Abstract
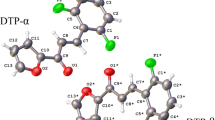
The crystal structure of the new chalcone derivative (E)-3-(5-bromothiophen-2-yl)-1-(2,5-dichlorothiophen- 3-yl)-2-propen-1-one is synthesized and structurally characterized by spectroscopic IR, NMR, and HRMS techniques. Its crystal and molecular structure is determined from the single crystal X-ray diffractionstudy. This compound crystallizes in the monoclinic system and space group P21/n (N 19). The crystal packing is governed by C—H...O non-conventional hydrogen bond-type intermolecular interactions, forming extended zigzag chains related by 21 screw axes running along the [010] direction, with graph-set C(8). These hydrogen bonds contribute to the stabilization of the crystal structure that packs with an efficiency of 71.4%.
Similar content being viewed by others
References
S. Cheenpracha, C. Karalai, C. Ponglimanont, et al. Bioorg. Med. Chem., 2006, 14(6), 1710–1714.
X. Wu, P. Wilairat, and M. L. Go. Bioorg. Med. Chem. Lett., 2002, 12(17), 2299–2302.
S. J. Won, C. T. Liu, L. T. Tsao, et al. Eur. J. Med. Chem., 2005, 40(1), 103–112.
O. Kayser and A. F. Kiderlen. Phytother. Res., 2001, 15(2), 148–152.
M. R. Ahmad, V. G. Sastry, and N. Bano. Int. J. Chem. Tech. Res., 2011, 3(3), 1462–1469.
B. P. Bandgar, S. S. Gawande, R. G. Bodade, et al. Bioorg. Med. Chem., 2010, 18(3), 1364–1370.
A. M. Asiri and S. A. Khan. Molecules, 2011, 16(1), 523–531.
C. Kumar, W. S. Loh, C. W. Ooi, et al. Molecules, 2013, 18(10), 12707–12724.
B. A. Bohm. Introduction to Flavonoids. London: Harwood Academic Publishers, 1998.
S. Eddarir, N. Cotelle, Y. Bakkour, et al. Tetrahedron Lett., 2003, 44(28), 5359–5363.
M. Jayapal and N. Sreedhar. J. Pharm. Sci. Res., 2010, 2(10), 644–647.
J. T. Li, W. Z. Yang, S. X. Wang, et al. Ultrason. Sonochem., 2002, 9(5), 237–239.
S. Zangade, S. Mokle, A. Vibhute, et al. J. Chem. Sci., 2011, 13(1), 1–6.
N. Kaur and D. Kishore. J. Chem. Sci., 2013, 125(3), 555–560.
H. P. Ávila, E. D. F. A. Smania, F. D. Monache, et al. Bioorg. Med. Chem., 2008, 16(22), 9790–9794.
H. M. Al–Maqtari, J. Jamalis, and H. M. Sirat. J. Teknologi., 2015, 77(3), 55–59.
CrystalClear. Rigaku Corporation, Tokyo, Japan, 2008.
G. M. Sheldrick. ActaCrystallogr., 2008, A64(1), 112–122.
G. M. Sheldrick. ActaCrystallogr., 2015, C71(1), 3–8.
K. Branderburg. DIAMOND, Visual Cristal Structure Information System, Crystal Impact GbR. Germany, 1998.
A. L. Spek. J. Appl. Crystallogr., 2003, 36(1), 7–13.
N. J. Lawrence, R. P. Patterson, L. Ooi, et al. Bioorg. Med. Chem. Lett., 2006, 16(15), 5844–5848.
C. R. Groom and F. H. Allen. Angew. Chem. Int. Ed. 2014, 53(3), 662–671.
W. T. A. Harrison, C. S. C. Kumar, H. S. Yathirajan, et al. Acta Crystallogr. E, 2010, 66(10), o2480.
W. T. A. Harrison, C. S. C. Kumar, H. S. Yathirajan, et al. Acta Crystallogr. E, 2010, 66(10), o2479.
G. Dutkiewicz, C. S. C. Kumar, H. S. Yathirajan, et al. Acta Crystallogr. E, 2010, 66(5), o1139.
J. P. Jasinski, A. E. Pek, C. S. C. Kumar, et al. Acta Crystallogr. E, 2010, 66(7), o1717.
T. Steiner. Angew. Chem. Int. Ed., 2002, 41(1), 48–76.
M. C. Etter. Acc. Chem. Res., 1990, 23(4), 120–126.
M. C. Etter, J. C. MacDonald, and J. Bernstein. Acta Crystallogr. B, 1990, 46(4), 256–262.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2018 G. E. Delgado, J. A. Henao, J. H. Quintana, H. M. Al-Maqtari, J. Jamalis, H. M. Sirat.
The text was submitted by the authors in English. Zhurnal Strukturnoi Khimii, Vol. 59, No. 6, pp. 1493–1498, July-August, 2018.
Rights and permissions
About this article
Cite this article
Delgado, G.E., Henao, J.A., Quintana, J.H. et al. Structural Characterization of a New Chalcone Compound Containing a Thiophene Moiety: (E)-3-(5-Bromothiophen-2-YL)-1- (2,5-Dichlorothiophen-3-YL)-2-Propen-1-One. J Struct Chem 59, 1440–1445 (2018). https://doi.org/10.1134/S0022476618060276
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476618060276