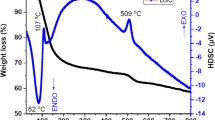
Abstract
We located multiple binding sites for doxycycline on DNA under physiological conditions, using spectroscopic methods and molecular modeling. Fourier-transformed infrared spectroscopy and UV-visible spectroscopy are used to determine the ligand intercalation and external binding modes, the binding constant, and the stability of doxycycline–DNA complexes in an aqueous solution. Spectroscopic evidence shows that the doxycycline (DOXY) complexation with DNA occurs via G–C and A–T, and a PO2 group with a binding constant K(DOXY–DNA) = 1.4×104 M–1. Uniform rare-earth gadolinium oxide (Gd2O3), as formed through a precipitation process using hexamine as template, are characterized using X-ray diffraction and scanning electron microscopy. Another aim of the study was to investigate the degradation of the DOXY antibiotic by nanosized Gd2O3 under ultraviolet irradiation. Various experimental parameters such as initial DOXY concentrations, initial Gd2O3 concentration, initial pH, reaction times are investigated. According to the results, this method can be good in the removal of DOXY.
Similar content being viewed by others
Change history
23 April 2019
In the original publication the affiliation was not correct. The correct affiliation is: Department of Chemistry, Central Tehran Branch, Islamic Azad University, Tehran, Iran.
06 May 2019
In the original publication the name of one of the authors had been misspelled and the authors had been listed in the wrong order. The correct spelling of the names of the authors and their order is: M. Masoudinia, A. Bagheri ghomi.
References
T. Onoda, T. Ono, D. K. Dhar, et al. J. Int. Du Cancer, 2006, 118, 1309–1315.
K. Son, S. Fujioka, T. Iida, et al. Anticancer Res., 2009, 29, 3995–4003.
Y. Charles Cao. J. Am. Chem. Soc., 2004, 126, 7456/7457.
K. Takahashi, S. Tazaki, J. Miyahara, Y. Karasawa, and N. Nimura. Nucl. Instrum. Methods Phys. Res., 1996, A377, 119.
G. Gunduz and I. Uslu. J. Nucl. Mater., 1996, 231, 113–118.
A. C. Faure, S. Dufort, V. Josserand, P. Perriat, J. L. Coll, and S. O. Roux. Small, 2009, 5, 2565–2575.
L. E. Van Vlerken and M. M. Amiji. Drug Delivery, 2006, 3, 205–216.
J. Ma, L. T. B. La, I. Zaman, Q. Meng, L. Luong, D. Ogilvie, and H. C. Kuan. Macromol. Mater. Eng., 2011, 296, 465–474.
P. Caravan, J. J. Ellison, T. J. McMurry, and R. B. Lauffer. Chem. Rev., 1999, 99, 2293–2352.
M. Shokouhimehr, E. S. Soehnlen, J. Hao, M. Griswold, C. Flask, X. Fan, J. P. Basilion, S. Basu, and S. D. Huang. J. Mater. Chem., 2010, 20, 5251–5259.
R. Lv, S. Gai, Y. Dai, N. Niu, F. He, and P. Yang. ACS. Appl. Mater. Interfaces, 2013, 5, 10806–10818.
Q. Ju, D. Tu, Y. Liu, R. Li, H. Zhu, J. Chen, Z. Chen, M. Huang, and X. Chen. J. Am. Chem. Soc., 2011, 134, 1323–1330.
H. K. Cho, H.–J. Cho, S. Lone, D.–D. Kim, J. H. Yeum, and I. W. Cheong. J. Mater. Chem., 2011, 21, 15486–15493.
L. B. T. La, Y. K. Leong, C. Leatherday, P. I. Au, K. J. Hayward, and L. C. Zhang. Colloids Surf., 2016, A501, 75–82.
M. A. Malik, M. Y. Wani, and M. A. Hashim. Arabian J. Chem., 2012, 5, 397–417.
D. Hari Prasad, H. R. Kim, J. S. Park, J. W. Son, B. K. Kim, H. W. Lee, and J. H. Lee. J. Alloys Compd., 2010, 495, 238–241.
I. Muneer, M. A. Farrukh, S. Javaid, and M. Shahid. Superlattices Microstruct., 2015, 77, 256–266.
N. Zhang, R. Yi, L. Zhou, G. Gao, R. Shi, G. Qiu, and X. Liu. Mater. Chem. Phys., 2009, 114, 160–167.
T. Tsuzuki, E. Pirault, and P. McCormick. Nanostruct. Mater., 1999, 11, 125–131.
T. Tsuzuki, W. T. Harrison, and P. G. McCormick. J. Alloys Compd., 1998, 281, 146–151.
R. Bazzi, M. Flores–Gonzalez, C. Louis, K. Lebbou, C. Dujardin, A. Brenier, W.Zhang, O. Tillement, E. Bernstein, and P. Perriat. J. Lumin, 2003, 102, 445–450.
J. L. Bridot, A. C. Faure, S. Laurent, C. Riviere, C. Billotey, B. Hiba, M. Janier, V. Josserand, J. L. Coll, and L. Vander. J. Am. Chem. Soc., 2007, 129, 5076–5084.
L. Faucher, M. L. Tremblay, J. Lagueux, Y. Gossuin, and M. A. Fortin. ACS. Appl. Mater. Interfaces, 2012, 4, 4506–4515.
F. Chen, M. Chen, C. Yang, J. Liu, N. Luo, G. Yang, D. Chen, and L. Li. Phys. Chem. Chem. Phys., 2015, 17, 1189–1196.
A. Mignot, C. Truillet, F. Lux, L. Sancey, C. Louis, F. Denat, F. Boschetti, L. Bocher, A. Gloter, and O. Stéphan. Chem. Eur. J., 2013, 19, 6122–6136.
A. Betke and G. Kickelbick, Bottom–up. Inorganics, 2014, 2, 1–15.
M. Khorasani–Motlagh, M. Noroozifar, and S. Mirkazehi–Rigi. Spectrochim. Acta A Mol. Biomol. Spectrosc., 2010, 75, 598–603.
N. Shahabadi and S. Hadidi. Spectrochim. Acta A, Mol. Biomol. Spectrosc., 2012, 96, 278–283.
C. Wei, J. Wang, and M. Zhang. Biophys. Chem., 2010, 148, 51–55.
K. Bhadra and G. S. Kumar. Biochim. Biophys. Acta, 2011, 1810, 485–496.
J. Perrin. Brownian Movement and Molecular Reality. London: Taylor & Francis, 1910.
W. Yu and H. Xie. J. Nanomater., 2012, 20, 435873–435880.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2018 A. Bagheri, M. Masoudinia.
The text was submitted by the authors in English. Zhurnal Strukturnoi Khimii, Vol. 59, No. 6, pp. 1530–1535, July-August, 2018.
Rights and permissions
About this article
Cite this article
Bagheri, A., Masoudinia, M. Catalytic Properties of Gadolinium Oxide in the Removal of Doxycycline with Anticancer Activity. J Struct Chem 59, 1478–1483 (2018). https://doi.org/10.1134/S0022476618060343
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476618060343