Abstract
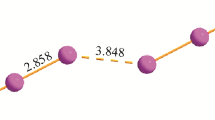
The structures of 1,4-diiodo-2,5-dimethylbenzene (1) and 1,3,4,6-tetraiodo-2,5-dimethylbenzene (2) are determined by single crystal X-ray diffraction. The packing motif in 2 is mainly determined by π-stacking and the system of halogen⋯halogen contacts between the stacks. In the structure of 1, it is determined by the presence of both π-stacking and the interaction between σ-holes of iodine atoms and the π-system of the neighboring molecules. The thermal stability of 1 and 2 is studied and the optical spectra are measured.





Similar content being viewed by others
REFERENCES
G. L. Ciamician and M. Dennstedt. Ber. Dtsch. Chem. Ges., 1882, 15, 2579-2585. https://doi.org/10.1002/cber.188201502221
E. Rupp. Ber. Dtsch. Chem. Ges., 1896, 29, 1625-1634. https://doi.org/10.1002/cber.18960290293
M. A. Bondarenko, A. S. Novikov, P. A. Abramov, I. F. Sakhapov, M. N. Sokolov, and S. A. Adonin. J. Mol. Struct., 2021, 1230, 129931. https://doi.org/10.1016/j.molstruc.2021.129931
E. V. Bartashevich and V. G. Tsirelson. Russ. Chem. Rev., 2014, 83, 1181-1203. https://doi.org/10.1070/RCR4440
E. V. Bartashevich, E. A. Grigoreva, I. D. Yushina, L. M. Bulatova, and V. G. Tsirelson. Russ. Chem. Bull., 2017, 66, 1345-1356. https://doi.org/10.1007/s11172-017-1898-1
G. Cavallo, P. Metrangolo, R. Milani, T. Pilati, A. Priimagi, G. Resnati, and G. Terraneo. Chem. Rev., 2016, 116, 2478-2601. https://doi.org/10.1021/acs.chemrev.5b00484
D. M. Ivanov, M. A. Kinzhalov, A. S. Novikov, I. V. Ananyev, A. A. Romanova, V. P. Boyarskiy, M. Haukka, and V. Y. Kukushkin. Cryst. Growth Des., 2017, 17, 1353-1362. https://doi.org/10.1021/acs.cgd.6b01754
A. S. Mikherdov, A. S. Novikov, V. P. Boyarskiy, and V. Y. Kukushkin. Nat. Commun., 2020, 11, 2921. https://doi.org/10.1038/s41467-020-16748-x
A. A. Eliseeva, D. M. Ivanov, A. S. Novikov, A. V. Rozhkov, I. V. Kornyakov, A. Y. Dubovtsev, and V. Y. Kukushkin. Dalton Trans., 2020, 49, 356-367. https://doi.org/10.1039/C9DT04221K
A. S. Novikov, D. M. Ivanov, Z. M. Bikbaeva, N. A. Bokach, and V. Y. Kukushkin. Cryst. Growth Des., 2018, 18, 7641-7654. https://doi.org/10.1021/acs.cgd.8b01457
Y. V. Matveychuk, M. V. Ilkaeva, E. A. Vershinina, V. I. Batalov, R. S. Morozov, and E. V. Bartashevich. J. Mol. Struct., 2016, 1119, 227-234. https://doi.org/10.1016/j.molstruc.2016.04.072
I. Yushina, N. Tarasova, D. Kim, V. Sharutin, and E. Bartashevich. Crystals, 2019, 9, 506. https://doi.org/10.3390/cryst9100506
U. Dabranskaya, D. M. Ivanov, A. S. Novikov, Y. V. Matveychuk, N. A. Bokach, and V. Y. Kukushkin. Cryst. Growth Des., 2019, 19, 1364-1376. https://doi.org/10.1021/acs.cgd.8b01757
M. A. Kinzhalov, M. V. Kashina, A. S. Mikherdov, E. A. Mozheeva, A. S. Novikov, A. S. Smirnov, D. M. Ivanov, M. A. Kryukova, A. Y. Ivanov, S. N. Smirnov, V. Y. Kukushkin, and K. V. Luzyanin. Angew. Chem., Int. Ed., 2018, 57, 12785-12789. https://doi.org/10.1002/anie.201807642
Y. V. Torubaev and I. V. Skabitsky. Z. Kristallogr. – Cryst. Mater., 2020, 235, 599-607. https://doi.org/10.1515/zkri-2020-0064
Y. V. Torubaev, I. V. Skabitsky, and K. A. Lyssenko. Mendeleev Commun., 2020, 30, 580-582. https://doi.org/10.1016/j.mencom.2020.09.009
A. S. Novikov, D. M. Ivanov, M. S. Avdontceva, and V. Y. Kukushkin. CrystEngComm, 2017, 19, 2517-2525. https://doi.org/10.1039/C7CE00346C
E. Albright, J. Cann, A. Decken, and S. Eisler. CrystEngComm, 2017, 19, 1024-1027. https://doi.org/10.1039/C6CE02339H
A. A. Eliseeva, D. M. Ivanov, A. S. Novikov, and V. Y. Kukushkin. CrystEngComm, 2019, 21, 616-628. https://doi.org/10.1039/C8CE01851K
S. V. Baykov, S. I. Filimonov, A. V. Rozhkov, A. S. Novikov, I. V. Ananyev, D. M. Ivanov, and V. Y. Kukushkin. Cryst. Growth Des., 2020, 20, 995-1008. https://doi.org/10.1021/acs.cgd.9b01334
P. A. Demakov, S. A. Sapchenko, D. G. Samsonenko, D. N. Dybtsev, and V. P. Fedin. Russ. Chem. Bull., 2018, 67, 490-496. https://doi.org/10.1007/s11172-018-2098-3
V. A. Bolotov, K. A. Kovalenko, D. G. Samsonenko, X. Han, X. Zhang, G. L. Smith, L. J. MCormick, S. J. Teat, S. Yang, M. J. Lennox, A. Henley, E. Besley, V. P. Fedin, D. N. Dybtsev, and M. Schröder. Inorg. Chem., 2018, 57, 5074-5082. https://doi.org/10.1021/acs.inorgchem.8b00138
A. V. Marakulin, A. A. Lysova, D. G. Samsonenko, P. V. Dorovatovskii, V. A. Lazarenko, D. N. Dybtsev, and V. P. Fedin. Russ. Chem. Bull., 2020, 69, 360-368. https://doi.org/10.1007/s11172-020-2768-9
A. A. Sapianik, E. N. Zorina-Tikhonova, M. A. Kiskin, D. G. Samsonenko, K. A. Kovalenko, A. A. Sidorov, I. L. Eremenko, D. N. Dybtsev, A. J. Blake, S. P. Argent, M. Schröder, and V. P. Fedin. Inorg. Chem., 2017, 56, 1599-1608. https://doi.org/10.1021/acs.inorgchem.6b02713
K. deKrafft, Z. Xie, G. Cao, S. Tran, L. Ma, O. Zhou, and W. Lin. Angew. Chem., Int. Ed., 2009, 48, 9901-9904. https://doi.org/10.1002/anie.200904958
M. A. Rahman, F. Shito, and T. Kitamura. Synthesis, 2010, 27-29. https://doi.org/10.1055/s-0029-1217072
E. R. T. Tiekink. CrystEngComm, 2021, 23, 904-928. https://doi.org/10.1039/D0CE01677B
Funding
The work was supported by the Russian Science Foundation (grant No. 21-73-20019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interests.
Additional information
Russian Text © The Author(s), 2022, published in Zhurnal Strukturnoi Khimii, 2022, Vol. 63, No. 4, pp. 504-509.https://doi.org/10.26902/JSC_id90869
Rights and permissions
About this article
Cite this article
Rajakumar, K., Sharutin, V.V., Adonin, S.A. et al. DI- AND TETRAIODOXYLENES: STRUCTURE AND FEATURES OF NON-COVALENT INTERACTIONS IN A SOLID STATE. J Struct Chem 63, 620–625 (2022). https://doi.org/10.1134/S0022476622040138
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476622040138