Abstract
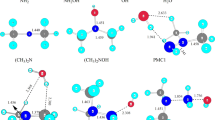
Chemisorption of C2H2 gas on the C20 bowl was studied at the LC-wPBE/6-311G(d,p) level of theory. Various models of chemisorption of C2H2 gas on the C20 bowl were considered. The illustration of chemisorption was performed using the energy decomposition analysis. Variations in the frontier orbitals energies and structural parameters were also considered. The electrophilicity-based charge transfer was used to illustrate charge transfer between fragments. Thermochemical parameters of this process were revealed.




Similar content being viewed by others
REFERENCES
A. N. R. Bos, E. S. Bootsma, F. Foeth, H. W. J. Sleyster, and K. R. Westerterp. Chem. Eng. Process., 1993, 32, 53. https://doi.org/10.1016/0255-2701(93)87006-G
A. S. Rad and K. Ayub. Mater. Chem. Phys., 2017, 194, 337. https://doi.org/10.1016/j.matchemphys.2017.04.002
A. S. Rad. Synth. Met., 2016, 211, 115. https://doi.org/10.1215/00382876-3425035
C. G. P. M. Bernardo and J. A. N. F. Gomes. In: Theoretical Aspects of Heterogeneous Catalysis: Progress in Theoretical Chemistry and Physics, Vol. 8 / Eds. W. N. Lipscomb, I. Prigogine, J. Maruani, S. Wilson, H. Ågren, D. Avnir, J. Cioslowski, R. Daudel, E. K. U. Gross, W. F. Gunsteren, K. Hirao, I. Hubač, M. P. Levy, G. L. Malli, R. McWeeny, P. G. Mezey, M. A. C. Nascimento, J. Rychlewski, S. D. Schwartz, Y. G. Smeyers, S. Suhai, O. Tapia, P. R. Taylor, and R. G. Woolley. Springer Science, 2001, 217. https://doi.org/10.1007/0-306-47667-3_9
Z. Kazemi, R. Ghiasi, and S. Jamehbozorgi. J. Nanoanal., 2019, 6, 121.
R. Ghiasi, F. Hadi, and A. H. Hakimyuon. J. Appl. Chem. Res., 2014, 8, 55.
P. Bechthold, M. Sandoval, E. A. González, G. Brizuela, A. Bonivardi, and P. V. Jasen. J. Phys. Chem. C, 2015, 119, 18229. https://doi.org/10.1021/acs.jpcc.5b04214
M. D. Esrafili and L. Dinparast. J. Phys. Chem. Solids, 2018, 117, 42. https://doi.org/10.1016/j.jpcs.2018.02.022
Y. Zhou, W. Sun, W. Chu, J. Zheng, X. Gao, X. Zhou, and Y. Xue. Appl. Surf. Sci., 2018, 435, 521. https://doi.org/10.1016/j.apsusc.2017.11.138
R. S. Shamsiev, F. O. Danilov, and T. A. Morozova. Russ. Chem. Bull., 2017, 66, 401. https://doi.org/10.1007/s11172-017-1747-2
P. Matczak. J. Struct. Chem., 2013, 54(1), 40. https://doi.org/10.1134/S002247661301006X
J. W. Medlin and M. D. Allendorf. J. Phys. Chem. B, 2003, 107, 217. https://doi.org/10.1021/jp026555t
A. Rezaei, R. Ghiasi, and A. Marjani. J. Nanostruct. Chem., 2020, 10, 179. https://doi.org/10.1007/s40097-020-00340-2
B. Chan. J. Phys. Chem. A, 2020, 124, 6688. https://doi.org/10.1021/acs.jpca.0c04732
L. Fulcheri, Y. Schwob, F. Fabry, G. Flamant, L. Chibante, and D. Laplaze. Carbon, 2000, 38, 797. https://doi.org/10.1016/S0008-6223(99)00153-0
L. D. Lamb and D. R. Huffman. J. Phys. Chem. Solids, 1993, 54, 1635. https://doi.org/10.1016/0022-3697(93)90277-X
H. Takehara, M. Fujiwara, M. Arikawa, M. D. Diener, and J. M. Alford. Carbon, 2005, 43, 311. https://doi.org/10.1016/j.carbon.2004.09.017
C. Crowley, R. Taylor, H. W. Kroto, D. R. Walton, P.-C. Cheng, and L. T. Scott. Synth. Met., 1996, 77, 17. https://doi.org/10.1016/0379-6779(96)80048-8
R. Dubrovsky, V. Bezmelnitsyn, and A. Eletskii. Carbon, 2004, 42, 1063. https://doi.org/10.1016/j.carbon.2003.12.010
M. C. Padole and P. A. Deshpande. J. Phys. Chem. C, 2016, 120, 12654. https://doi.org/10.1021/acs.jpcc.6b03747
F. Zhao, Y. Wang, M. Zhu, and L. Kang. RSC Adv., 2015, 5, 56348. https://doi.org/10.1039/C5RA08266H
Y. Wang and L. Kang. Catalysts, 2020, 10, 115. https://doi.org/10.3390/catal10010115
K. D. Sattler. Handbook of Nanophysics: Clusters and Fullerenes. CRC, 2010.
P. Pyykkö, C. Wang, M. Straka, and J. Vaara. Phys. Chem. Chem. Phys., 2007, 9, 2954. https://doi.org/10.1039/b704695b
H. Prinzbach, A. Weiler, P. Landenberger, F. Wahl, J. Wörth, L. T. Scott, M. Gelmont, D. Olevano, and B. Issendorff. Nature, 2000, 407, 60. https://doi.org/10.1038/35024037
H. Prinzbach, F. Wahl, W. A. P. Landenberger, J. Woerth, L. T. Scott, M. Gelmont, D. Olevano, F. Sommer, and B. Issendorff. Chem. – Eur. J., 2006, 12, 6268. https://doi.org/10.1002/chem.200501611
W. An, Y. Gao, S. Bulusu, and X. C. Zeng. J. Chem. Phys., 2005, 122, 204109. https://doi.org/10.1063/1.1903946
C. Zhang, W. Sun, and Z. Cao. J. Chem. Phys., 2007, 126, 144306. https://doi.org/10.1063/1.2716642
A. K. Ray and M. N. Huda. J. Comput. Theor. Nanosci., 2006, 3, 315. https://doi.org/10.1166/jctn.2006.3014
F. N. Ajeel. Chin. J. Phys., 2017, 55, 2134. https://doi.org/10.1016/j.cjph.2017.05.031
R. Ghiasi and A. Valizadeh. Main Group Chem., 2022, 21, 43. https://doi.org/10.3233/MGC-210076
Ö. Alver, C. Parlak, and P. Ramasami. Adsorpt. Sci. Technol., 2018, 36, 788. https://doi.org/10.1177/0263617417722922
L. Zoppi, L. Martin-Samos, and K. K. Baldridge. Acc. Chem. Res., 2014, 47, 3310. https://doi.org/10.1021/ar5001132
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalman, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox. Gaussian09, Revision A.02. Wallingford, CT: Gaussian, 2009.
R. Krishnan, J. S. Binkley, R. Seeger, and J. A. Pople. J. Chem. Phys., 1980, 72, 650. https://doi.org/10.1063/1.438955
A. D. McLean and G. S. Chandler. J. Chem. Phys., 1980, 72, 5639. https://doi.org/10.1063/1.438980
L. A. Curtiss, M. P. McGrath, J.-P. Blandeau, N. E. Davis, R. C. Binning, and J. L. Radom. J. Chem. Phys., 1995, 103, 6104. https://doi.org/10.1063/1.470438
O. A. Vydrov, G. E. Scuseria, and J. P. Perdew. J. Chem. Phys., 2007, 126, 154109. https://doi.org/10.1063/1.2723119
O. A. Vydrov and G. E. Scuseria. J. Chem. Phys., 2006, 125, 234109. https://doi.org/10.1063/1.2409292
Y. Tawada, T. Tsuneda, S. Yanagisawa, T. Yanai, and K. Hirao. J. Chem. Phys., 2004, 120, 8425. https://doi.org/10.1063/1.1688752
O. A. Vydrov, J. Heyd, A. Krukau, and G. E. Scuseria. J. Chem. Phys., 2006, 125, 074106. https://doi.org/10.1063/1.2244560
I. H. Nayyar, E. R. Batista, S. Tretiak, A. Saxena, D. L. Smith, and R. L. Martin. J. Chem. Theory Comput., 2013, 9, 1144. https://doi.org/10.1021/ct300837d
I. H. Nayyar, E. R. Batista, S. Tretiak, A. Saxena, D. L. Smith, and R. L. Martin. J. Phys. Chem. Lett., 2011, 2, 566. https://doi.org/10.1021/jz101740w
C. Møller and M. S. Plesset. Phys. Rev. B, 1934, 46, 0618. https://doi.org/10.1103/PhysRev.46.618
M. Head-Gordon, J. A. Pople, and M. J. Frisch. Chem. Phys. Lett., 1988, 153, 503. https://doi.org/10.1016/0009-2614(88)85250-3
The Challenge of d and f Electrons / Ed. M. C. Zerner. Washington, D.C.: ACS, 1989.
M. J. Frisch, M. Head-Gordon, and J. A. Pople. Chem. Phys. Lett., 1990, 166, 275. https://doi.org/10.1016/0009-2614(90)80029-D
M. J. Frisch, M. Head-Gordon, and J. A. Pople. Chem. Phys. Lett., 1990, 166, 281. https://doi.org/10.1016/0009-2614(94)00116-2
M. Head-Gordon and T. Head-Gordon. Chem. Phys. Lett., 1994, 220, 122. https://doi.org/10.1016/0009-2614(94)00116-2
T. Clark, J. Chandrasekhar, G. W. Spitznagel, and P. V. R. Schleyer. J. Comput. Chem., 1983, 4, 294. https://doi.org/10.1002/jcc.540040303
D. Feller. J. Comput. Chem., 1996, 17, 1571. https://doi.org/10.1002/(SICI)1096-987X(199610)17:13%3C1571::AID-JCC9%3E3.0.CO;2-P
B. P. Pritchard, D. Altarawy, B. Didier, T. D. Gibsom, and T. L. Windus. J. Chem. Inf. Model., 2019, 59, 4814. https://doi.org/10.1021/acs.jcim.9b00725
K. L. Schuchardt, B. T. Didier, T. Elsethagen, L. Sun, V. Gurumoorthi, J. Chase, J. Li, and T. L. Windus. J. Chem. Inf. Model., 2007, 47, 1045. https://doi.org/10.1021/ci600510j
T. Lu and F. Chen. J. Mol. Graphics Modell., 2012, 38, 314. https://doi.org/10.1016/j.jmgm.2012.07.004
T. Lu and F. Chen. J. Comput. Chem., 2012, 33, 580. https://doi.org/10.1002/jcc.22885
M. E. Foster and B. M. Wong. J. Chem. Theory Comput., 2012, 8, 2682. https://doi.org/10.1021/ct300420f
L. N. Anderson, M. B. Oviedo, and B. M. Wong. J. Chem. Theory Comput., 2017, 13, 1656. https://doi.org/10.1021/acs.jctc.6b01249
J. Padmanabhan, R. Parthasarathi, V. Subramanian, and P. K. Chattaraj. J. Phys. Chem. A, 2007, 111, 1358. https://doi.org/10.1021/jp0649549
R. G. Pearson. J. Org. Chem., 1989, 54, 1430. https://doi.org/10.1021/jo00267a034
R. G. Parr and R. G. Pearson. J. Am. Chem. Soc., 1983, 105, 7512. https://doi.org/10.1021/ja00364a005
P. Geerlings, F. D. Proft, and W. Langenaeker. Chem. Rev., 2003, 103, 1793. https://doi.org/10.1021/cr990029p
R. G. Parr, L. Szentpály, and S. Liu. J. Am. Chem. Soc., 1999, 121, 1922. https://doi.org/10.1021/ja983494x
R. G. Parr and W. Yang. Density Functional Theory of Atoms and Molecules. Oxford, New York: Oxford University Press, 1989.
K. B. Wiberg. Tetrahedron, 1968, 24, 1083. https://doi.org/10.1016/0040-4020(68)88057-3
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interests.
Additional information
Text © The Author(s), 2022, published in Zhurnal Strukturnoi Khimii, 2022, Vol. 63, No. 10, 99318.https://doi.org/10.26902/JSC_id99318
Rights and permissions
About this article
Cite this article
Sarghein, M.G., Ghiasi, R. & Baniyaghoob, S. CHEMISORPTION OF C2H2 ON C20 BOWL: A COMPUTATIONAL INVESTIGATION. J Struct Chem 63, 1600–1609 (2022). https://doi.org/10.1134/S0022476622100067
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476622100067