Abstract
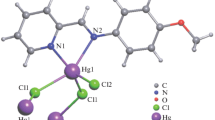
The title complex has been prepared using (4-fluoro-phenyl)-pyridin-2-ylmethylene-amine bidentate Schiff base ligand (L) and HgCl2. The Hg(II) center in the title compound, [HgCl2(C12H9FN2)], is covalently bonded to three Cl atoms and two N atoms of the diimine ligand. Mercury ion mononuclear fragment that forms a polymeric structure due to bridging chloride ligands is in a distorted trigonal-bipyramidal coordination environment formed by the bis-chelating N-heterocyclic ligand, two bridging Cl ligands and one terminal Cl ligand. One of the bridging Hg–Cl bonds is significantly longer than the other. In the crystal, weak C–H⋯Cl hydrogen bonds are observed in addition to π–π stacking interactions. It crystallizes in the orthorhombic space group P212121 with a = 7.2248(3) Å, b = 8.7713(3) Å, c = 20.0217(7) Å, α = 90.00°, β = 90.00°, γ = 90.00°, V = 1268.79(8) Å3, R1 = 0.023 and wR2 = 0.062 (I > 2σ(I)]. The antibacterial activities of the complex, ligand and metal salt were tested against Gram-positive bacteria Staphylococcus aureus and Gram-negative bacteria Escherichia coli studied by disk diffusion method.


Similar content being viewed by others
REFERENCES
S. AziziTalouki, G. Grivani, P. Crochet, and V. Cadierno. Inorg. Chim. Acta, 2017, 456, 142. https://doi.org/10.1016/j.ica.2016.10.041
I. Gonul, M. Kose, G. Ceyhan, and S. Serin. Inorg. Chim. Acta, 2016, 453, 522. https://doi.org/10.1016/j.ica.2016.09.004
A. Bhattacharyya, B. NathGhosh, K. Rissanen, and S. Chattopadhyay. Polyhedron, 2016, 117, 138. https://doi.org/10.1016/j.poly.2016.04.037
S. C. Bhatia, J. M. Bindlish, A. R. Saini, and P. C. Jain. J. Chem. Soc., Dalton Trans., 1981, 1773. https://doi.org/10.1039/dt9810001773
J. Costamagna, J. Vargas, R. Latorre, A. Alvarado, and G. Mena. Coord. Chem. Rev, 1992, 119, 67. https://doi.org/10.1016/0010-8545(92)80030-U
B. L. Vallee and D. S. Auld. Acc. Chem. Res., 1993, 26, 543. https://doi.org/10.1021/ar00034a005
J. W. Shin, J. M. Bae, Ch. Kim, and K. S. Min. Inorg. Chem., 2013, 52, 2265. https://doi.org/10.1021/ic302375z
H. Abu Ali, M. D. Darawsheh, and E. Rappocciolo. Polyhedron, 2013, 61, 235-241. https://doi.org/10.1016/j.poly.2013.06.015
Sh. Geranmayeh, A. Abbasi, A. H. Zarnani, and M. Y. Skripkin. Polyhedron, 2013, 61, 6. https://doi.org/10.1016/j.poly.2013.05.030
A. Lalegani, M. KhalediSardashti, H. Salavati, A. Asadi, R. Gajda, and K. Wozniak. J. Mol. Struct., 2016, 1108, 288. https://doi.org/10.1016/j.molstruc.2015.12.017
L. Tahmasbi, T. Sedaghat, H. Motamedi, and M. Kooti. J. Solid State Chem., 2018, 258, 517. https://doi.org/10.1016/j.jssc.2017.11.015
M. Azarkish, A. Akbari, T. Sedaghat, and J. Simpson. J. Mol. Struct, 2018, , 34. https://doi.org/10.1016/j.molstruc.2017.11.080
G. Q. Hu, X. K. Wu, and G. Q. Wang. Chin. Chem. Lett, 2012, 23, 515.
M. Sonmez, M. Celebi, and I. Berber. Eur. J. Med. Chem, 2010, 45, 1935. https://doi.org/10.1016/j.ejmech.2010.01.035
K. Venkatesan, V. S. V. Satyanarayana, and A. Sivakumar. J. Chin. Chem. Soc., 2011, 58, 583. https://doi.org/10.1002/jccs.201190091
S. R. Collinson and D. E. Fenton. Coord. Chem. Rev., 1996, 148, 19. https://doi.org/10.1016/0010-8545(95)01156-0
T. Sedaghat, M. Monajemzadeh, and H. Motamedi. J. Coord. Chem., 2011, 64, 3169. https://doi.org/10.1080/00958972.2011.616197
T. Sedaghat, M. Naseh, H. R. Khavasi, and H. Motamedi. Polyhedron, 2012, 33(1), 435-440. https://doi.org/10.1016/j.poly.2011.12.007
S. Dehghanpour and A. Mahmoudi. Main Group Chem., 2007, 6, 121. https://doi.org/10.1080/10241220801889025
Z. Otwinowski and W. Minor. In: Macromolecular Crystallography, Part A. / Eds. C. W. Carter and R. M. Sweet. New York: Academic, 1997, 307. https://doi.org/10.1016/S0076-6879(97)76066-X
R. H. Blessing. Acta Crystallogr., Sect. A, 1995, 51, 33. https://doi.org/10.1107/S0108767394005726
G. M. Sheldrick. Acta Crystallogr., Sect. A, 2008, 64, 112. https://doi.org/10.1107/S0108767307043930
A. W. Addison and T. N. Rao. J. Chem. Soc., Dalton Trans., 1984, 1349. https://doi.org/10.1039/DT9840001349
S. Salehzadeh, M. Khalaj, S. Dehghanpour, and A. Mahmoudi. J. Iran. Chem. Soc., 2013, 10, 921. https://doi.org/10.1007/s13738-013-0229-1
W. L. Drew, A. L. Barry, R. O, Toole, and J. C. Sherris. Appl. Environ. Microbiol., 1972, 24, 240. https://doi.org/10.1128/am.24.2.240-247.1972
N. S. Gwaram, H. M. Ali, H. Khaledi, and M. A. Abdulla. Molecules, 2012, 17, 5952. https://doi.org/10.3390/molecules17055952
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interests.
Additional information
Text © The Author(s), 2022, published in Zhurnal Strukturnoi Khimii, 2022, Vol. 63, No. 11, 101498.https://doi.org/10.26902/JSC_id101498
Rights and permissions
About this article
Cite this article
Aminian, M., Khalaj, M. SYNTHESIS, SPECTRAL CHARACTERIZATION, CRYSTAL STRUCTURE AND ANTIBACTERIAL ACTIVITY OF DICHLORO-{2-[(4-FLUOROPHENYL) IMINOMETHYL]PYRIDINE-κ2N,N′}MERCURY(II). J Struct Chem 63, 1797–1801 (2022). https://doi.org/10.1134/S0022476622110099
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476622110099