Abstract
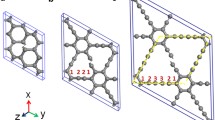
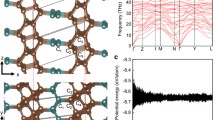
Thermal stability of a recently proposed carbon allotrope based on Ladenburg benzene, superprismane, is studied by the molecular dynamics method. It is shown that the superprismane structure destroys at 500 K and its lattice transforms from the hexagonal one to the tetragonal one. The resulting crystal with a tetragonal lattice possesses high strength and is stable up to 1000 K. It has channels with a diameter of ~1 nm allowing for the diffusion of small molecules along certain crystallographic directions. It is shown that the penetration of hydrogen, nitrogen, water, and carbon monoxide molecules into the channel is accompanied by energy release, i.e. the molecules are “pulled” inside the channel.



Similar content being viewed by others
REFERENCES
K. P. Katin and M.M. Maslov. Mol. Simul., 2018, 44(9), 703-707. https://doi.org/10.1080/08927022.2018.1434720
M. A. Forman and W. P. Dailey. Org. Prep. Proced. Int., 1994, 26(3), 291-320. https://doi.org/10.1080/00304949409458427
T. J. Katz and N. Acton. J. Am. Chem. Soc., 1973, 95(8), 2738-2739. https://doi.org/10.1021/ja00789a084
P. E. Eaton and T. W. Cole. J. Am. Chem. Soc., 1964, 86(15), 3157/3158. https://doi.org/10.1021/ja01069a041
P. E. Eaton, Y. S. Or, and S. J. Branca. J. Am. Chem. Soc., 1981, 103(8), 2134-2136. https://doi.org/10.1021/ja00398a062
K. P. Katin and M. M. Maslov. Adv. Condens. Matter Phys., 2015, 1-6. https://doi.org/10.1155/2015/754873
H. A. Favre and W. H. Powell. Nomenclature of Organic Chemistry. Royal Society of Chemistry, 2013, Ch. P-2. https://doi.org/10.1039/9781849733069-00130
K. P. Katin, V. S. Prudkovskiy, and M. M. Maslov. Phys. E, 2016, 81, 1-6. https://doi.org/10.1016/j.physe.2016.02.010
H. Xing, S. D. Houston, X. Chen, S. Ghassabian, T. Fahrenhorst-Jones, A. Kuo, C. P. Murray, K. Conn, K. N. Jaeschke, D. Jin, C. Pasay, P. V. Bernhardt, J. M., Burns, J. Tsanaktsidis, G. P. Savage, G. M. Boyle, J. J. De Voss, J. McCarthy, G. H. Walter, T. H. J. Burne, M. T. Smith, J.-K. Tie, and C. M Williams. Chem. - Eur. J., 2019, 25(11), 2729-2734. https://doi.org/10.1002/chem.201806277
M. M. Maslov, K. P. Katin, A. I. Avkhadieva, and A. I. Podlivaev. Russ. J. Phys. Chem. B, 2014, 8(2), 152-157. https://doi.org/10.1134/s1990793114020195
K. Srinivasu and S. K. Ghosh. J. Phys. Chem. C, 2012, 116(47), 25015-25021. https://doi.org/10.1021/jp3104479
M. M. Maslov and K. P. Katin. Chem. Phys. Lett., 2016, 644, 280-283. https://doi.org/10.1016/j.cplett.2015.12.022
K. Lonsdale, H. J. Milledge, and E. Nave. Mineral. Mag. J. Mineral. Soc., 1959, 32(246), 185-201. https://doi.org/10.1180/minmag.1959.032.246.02
V. A. Greshnyakov, E. A. Belenkov, and V. M. Berezin. Crystal Structure and Properties of Diamond-Like Carbon Phase (Kristallicheskaya Struktura i Svoistva Uglerodnykh Almazopodobnykh Faz). Chelyabinsk, Russia: South-Ural State University, 2012. [In Russian]
D. S. Lisovenko, J. A. Baimova, L. Kh. Rysaeva, V. A. Gorodtsov, A. I. Rudskoy, and S. V. Dmitriev. Phys. Status Solidi B, 2016, 253(7), 1295-1302. https://doi.org/10.1002/pssb.201600049
K. A. Krylova, Yu. A. Baimova, S. V. Dmitriev, and R. R. Mulyukov. Phys. Solid State, 2016, 58(2), 394-401. https://doi.org/10.1134/s1063783416020189
J. A. Baimova, L. Kh. Rysaeva, and A. I. Rudskoy. Diamond Relat. Mater., 2018, 81, 154-160. https://doi.org/10.1016/j.diamond.2017.12.001
N. V. R. Nulakani and V. Subramanian. Chem. Phys. Lett., 2019, 715, 29-33. https://doi.org/10.1016/j.cplett.2018.11.006
S. A. Shostachenko, M. M. Maslov, V. S. Prudkovskii, and K. P. Katin. Phys. Solid State, 2015, 57(5), 1023-1027. https://doi.org/10.1134/s1063783415050261
M. M. Maslov, A. I. Podlivaev, and K. P. Katin. Mol. Simul., 2015, 42(4), 305-311. https://doi.org/10.1080/08927022.2015.1044453
K. P. Katin, K. S. Grishakov, A. I. Podlivaev, and M. M. Maslov. J. Chem. Theor. Comput., 2020, 16(4), 2065-2070. https://doi.org/10.1021/acs.jctc.9b01229
M. M. Maslov and K. P. Katin. Chem. Phys., 2011, 387(1-3), 66-68. https://doi.org/10.1016/j.chemphys.2011.06.035
K. P. Katin and M. M. Maslov. Russ. J. Phys. Chem. B, 2011, 5(5), 770-779. https://doi.org/10.1134/s1990793111090181
O. R. Rakhmanova and A. E. Galashev. Russ. J. Phys. Chem. A, 2017, 91(5), 921-925. https://doi.org/10.1134/s003602441705020x
Y. Galashev and O. R. Rakhmanova. Phys. E, 2021, 126, 114446. https://doi.org/10.1016/j.physe.2020.114446
V. Savin and O. I. Savina. Fiz. Tverd. Tela, 2021, 63(4), 564. https://doi.org/10.21883/ftt.2021.04.50726.239
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author declares that he has no conflict of interests.
Additional information
Russian Text © The Author(s), 2021, published in Zhurnal Strukturnoi Khimii, 2021, Vol. 62, No. 10, pp. 1621-1626.https://doi.org/10.26902/JSC_id80792
Rights and permissions
About this article
Cite this article
Kuandykov, R.R. STRUCTURE AND PROPERTIES OF A NEW CARBON ALLOTROPE BASED ON LADENBURG BENZENE. J Struct Chem 62, 1516–1521 (2021). https://doi.org/10.1134/S002247662110005X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002247662110005X