Abstract
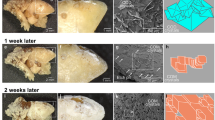
The work considers the aggregation process of calcium oxalate monohydrate crystals (whewellite) during their crystallization from aqueous solutions. The intensive crystal aggregation during the precipitation is evidenced by a decrease in the bulk concentration against an increase in the crystal size. At the initial stages of crystallization fine particles are mainly captured by larger ones in the form of substance deposition on the initiating centers: organic blobs, dead cells, and so on. During the crystallization the effect of the dominant growth of large aggregates increases; the process develops autocatalytically; fine particles disappear more rapidly than large ones, and the crystal size distribution compresses simultaneously with an increase in the average particle size.
Similar content being viewed by others
References
A. A. Korago, Introduction to Biomineralogy [in Russian], Nauka, St. Petersburg (1992).
F. V. Zuzuk, Mineralogy of Uroliths [in Ukrainian], Frank Lviv National University, Lutsk (2005).
O. A. Golovanova, Yu. O. Punin, A. R. Izatulina, et al., Vestn. St.Petersburg Univ., 7, No. 1, 26 (2009).
Yu. O. Punin, O. G. Smetannikova, G. E. Demidova, et al., Dokl. RAN., 332, No. 2, 216 (1993).
N. E. Baranova, Yu. O. Punin, M. D. Evdokimov, et al., Vestn. St.Petersburg Univ., 7, No. 3, 18 (2001).
E.V. Rosseeva, Ja. Buder, P. Simon, et al., Chem. Mater., 20, No. 19, 6003 (2008).
A. Mersmann, Crystallization technology handbook, Marsel Dekker Inc., NY, Basel (1994).
O. A. Golovanova, Pathogenic Minerals in a Human Organism [in Russian], Izd. OmGU, Omsk (2006).
A. Hansen, R. Felix, S. Bisaz, et al., Biochim. Biophys. Acta, 451, No. 2, 549 (1976).
D. Skric, M. Marcovic, L. J. Komunjer, et al., J. Cryst. Growth, 66, 431 (1984).
O. D. Linnikov, Regularities of the Crystallization of Inorganic Salts from Aqueous Solutions [in Russian], Doct. Diss. of Chem. Sc., Ekaterinburg (2011).
I. V. Melikhov, Uspekhi Khimii, 62, No. 3, 249 (1993).
O. A. Golovanova, Yu. O. Punin, A. S. Vysotskii, et al., Khimiya v Interesakh Ustoichivogo Razvitiya, No. 4, 501 (2011).
O. A. Golovanova, V. V. Korol`kov, Yu. O. Punin, et al., Khimiya v Interesakh Ustoichivogo Razvitiya, 21, No. 4, 401 (2013).
O. A. Golovanova, E. Yu. Achkasova, E. V. Rosseeva, et al., Mineralogy of Technogenesis [in Russian], IMin UrO RAN, Miass (2005), p. 123.
Yu. O. Punin, Zap. VMO, 93, No. 3, 459 (1964).
E. B. Treivus and G. M. Kovnurko, Zap. VMO, No. 3, 313 (1985).
H. R. Kruyt (ed.), Colloid Science, V. 1, Elsevier, Amsterdam (1952).
V. G. Levich, Physicochemical Hydrodynamics [in Russian], Fizmatgiz, Moscow (1959).
O. A. Golovanova, Yu. O. Punin, A. R. Izatulina, et al., J. Struct. Chem., 55, No. 7, 1356–1371 (2014).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2014 A. R. Izatulina, Yu. O. Punin, O. A. Golovanova.
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 55, Supplement 1, pp. S41–S47, 2014.
Rights and permissions
About this article
Cite this article
Izatulina, A.R., Punin, Y.O. & Golovanova, O.A. To the formation of aggregate structures of kidney stones. J Struct Chem 55, 1225–1231 (2014). https://doi.org/10.1134/S0022476614070063
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476614070063