Abstract
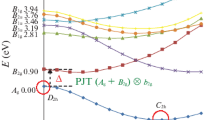
The instability of planar structures in [C2X4]− (X = H, F, Cl, Br) molecules due to the pseudo Jahn-Teller effect (PJTE) is investigated as an original PJTE study. Optimization and the following frequency calculations in these molecules illustrate that all of these compounds are unstable in high symmetry planar (with D2h symmetry) geometry and their structures are distorted to a lower C2h symmetry stable geometry. Moreover, the vibronic coupling interaction between 2B2g ground and 2Ag excited states via the (2B2g + 2Ag) ⊗B2g PJTE problem is the reason for the symmetry breaking phenomenon and non-planarity in those series. Natural bond analysis (NBO) is used for illustrating the strongest interaction and natural atomic charges of these structures.
Similar content being viewed by others
References
R. A. King, N. D. Pettigrew, and H. F. Schaefer. J. Chem. Phys., 1997, 107, 8536.
X.-J. Hou and M.-B. Huang. J. Phys. Chem. A, 2002, 106, 10655.
M. N. Paddon-Row, N. G. Rondan, K. N. Houk, and K. D. Jordan. J. Am. Chem. Soc., 1982, 104, 1143.
S. Merry and C. Thomson. Chem. Phys. Lett., 1981, 82, 373.
J. T. Wang and F. Williams. J. Am. Chem. Soc., 1981, 103, 2902.
R. I. McNeil, M. Shiotani, F. Williams, and M. B. Yim. Chem. Phys. Lett., 1977, 51, 433.
J. R. Morton, K. F. Preston, J. T. Wang, and F. Williams. Chem. Phys. Lett., 1979, 64, 71.
M. N. Paddon-Row, N. G. Rondan, K. N. Houk, and K. D. Jordan. J. Am. Chem. Soc., 1982, 104, 1143.
Topics in Molecular Organization and Engineering / Eds. A. Lund, M. Shiotani. Dordrecht: Springer Science and Business Media, 1991, 6.
I. B. Bersuker. The Jahn-Teller Effect. Cambridge University Press, 2006.
R. G. Pearson. Symmetry Rules for Chemical Reactions. Wiley-Interscience: New York, 1976.
A. R. Ilkhani. Russ. J. Phys. Chem. A, 2017, 91, 1743.
Y. Shen and J. J. BelBruno. J. Phys. Chem. A, 2005, 109, 512.
P. Garcia-Fernandez, J. A. Aramburu, M. Moreno, M. Zlatar, and M. G-Pavlovic. J. Chem. Theory Comput., 2014, 10, 1824.
X.-J. Feng, M. Zhang, L.-X. Zhao, H.-Y. Zhang, and Y.-H. Luo. Comput. Theor. Chem., 2014, 1029, 84.
A. R. Ilkhani. J. Mol. Struct., 2015, 1098, 21.
Y. Liu. Comput. Theor. Chem., 2014, 1044, 94.
F. Grein. Theor. Chem. Ace, 2014, 133, 1482.
Y. Kameoka, T. Sato, T. Koyama, K. Tanaka, and T. Kato. Chem. Phys. Lett., 2014, 598, 69.
A. R. Ilkhani. J. Theor. Comput. Chem., 2015, 14, 1550045.
N. N. Gorinchoy, I. I. Balan, and I. B. Bersuker. Comput. Theor. Chem., 2011, 976(2011), 113.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalman, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox. Revision A.02 ed., Gaussian, Inc.: Wallingford CT, 2009.
F. Weigend and R. Ahlrichs. Phys. Chem. Chem. Phys., 2005, 7, 3297.
C. C. J. Roothaan. Rev. Mod. Phys., 1951, 23, 69.
J. A. Pople and R. K. Nesbet. J. Chem. Phys., 1954, 22, 571.
R. McWeeny and G. Dierksen. J. Chem. Phys., 1968, 49, 4852.
J. B. Foresman, M. Head-Gordon, J. A. Pople, and M. J. Frisch. J. Phys. Chem. A, 1992, 96, 135.
A. E. Reed, L. A. Curtiss, and F. Weinhold. Chem. Rev., 1988, 88, 899.
E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, C. R. Landis, and F. Weinhold. Theoretical Chemistry Institute, University of Wisconsin, Madison, WI, 2013.
L. Pauling. The Nature of the Chemical Bond. 3rd ed. Cornell University Press: Ithaca, 1960.
P. W. Ayers and R. G. Parr. J. Am. Chem. Soc., 2000, 422, 2010.
R. G. Parr and P. K. Chattaraj. J. Am. Chem. Soc., 1991, 113, 1854.
R. G. Pearson. J. Chem. Educ., 1987, 64, 561.
R. G. Pearson. Acc. Chem. Res., 1993, 26, 250.
R. G. Pearson. J. Chem. Educ., 1999, 76, 267.
E. Chamorro, P. K. Chattaraj, and P. Fuentealba. J. Phys. Chem. A, 2003, 107, 7068.
R. Parthasarathi, M. Elango, V. Subramanian, and P. K. Chattaraj. Theor. Chem. Acc., 2005, 113, 257.
P. A. Christiansen and W. E. Palke. J. Chem. Phys., 1977, 67, 57.
V. F. Weisskopf. Science, 1975, 187, 605.
J. K. Badenhoop and F. Weinhold. Int. J. Quantum. Chem., 1999, 72, 269.
Author information
Authors and Affiliations
Corresponding author
Additional information
Text © The Author(s), 2019, published in Zhurnal Strukturnoi Khimii, 2019, Vol. 60, No. 5, pp. 770-778.
Rights and permissions
About this article
Cite this article
Mahmoudzadeh, G., Ghiasi, R. & Pasdar, H. Computational Investigation of the Pseudo Jahn–Teller Effect on the Structure and Chemical Properties of Perhaloethene Anions. J Struct Chem 60, 736–745 (2019). https://doi.org/10.1134/S0022476619050056
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476619050056