Abstract
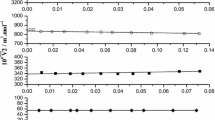
Based on the experimental data, the molar isothermal compressibilities, molar isobaric expansibilities, thermal pressure coefficients, internal pressures of a liquid phase mixture {ethylene glycol (1) + tert-butanol (2)} are calculated for a wide spectrum of compositions in the range of pressures of 0.1–100 MPa and temperatures of 278.15–323.15 K. Shown that the dependences of molar isothermal compressibilities K T, m , molar isobaric expansibilities E P, m , and isochoric thermal pressure coefficients β on the mole fraction of tert-butanol in the mixture are characterized by the absence of extrema typical of aqueous systems. The manifestation of negative partial expansibility and negative partial expansibility of ethylene glycol in the mixture is found. The thermal pressure coefficients decrease with an increase in the mole fraction of tert-butanol at all pressures and temperatures. A rise in the pressure increases the thermal pressure coefficient, while a rise in the temperature decreases its value due to a decrease of free space in the mixture. An increase in the concentration of tert-butanol leads to an increase in the negative temperature coefficient of internal pressure ΔP int/ΔT, which indicates a weakening of intermolecular interaction at these compositions.
Similar content being viewed by others
References
G. I. Egorov, D. M. Makarov, and A. M. Kolker, J. Struct. Chem., 54,Suppl. 2, S305–S320 (2013).
J.-Y. Huot, E. Battistel, R. Lumry, G. Villeneuve, J.-F. Lavallee, A. Anusiem, and C. Jolicoeur, J. Solut. Chem., 17, 601 (1988).
E. Zorebski and A. Waligóra, J. Chem. Eng. Data, 53, 591 (2008).
J. A. Riddick, W. B. Bunger, and T. B. Sakano, Techniques of Chemistry. Organic Solvents, 4th ed., Wiley-Interscience, New York (1986), Vol. II.
E. A. Moelwin-Hughes, Physical Chemistry, Pergamon Press, London (1957).
J. H. Hildebrand and R. L. Scott Regular Solutions, Englewood Cliffs, Prentice-Hall, New Jersey (1962).
I. Prigozhin and R. Defei, Chemical Thermodynamics [in Russian], Nauka, Novosibirsk (1966).
V. N. Kartsev, M. N. Rodnikova, and S. N. Shtykov, J. Struct. Chem., 45, No. 1, 91–95 (2004).
R. O. Watts and I. J. McGee, Liquid State Chemical Physics, Wiley, New York (1976).
E. V. Ivanov and V. K. Abrosimov, J. Struct. Chem., 46, No. 5, 856–861 (2005).
A. M. Kolker, V. P. Korolev, and D. V. Batov, J. Struct. Chem., 46, No. 5, 927–930 (2005).
V. N. Kartsev and I. V. Bogomolova, J. Struct. Chem., 47,Suppl., S159–S156 (2006).
V. N. Kartsev, K. E. Pankin, and D. V. Batov, J. Struct. Chem., 47, No. 2, 277 (2006).
M. J. Blandamer and H. Hoiland, Phys. Chem. Chem. Phys., 1, 1873 (1999).
P. Buckley and P. A. Giguère, Can. J. Chem., 45, 397–407 (1967).
Yu. Ia. Kharitonov, E. G. Khoshabova, M. N. Rodnikova, K. T. Dudnikova, and A. B. Razumova, Dokl. Akad. Nauk SSSR, 304, No. 4, 917 (1989).
R. Boese and H.-C. Weiss, Acta Crystallogr. Sect. C: Cryst. Struct. Commun., C54, IUC9800024 (1998).
L. Saiz, J. A. Padro, and E. Guardia, J. Chem. Phys., 114, 3187 (2001).
I. Bakó, T. Grósz, G. Pálinkás, and M. C. Bellisent-Funel, J. Chem. Phys., 118, 3215 (2003).
A. V. Gubskaya and P. G. Kusalik, J. Phys. Chem. A, 108, 7151 (2004).
A. V. Gubskaya and P. G. Kusalik, J. Phys. Chem. A, 108, 7165 (2004).
A. G. Novikov, M. N. Rodnikova, and O. V. Sobolev, Physica B, 350, E363 (2004).
N. A. Chumaevskii, M. N. Rodnikova, and J. Barthel, J. Mol. Liq., 115/2-3, 63 (2004).
M. Matsugami, T. Takamuku, T. Otomo, and T. Yamaguchi, J. Phys. Chem. B, 110, 12372 (2006).
M. N. Rodnikova, N. A. Chumaevskii, V. M. Troitskii, and D. B. Kayumova, J. Phys. Chem., 80, No. 5, 947 (2006).
G. I. Egorov, D. M. Makarov, and A. M. Kolker, J. Chem. Thermodyn., 61, 169 (2012).
A. H. Narten and S. I. Sandler, J. Chem. Phys., 71, 2069 (1979).
P. G. Kusalik, A. P. Lyubartsev, D. L. Bergman, and A. Laaksonen, J. Phys. Chem. B, 104, 9526 (2000).
D. Wojtkow and M. A. Czarnecki, J. Phys. Chem. A, 109, 8218 (2005).
T. Fukasawa, Y. Tominaga, and A. Wakisaka, J. Phys. Chem. A, 108, 59 (2004).
D. T. Bowron, J. L. Finney, and A. K. Soper, Mol. Phys., 93, 531 (1998).
K. Yoshida, T. Yamaguchi, A. Kovalenko, and F. Hirata, J. Phys. Chem. B, 106, 5042 (2002).
M. Sakurai, K. Nakamura, and K. Nitta, Bull. Chem. Soc. Jpn., 67, 1580 (1994).
V. I. Grasin and V. K. Abrosimov, Izv. Akad. Nauk SSSR, Ser. Khim., No. 2, 317 (1991).
M. Sakurai and T. Nakagawa, J. Chem. Thermodyn., 14, 269 (1982).
M. Sakurai and T. Nakagawa, J. Chem. Thermodyn., 16, 171 (1984).
E. V. Ivanov and V. K. Abrosimov, Zh. Neorg. Khim., 40, No. 6, 1047 (1995).
E. V. Ivanov, J. Chem. Thermodyn., 47, 162 (2012).
M. Sakurai, Bull. Chem. Soc. Jpn., 60, 1 (1987).
C. de Visser, G. Perron, and J. E. Desnoyers, Can. J. Chem., 55, 856 (1977).
P. Hynčica, L. Hnedkovsky, and I. Cibulka, J. Chem. Thermodyn., 38, 418 (2006).
M. I. Origlia and E. M. Wooley, J. Chem. Thermodyn., 33, 451 (2001).
C. Jolicouer and G. Lacroix, Can. J. Chem., 54, 624 (1976).
G. I. Egorov and D. M. Makarov, J. Chem. Thermodyn., 43, 430 (2011).
M. Sakurai, J. Solut. Chem., 18, 37 (1989).
A. F. S. S. Mendonca, M. J. A. Barbas, J. M. Freitas, and I. M. S. Lampreia, J. Chem. Thermodyn., 36, 965 (2004).
M. Sakurai, T. Komatsu, and T. Nakagawa, J. Solut. Chem., 4, 511 (1975).
D. Hamilton and R. H. Stokes, J. Solut. Chem., 1, 213 (1972).
G. I. Egorov and D. M. Makarov, J Solut. Chem., 41, 536 (2012).
V. N. Kartsev, M. N. Rodnikova, J. Bartel, and S. N. Shtykov, Zh. Fiz. Khim., 76, No. 6, 1016 (2002).
V. N. Kartsev, M. N. Rodnikova, S. N. Shtykov, and J. Bartel, Zh. Fiz. Khim., 77, No. 8, 1456 (2003).
V. N. Kartsev, M. N. Rodnikova, and S. N. Shtykov, J. Struct. Chem., 45, No. 1, 91–95 (2004).
V. N. Kartsev, J. Struct. Chem., 45, No. 5, 832–837 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2013 G. I. Egorov, D. M. Makarov.
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 54, Supplement 2, pp. S325–S340, 2013.
Rights and permissions
About this article
Cite this article
Egorov, G.I., Makarov, D.M. Bulk properties of a liquid phase mixture {ethylene glycol+tert-butanol} in the temperature range 278.15–348.15 K and pressures of 0.1–100 MPa. II. Molar isothermal compressibility, molar isobaric expansibility, thermal pressure coefficient, and internal pressure. J Struct Chem 54 (Suppl 2), 320–335 (2013). https://doi.org/10.1134/S002247661308012X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002247661308012X