Abstract
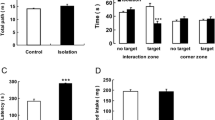
The effect of social isolation or enriched environment on fear conditioning and extinction was studied in 3.5–4-month-old rats exposed to early proinflammatory stress via LPS administration on the 3rd and 5th day after birth. Social isolation reduced the expression of conditioned fear in males and females, as measured by a decrease in time of the freezing response 24 h post conditioning. The greatest sensitivity to social isolation was observed in rats with early proinflammatory stress, for which the freezing time was reduced after exposure to both the cue and the context. We hypothesize that the decrease in fear is associated with a memory deficit. The enriched environment also reduced the freezing time, but to a lesser extent than social isolation, and only the contextual fear response was reduced. The extinction of fear in males of the LPS group was accelerated after exposure to social isolation and enriched environment compared to animals of this group kept in standard conditions. Exposure to early proinflammatory stress resulted in an increased vulnerability of adult animals to changes in housing conditions.




Similar content being viewed by others
REFERENCES
Maynard TM, Sikich L, Lieberman JA, Lamantia S (2001) Neural development, cell—cell signaling, and the ‘‘Two-Hit’’ hypothesis of schizophrenia. Schizophr Bull 27:457-476. https://doi.org/10.1093/oxfordjournals.schbul.a006887
Walker AK, Nakamura T, Byrne RJ, Sundresan N, Tynan RJ, Hunter M, Hodgson DM (2009) Neonatal lipopolysaccharide and adult stress exposure predisposes rats to anxiety-like behaviour and blunted corticosterone responses: implications for the double-hit hypothesis. Psychoneuroendocrinology 34:1515-1525. https://doi.org/10.1016/j.psyneuen.2009.05.010
Lahiri DK, Maloney B, Zawia NH (2009) The LEARn model: an epigenetic explanation for idiopathic neurobiological diseases. Mol Psychiatry 14: 992-1003. https://doi.org/10.1038/mp.2009.82
Custódio CS, Mello BSF, Filho AJMC, de Carvalho Lima CN, Cordeiro RC, Miyajima F, Réus GZ, Vasconcelos SMM, Barichello T, Quevedo J, de Oliveira AC, de Lucena DF, Macedo DS (2018) Neonatal immune challenge with lipopolysaccharide triggers long-lasting sex- and age related behavioral and immune/neurotrophic alterations in mice: relevance to autism spectrum disorders. Mol Neurobiol 55:3775-3788. https://doi.org/10.1007/s12035-017-0616-1
Grigoryan GA, Dygalo NN, Geht AB, Stepanichev MYu, Gulyaeva NV (2014) Molecularcellular mechanisms of depression. The role of glucocorticoids, cytokines, and neurotrophic factors in the genesis of depressive disorders. Usp Fiziol Nauk 44:3-20. (In Russ).
Parker G1, Brotchie H (2010) Gender differences in depression. Int Rev Psychiatry 22:429-36. https://doi.org/10.3109/09540261.2010.492391
Alexander C, Rietschel ET (2001) Bacterial lipopolysaccharides and innate immunity. J Endotoxin Res 7:167-202. PMID: 11581570
Bilbo SD, Rudy JW, Watkins LR, Maier SF (2006) A behavioural characterization of neonatal infection-facilitated memory impairment in adult rats. Behav Brain Res 169:39-47. https://doi.org/10.1016/j.bbr.2005.12.002
Tchessalova D, Tronson NC (2019) Memory deficits in males and females long after subchronic immune challenge. Neurobiol Learn Mem 158:60-72. https://doi.org/10.1016/j.nlm.2019.01.003
Osborne BF, Caulfield JI, Solomotis SA, Schwarz JM (2017) Neonatal infection produces significant changes in immune function with no associated learning deficits in juvenile rats. Dev Neurobiol 77:1221-1236. https://doi.org/10.1002/dneu.22512
Tishkina A, Stepanichev M, Kudryashova I, Freiman S, Onufriev M, Lazareva N, Gulyaeva N (2016) Neonatal proinflammatory challenge in male Wistar rats: Effects on behavior, synaptic plasticity, and adrenocortical stress response. Behav Brain Res 304:1-10. https://doi.org/10.1016/j.bbr.2016.02.001
Broshevitskaya ND, Pavlova IV, Zaichenko MI, Onufriev MV, Moiseeva YuV, Grigoryan GA (2020) The Sex Differences in Defensive Behavior of Adult Rats in Response to Early Neuroinflammatory Stress. Zh Vyssh Nerv Deyat 70:261-278. (In Russ). https://doi.org/10.31857/S0044467720020057
Doenni VM, Song CM, Hill MN, Pittman QJ (2017) Early-life inflammation with LPS delays fear extinction in adult rodents. Brain Behav Immun 63:176-185. https://doi.org/10.1016/j.bbi.2016.11.022
Harre EM, Galic MA, Mouihate A, Noorbakhsh F, Pittman QJ (2008) Neonatal inflammation produces selective behavioral deficits and alters N-metyl-D-aspartate receptor subunit mRNA in the adult rat brain. Eur J Neurosci 27:644-653. https://doi.org/10.1111/j.1460-9568.2008.06031.x
Kohman RA, Tarr AJ, Sparkman NL, Bogale TM, Boehm GW (2008) Neonatal endotoxin exposure impairs avoidance learning and attenuates endotoxin-induced sickness behavior and central IL-1beta gene transcription in adulthood. Behav Brain Res 194:25-31. https://doi.org/10.1016/j.bbr.2008.06.018
Lukkes JL, Watt MJ, Lowry CA, Forster GL (2009) Consequences of post-weaning social isolation on anxiety behavior and related neural circuits in rodents. Front Behav Neurosci 3:18. https://doi.org/10.3389/neuro.08.018.2009
Guarnieri LO, Pereira-Caxeta AR, Medeiros DC, Aquino NSS, Szawka RE, Mendes EMA, Moraes MFD, Pereira GS (2020) Pro-neurogenic effect of fluoxetine in the olfactory bulb is concomitant to improvements in social memory and depressive-like behavior of socially isolated mice. Transl Psychiatry 10:33. https://doi.org/10.1038/s41398-020-0701-5
Wang HT, Huang FL, Hu ZL, Zhang WJ, Qiao XQ, Huang YQ, Dai RP, Li F, Li CQ (2017) Early-life social isolation-induced depressive-like behavior in rats results in microglial activation and neuronal histone methylation that are mitigated by minocycline. Neurotox Res 31:505-520. https://doi.org/10.1007/s12640-016-9696-3
Pavlova IV, Broshevitskaya ND, Zaichenko MI, Grigoryan GA (2021) The Influence of Social Isolation and Enriched Environment on Anxious and Depressive-like Behavior of Rats in Norm and After the Early Proinflammatory Stress. Zh Vyssh Nerv Deyat 71(5):655-667. (In Russ).
Kulesskaya N, Rauvala H, Voikar V (2011) Evaluation of social and physical enrichment in modulation of behavioural phenotype in C57BL/6J female mice. PLoS One 6:e24755. https://doi.org/10.1371/journal.pone.0024755
Krupina NA, Shirenova SD, Khlebnikova NN (2020) Prolonged social isolation, started early in life, impairs cognitive abilities in rats depending on sex. Brain Sci 10: 799. https://doi.org/10.3390/brainsci10110799
Hori M, Yamada K, Ohnishi J, Sakamoto S, Furuie H, Murakami K, Ichitani Y (2014) Tickling during adolescence alters fear-related and cognitive behaviors in rats after prolonged isolation. Physiol Behav 131:62-67. https://doi.org/10.1016/j.physbeh.2014.04.008
Võikar V, Polus A, Vasar E, Rauvala H (2005) Long-term individual housing in C57BL/6J and DBA/2 mice: assessment of behavioral consequences. Genes Brain Behav 4:240-252. https://doi.org/10.1111/j.1601-183X.2004.00106.x
Mora-Gallegos A, Fornaguera J (2019) The effects of environmental enrichment and social isolation and their reversion on anxiety and fear conditioning. Behav Processes 158:59-69. https://doi.org/10.1016/j.beproc.2018.10.022
Badowska DM, Brzozka MM, Chowdhury A, Malzahn D, Rossner MJ (2015) Data calibration and reduction allows to visualize behavioural profiles of psychosocial influences in mice towards clinical domains. Eur Arch Psychiatry Clin Neurosci 165:483-496. https://doi.org/10.1007/s00406-014-0532-6
Ouchi H, Ono K, Murakami Y, Matsumoto K (2013) Social isolation induces deficit of latent learning performance in mice: a putative animal model of attention deficit/hyperactivity disorder. Behav Brain Res 238:146-153. https://doi.org/10.1016/j.bbr.2012.10.029.
Okada R, Marsumoto K, Tsushima R, Fujiwara H, Tsuneyama K (2014) Social isolation stress-induced fear memory deficit is mediated by down-regulated neuro-signaling system and Egr-1 expression in the brain. Neurochem Res 39:875-882. https://doi.org/10.1007/s11064-014-1283-5
Matsumoto K, Fujiwara H, Araki R, Yabe T (2019) Post-weaning social isolation of mice: a putative animal model of developmental disorders. J Pharmacol Sci 141:111-118. https://doi.org/10.1016/j.jphs.2019.10.002
Mileva GR, Bielajew C (2015) Environmental manipulation affects depressive-like behaviours in female Wistar-Kyoto rats. Behav Brain Res 293:208-216. https://doi.org/10.1016/j.bbr.2015.07.035
Grippo AJ, Ihm E, Wardwell J, McNeal N, Scotti MA, Moenk DA, Chandler DL, LaRocca MA, Preihs K (2014) The effects of environmental enrichment on depressive and anxiety-relevant behaviors in socially isolated prairie voles. Psychosom Med 76:277-284. https://doi.org/10.1097/PSY.0000000000000052
Brenes JC, Fornaguera J, Sequeira-Cordero A (2020) Environmental enrichment and physical exercise attenuate the depressive-like effects induced by social isolation stress in rats. Front Pharmacol 11:804. https://doi.org/10.3389/fphar.2020.00804
Pietropaolo S, Feldon J, Alleca E, Cirulli F, Yee BK (2006) The role of voluntary exercise in enriched rearing: a behavioral analysis. Behav Neurosci 4:787–803. https://doi.org/10.1037/0735-7044.120.4.787
Pietropaolo S, Feldon J, Yee BK (2014) Environmental enrichment eliminates the anxiety phenotypes in a triple transgenic mouse model of Alzheimer’s disease. Cogn Affect Behav Neurosci 14:996-1008. https://doi.org/10.3758/s13415-014-0253-3
Weiss IC, Pryce CR, Jongen-Relo AL, Nanz-Bahr NI, Feldon J (2004) Effect of social isolation on stress-related behavioural and neuroendocrine state in the rat. Behav Brain Res 152:279–295. https://doi.org/10.1016/j.bbr.2003.10.015
Hsiao YH, Kuo JR, Chen SH, Gean PW (2012) Amelioration of social isolation-triggered onset of early Alzheimer’s desease-related cognitive deficit by N-acetylcysteine in a transgenic mouse model. Neurobiol Dis 45:1111-1120. https://doi.org/10.1016/j.nbd.2011.12.031
Zelikowsky M, Hui M, Karigo T, Gradinaru V, Deverman BE, Anderson D.J. (2018) The neuropeptide Tac2 controls a distributed brain state induced by chronic social isolation stress. Cell 173:1265-1279. https://doi.org/10.1016/j.cell.2018.03.037
Hellemans KG, Benge LC, Olmstead MC (2004) Adolescent enrichment partially reverses the social isolation syndrome. Dev Brain Res 150:103-15. https://doi.org/10.1016/j.devbrainres.2004.03.003
Heidbreder CA, Weiss IC, Domeney AM, Pryce C, Homberg J, Hedou G, Feldon J, Moran MC, Nelson P (2000) Behavioral, neurochemical and endocrinological characterization of the early social isolation syndrome. Neuroscience 100:749-68. https://doi.org/10.1016/s0306-4522(00)00336-5
Green MR, McCormick CM (2013) Effects of social instability stress in adolescence on Long-Term, Not short-term, spatial memory performance. Behav Brain Res 256:165-171. https://doi.org/10.1016/j.bbr.2013.08.011
Wang B, Wu Q, Lei L, Sun H, Michael N, Zhang X, Wang Y, Zhang Y, Ge B, Wu X, Wang Y, Xin Y, Zhao J, Li S (2019) Long-term social isolation inhibits autophagy activation, induces postsynaptic dysfunctions and impairs spatial memory. Exp Neurol 311:213-224. https://doi.org/10.1016/j.expneurol.2018.09.009
Leger M, Paizanis E, Dzahini K, Quiedeville A, Bouet V, Cassel JC, Freret T, Schumann-Bard P, Boulouard M (2015) Environmental enrichment duration differentially affects behavior and neuroplasticity in adult mice. Cereb Cortex 25:4048-4061. https://doi.org/10.1093/cercor/bhu119
Walasek G, Wesierska M, Werka T (2002) Effects of social rearing conditions on conditioned suppression in rats. Acta Neurobiol Exp (Wars) 62:25-31. PMID: 12004570
Pavlova IV, Rysakova MP (2015) The manifestation of the anxiety during fear conditioning in Wistar rats. Zh Vyssh Nerv Deyat 65:720-734. (In Russ). https://doi.org/10.7868/S0044467715050123
Skelly MJ, Chapell AE, Carter E, Weiner JL (2015) Adolescent social isolation increases anxiety-like behavior and ethanol intake and impairs fear extinction in adulthood: possible role of disrupted noradrenergic signaling. Neuropharmacology 97:149-159. https://doi.org/10.1016/j.neuropharm.2015.05.025
Noble LJ, Gonzalez IJ, Meruva VB, Callahan KA, Belfort BD, Ramanathan KR, Meyers E, Kilgard MP, Rennaker RL, McIntyre CK (2017) Effects of vagus nerve stimulation on extinction of conditioned fear and post-traumatic stress disorder symptoms in rats. Transl Psychiatry 7:e1217. https://doi.org/10.1038/tp.2017.191
Alshammari TK, Alghamdi H, Alkhader LF, Alqahtani Q, Alrasheed NM, Yacoub H, Alnaem N, AlNakiyah M, Alshammari MA (2020) Analysis of the molecular and behavioral effects of acute social isolation on rats. Behav Brain Res 377:112191. https://doi.org/10.1016/j.bbr.2019.112191
Gibb J, Hayley S, Gandhi R, Poulter MO, Anisman H (2008) Synergistic and additive actions of a psychosocial stressor and endotoxin challenge: Circulating and brain cytokines, plasma corticosterone and behavioral changes in mice. Brain Behav Immun 22:573-589. https://doi.org/10.1016/j.bbi.2007.12.001
Gandhi R, Hayley S, Gibb J, Merali Z, Anisman H (2007) Influence of poly I:C on sickness behaviors, plasma cytokines, corticosterone and central monoamine activity: moderation by social stressors. Brain Behav Immun 21:477-489. https://doi.org/10.1016/j.bbi.2006.12.005
Funding
This work was carried out within the State assignment of the Ministry of Education and Science of the Russian Federation for 2021–2023 on the topic “Fundamental neurobiological mechanisms of behavior, memory and learning in normal and pathological conditions” (reg. No. AAAAA-A17-117-92040002-6), and with the support of the Russian Foundation for Basic Research (projects No. 19-015-00129A and 19-34-90022).
Author information
Authors and Affiliations
Contributions
I.V. Pavlova—planning and conducting experiments, data processing, writing and editing the article; N.D. Broshevitskaya—methodology, conducting experiments.
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest related to the publication of this article.
Additional information
Russian Text © The Author(s), 2021, published in Zhurnal Evolyutsionnoi Biokhimii i Fiziologii, 2021, Vol. 57, No. 4, pp. 331–343https://doi.org/10.31857/S0044452921040057.
Rights and permissions
About this article
Cite this article
Pavlova, I.V., Broshevitskaya, N.D. The Influence of Social Isolation and Enriched Environment on Fear Conditioning in Rats after Early Proinflammatory Stress. J Evol Biochem Phys 57, 803–816 (2021). https://doi.org/10.1134/S0022093021040062
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093021040062