Abstract
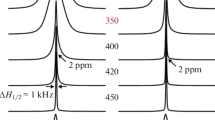
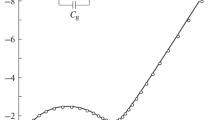
(7Li, 1H) NMR and impedance spectroscopy methods are used to study the ion mobility and conductivity in a complex of the composition Li(NH3CH2COO)(NO3) (I), which has a layered crystal structure. The character of ion motions in lithium and proton sublattices with temperature variation is considered; the types of motions and temperature ranges in which they occur are determined. It is found that above 350 K the dominant process in the lithium sublattice of the compound is Li+ ion diffusion. Possible migration paths of lithium ions in the lattice of the compound are analyzed. The specific conductivity of the compound is found to be 2.4×10–6 S/cm at 393 K.
Similar content being viewed by others
References
A. K. Ivanov-Shits and I. V. Murin, Ionics of Solid State [in Russian], vol. 1, Izd. S.-Pb. Univ., Saint Petersburg (2000).
R. K. B. Gover and P. R. Slater, Annu. Rep. Prog. Chem., Sect. A, 99, 477–504 (2003).
A. A. Kuzubov, N. S. Eliseeva, P. O. Krasnov, F. N. Tomilin, and A. S. Fedorov, J. Sib. Fed. Univ., Chem., 5, No. 2, 209–215 (2012).
R. Bohmer, K. R. Jeffrey, and M. Vogel, Prog. Nucl. Magn. Reson. Spectrosc., 50, 87–174 (2007).
V. Thangadurai and W. Weppner, Ionics, 12, 81–92 (2006).
M. Forsyth, J. Huang, and D. R. MacFarlane, J. Mater. Chem., 10, 2259–2265 (2000).
T. Fujinami, M. A. Mehta, K. Sugie, and K. Mori, Electrochim. Acta, 45, 1181–1186 (2000).
L. V. S. Lopes, D. C. Dragunski, A. Pawlicka, and J. P. Donoso, Electrochim. Acta, 48, 2021–2027 (2003).
N. H. Kaus, N. Lahazan, and A. H. Ahmad, Polym. Adv. Technol., 20, No. 3, 156–160 (2009).
R. Rohan, K. Pareek, W. W. Cai, Y. F. Zhang, G. D. Xu, Z. X. Chen, Z. Q. Gao, Z. Dan, and H. S. Cheng, J. Mater. Chem. A, 3, No. 9, 5132–5139 (2015).
M. Shkir, H. Abbas, S. Kumar, G. Bhagavannarayana, and S. AlFaify, J. Phys. Chem. Solids, 75, No. 8, 959–965 (2014).
J. Baran, M. Drozd, H. Ratajczak, and A. Pietraszko, J. Mol. Struct., 927, Nos. 1-3, 43–53 (2009).
R. G. Kadyrova, I. F. Kabirov, and R. R. Mullakhmetov, Uch. Zap. Kazan. Gos. Acad. Vet. Med. im. N. E. Baumana, No. 215, 141–147 (2013).
Bruker, APEX2, Bruker AXS Inc., Madison, Wisconsin, USA (2005).
G. M. Sheldrick, Acta Crystallogr., A 64, No. 1, 112–122 (2008).
S. P. Gabuda, Yu. V. Gagarinskii, and S. A. Polishchuk, NMR in Inorganic Fluorides [in Russian], Atomizdat, Moscow (1978).
V. Ya. Kavun, T. F. Antokhina, N. N. Savchenko, et al., Russ. J. Inorg. Chem., 60, No. 5, 681–690 (2015).
J. Baran, M. Drozd, H. Ratajczak, A. Pietraszko, M. Trzebiatowska, and H. Ratajczak, Polish. J. Chem., 77, 1561–1577 (2013).
R. M. Silverstein, F. X. Webster, and D. J. Kiemle, Spectrometric Identification of Organic Compounds, 7th ed., John Wiley & Sons, New York (2005).
A. G. Lundin, and E. I. Fedin, NMR Spectroscopy [in Russian], Nauka, Moscow (1986).
V. Ya. Kavun, and V. I. Sergienko, Diffusion Mobility and Ionic Transport in Crystalline and Amorphous Fluorides of the Group 4 Elements and Antimony(III) [in Russian], Dalnauka, Vladivostok (2004).
N. Bloembergen, E. M. Purcell, and R. V. Pound, Phys. Rev., 73, No. 7, 679–712 (1948).
J. M. Bobe, J. M. Réau, J. Senegas, and M. Poulain, J. Non-Cryst. Solids, 209, No. 2, 122–136 (1997).
Y. Kawamoto, J. Fujiwara, and C. Ichimura, J. Non-Cryst. Solids, 111, Nos. 2/3, 245–251 (1989).
E. R. Andrew and R. Bersohn, J. Chem. Phys., 18, 159–161 (1950).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Strukturnoi Khimii, Vol. 57, No. 4, pp. 697-703, May-June, 2016.
Devoted to the 80th anniversary of Professor S. P. Gabuda
Rights and permissions
About this article
Cite this article
Kavun, V.Y., Udovenko, A.A., Makarenko, N.V. et al. Ion mobility and conductivity in the Li(NH3CH2COO)(NO3) compound. J Struct Chem 57, 658–664 (2016). https://doi.org/10.1134/S0022476616040041
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476616040041