Abstract
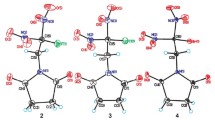
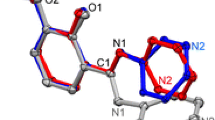
Diethylaminovinyl derivatives of halogenated 1,4-quinones (enaminoquinones) are produced by the interaction of halogenated 1,4-quinones with N,N-diethyl-N-vinylamine obtained in situ from Et2NH and MeCHO. Molecular and crystal structures of enaminoquinones halogenated in the quinone fragment are determined. It is shown that in the solid phase, atoms of the entire push-pull system of enaminoquinones lie practically in one plane.






Similar content being viewed by others
REFERENCES
H. S. Nalwa. Adv. Mater., 1993, 5, 341–358.
J. L. Brédas, C. Adant, P. Tackx, and A. Persoons. Chem. Rev., 1994, 94, 243–278.
S. D. Bella, I. L. Fragalá, M. A. Ratner, and T. J. Marks. J. Am. Chem. Soc., 1993, 115, 682–686.
R. Pilot and R. Bozio. Phys. Chem. Chem. Phys., 2011, 13, 230–239.
B. I. Kidyarov, V. V. Atuchin, and N. V. Pervukhina. J. Struct. Chem., 2010, 51(6), 1119–1125.
R. M. Metzger and C. A. Panetta. New J. Chem., 1991, 15, 209–221.
A. Aviram. Molecular Electronic Science and Technology. Engineering Foundation: New York, 1989.
S .R. Marder and J. W. Perry. Adv. Mater., 1993, 5, 804–815.
J. A. Cowan, J. K. M. Sanders, G. S. Beddard, and R. J. Harrison. J. Chem. Soc., Chem. Commun., 1987, 55–58.
D. Gust and T. A. Moore. Science, 1989, 244, 35–41.
H. Tamiaki and K. Maruyama. J. Chem. Soc. Perkin Trans. 1, 1992, 2431–2435.
B. Illescas, N. Martín, J. L. Segura, and C. Seoane. J. Org. Chem., 1995, 60, 5643–5650.
E. P. Fokin and E. P. Prudchenko. Zh. Org. Khim., 1970, 6, 91–93.
E. P. Fokin and E. P. Prudchenko. Zh. Org. Khim., 1970, 6, 1251–1255.
D. Buckley, S. Dunstan, and H. B. Henbest. J. Chem. Soc., 1957, 4880–4891.
L. I. Goryunov, N. M. Troshkova, G. A. Nevinskii, and V. D. Shteingarts. Russ. J. Org. Chem., 2009, 45, 835–841.
M. Alnabari and S. Bittner. Synthesis, 2000, 8, 1087–1090.
SADABS, v. 2008-1. Bruker AXS: Madison, WI, USA, 2008.
G. M. Sheldrick. Acta Crystallogr., Sect. C, 2015, 71, 3–8.
A. L. Spek. J. Appl. Crystallogr., 2003, 36, 7–13.
C. F. Macrae, P. R. Edgington, P. McCabe, E. Pidcock, G. P. Shields, R. Taylor, M. Towler, and J. van de Stree. J. Appl. Crystallogr., 2006, 39, 453–457.
H.-J. Kallmayer and B. Thierfelder. Pharmazie, 2002, 57, 456–459.
D. Buckley, H. B. Henbest, and P. Slade. J. Chem. Soc., 1957, 4891–4900.
E. P. Fokin, E. P. Prudchenko, and Yu. T. Glushkov. Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, 1970, 3, 123–125.
F. H. Allen, O. Kenard, D. G. Watson, L. Bramer, A. G. Orpen, and R. Taylor. J. Chem. Soc., Perkin Trans. 2, 1987, S1–S19.
G. V. Noshchenko, N. F. Salivon, B. Zarychta, and V. V. Olijnyk. J. Struct. Chem., 2013, 54(1), 136–140.
A. Krivokapic and H. L. Anderson. Acta Crystallogr., Sect. E, 2002, 58, o259–o260.
N. Batenko, O. Popova, S. Belyakov, and R. Valters. Chem. Heterocycl. Compd., 2012, 48(6), 955–959.
T. V. Rybalova and I. Yu. Bagryanskaya. J. Struct. Chem., 2009, 50(4), 767–779.
Funding
The study was supported by the Russian Science Foundation (project No. 17-73-10238).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Zhivetyeva, S.I., Zayakin, I.A., Bagryanskaya, I.Y. et al. 2-DIETHYLAMINOVINYL DERIVATIVES OF HALOGENATED 1,4-QUINONES: SYNTHETIC AND STRUCTURAL ASPECTS. J Struct Chem 61, 1253–1259 (2020). https://doi.org/10.1134/S0022476620080107
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476620080107