Abstract
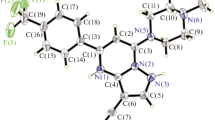
The title compound 2-(pyridin-4-yl)-5-(undecylthio)-1,3,4-oxadiazole (C18H27ON3S) is synthesized, and its structure is confirmed by 1H NMR, MS, elemental analyses and X-ray diffraction. It crystallizes in the monoclinic system, space group P2(1)/c with a = 24.453(17) Å, b = 10.604(7) Å, c = 7.095(5) Å, β = 91.60(2)°, V = 1839(2) Å3, Z = 4, and R = 0.086 for 2295 observed reflections with I > 2σ(I). The preliminary biological test shows that the title compound has good activity against Pythium ultimum with inhibitory to be 77.78%.
Similar content being viewed by others

References
A. Mumtaz, A. Saeed, and I. Malik. J. Chem. Soc. Pak., 2014, 36(5), 852–857.
X. H. Liu, Z. W. Zhai, X. Y. Xu, et al. Bioorg. Med. Chem. Lett., 2015, 25(23), 5524–5528.
L. J. Zhang, M. Y. Yang, Z. H. Sun, et al. Lett. Drug Des. Discovery, 2014, 11(9), 1107–1111.
N. R. Kode, J. J. Vanden Eynde, A. Mayence, et al. Molecules, 2013, 18(9), 11250–11263.
A. R. Saundane, V. A. Verma, and V. T. Katkar. J. Chem., 2013, 2013(2), 148412.
M. J. Ahsan, J. Sharma, M. Singh, et al. Biomed. Res. Int., 2013, 2013(2), 814984.
Y. Q. Zhu, W. M. Liu, B. Liu, et al. Chin. J. Org. Chem., 2009, 29(4), 638–642.
Aziz–ur–Rehman, S. Gul, and M. A. Abbasi. J. Chem. Soc. Pak., 2014, 36(3), 503–511.
Y. Y. Zhou, B. L. Wang, F. J. Di, et al. Bioorg. Med. Chem. Lett., 2014, 24(10), 2295–2299.
B. L. Wang, H. W. Zhu, Y. Ma, et al. J. Agric. Food Chem., 2013, 61(23), 5483–5493.
Z. H. Shen, Y. X. Shi, M. Y. Yang, et al. Chin. J. Struct. Chem., 2016, 35(3), 457–464.
X. H. Liu, W. Zhao, Z. H. Shen, et al. Bioorg. Med. Chem. Lett., 2016, 26(15), 3626–3628.
W. Zhao, Z. H. Shen, T. M. Xu, et al. J. Heterocycl. Chem., 2017, 54(3), 1751–1756.
W. Zhao, Z. H. Shen, J. H. Xing, et al. Chem. Pap., 2017, 71(5), 921–928.
T. I. El–Emary and S. A. El–Mohsen. Molecules, 2012, 17(12), 14464–14483.
X.H. Liu, X.Y. Xu, C.X. Tan et al. Pest Manag. Sci., 2015, 71(2), 292–301.
A. Kojima, S. Takita, T. Sumiya, et al.Bioorg. Med. Chem. Lett., 2013, 23(19), 5311–5316.
W. Zhao, J. H. Xing, T. M. Xu, et al. Front. Chem. Sci. Eng., 2017, 11(3), 363–368.
W. Zhao, Z. H. Shen, T. M. Xu, et al. Lett. Drug Des. Discovery, 2017, 14(3), 323–329.
X. H. Liu, W. Zhao, Z. H. Shen, et al. Eur. J. Med. Chem., 2017, 125(1), 881–889.
S. L. Yan, M. Y. Yang, Z. H. Sun, et al. Lett. Drug Des. Discovery, 2014, 11(7), 940–943.
X. H. Liu, L. Pan, Y. Ma, et al. Chem. Biol. Drug Des., 2011, 78(4), 689–694.
K. P. Harish, K. N. Mohana, L. Mallesha, et al. Lett. Drug Des. Discovery, 2013, 10(8), 783–791.
X. H. Liu, Z. H. Sun, M. Y. Yang, et al. Chem. Biol. Drug Des., 2014, 84(3), 342–347.
Q. Wang, Z. W. Zhai, Z. H. Sun, et al. Chin. J. Struct. Chem., 2016, 35(4), 651–655.
Z. H. Sun, Z. W. Zai, M. Y. Yang, et al. Chin. J. Struct. Chem., 2014, 33(12), 1779–1783.
J. Q. Weng, L. Wang, and X. H. Liu. J. Chem. Soc. Pak., 2012, 34(5), 1248–1252.
X. H. Liu, C. X. Tan, and J. Q. Wenig. Phosphorus Sulfur Silicon Relat. Elem., 2011, 186(3), 558–564.
M. Y. Yang, W. Zhao, X. H. Liu, et al. Chin. J. Struct. Chem., 2015, 34(2), 203–207.
Z. W. Zhai, Y. X. Shi, M. Y. Yang, et al. Chin. J. Struct. Chem., 2016, 35(1), 25–33.
G. X. Sun, Y. X. Shi, Z. W. Zhai, et al. Chin. J. Struct. Chem., 2016, 35(4), 640–644.
X. H. Liu, Q. Wang, Z. H. Sun, et al. Pest Manage. Sci., 2017, 73(5), 953–959.
G. X. Sun, Y. X. Shi, Z. H. Sun, et al. Lett. Drug Des. Discovery, 2014, 11(9), 1119–1123.
G. M. Sheldrick. SHELXL97, Program for the Refinement of Crystal Structures. University of Göttingen, Germany, 1997.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al. Gaussian 03, revision C.01 Gaussian, Inc.: Wallingford, CT, USA, 2014.
X. H. Liu, C. X. Tan, and J. Q. Wenig. Phosphorus Sulfur Silicon Relat. Elem., 2011, 186(3), 552–557.
Z. W. Zhai, Y. X. Shi, M. Y. Yang, et al. Lett. Drug Des. Discovery, 2014, 13(6), 521–525.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2018 Z.-H. Shen, Q. Wang, Z.-H. Sun, H.-K. Wu, C.-X. Tan, J.-Q. Weng, X.-H. Liu.
The text was submitted by the authors in English. Zhurnal Strukturnoi Khimii, Vol. 59, No. 5, pp. 1275–1279, June-July, 2018.
Rights and permissions
About this article
Cite this article
Shen, ZH., Wang, Q., Sun, ZH. et al. Crystal Structure of 2-(Pyridin-4-yl)-5-(Undecylthio)-1,3,4-Oxadiazole. J Struct Chem 59, 1236–1240 (2018). https://doi.org/10.1134/S0022476618050323
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476618050323