Abstract
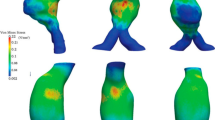
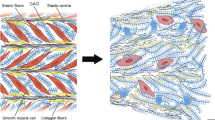
The aorta is the main vessel in the human circulatory system which plays a critical role in oxygen and nutrient supply to all abdominal organs and to the lower limbs. A critical pathology of this vessel is an aneurysm. An important problem in medical practice is to predict aortic aneurysm rupture and plan surgery since aneurysm rupture outside a hospital, as a rule, leads to a lethal outcome. To develop adequate mathematical models that predict such an outcome, it is necessary to determine the strength characteristics of tissues of the aorta, aortic aneurysm, and iliac arteries at various stages of aneurysm growth based on individual patient data. The strength characteristics of the tissues of the human aorta, aortic aneurysm, and common iliac arteries have been studied. It has been experimentally proved that in healthy aorta tissue specimens, the differences between the ultimate relative strains in the axial and circumferential directions are statistically significant (\(p=0.033\)), which is not observed in the case of the aortic aneurysm. The results can also be treated as remodeling of the aortic aneurysm wall compared to the healthy aorta. These data can be used to develop predictive models for aortic aneurysm rupture in personalized hydroelastic modeling.







Similar content being viewed by others
REFERENCES
T. McGloughlin, Biomechanics and Mechanobiology of Aneurysms (Springer, Berlin, Heidelberg, 2011).
F. L. Moll, J. T. Powell, G. Fraedrich, et al., “Management of Abdominal Aortic Aneurysms Clinical Practice Guidelines of the European Society for Vascular Surgery," Europ. J. Vascular Endovascular Surgery 41, S1–S58 (2011).
N. Sakalihasan, H. Kuivaniemi, B. Nusgens, et al., “Aneurysm: Epidemiology Aetiology and Pathophysiology," Biomechanics and Mechanobiology Aneurysms (Springer, Berlin; Heidelberg, 2010), pp. 1–33.
H. E. Salman, B. Ramazanli, M. M. Yavus, and H. C. Yalcin, “Biomechanical Investigation of Disturbed Hemodynamics-Induced Tissue Degeneration in Abdominal Aortic Aneurysms Using Computational and Experimental Techniques," Frontiers Bioengng Biotechnol 7, 111 (2019).
R. Darling, C. Messina, D. Brewster, and L. Ottinger, “Autopsy Study of Unoperated Abdominal Aortic Aneurysms. The Case for Early Resection," Circulation 56 (3): Suppl. II161–4 (1977).
J. P. V. Geest, M. S. Sacks, and D. A. Vorp, “The Effects of Aneurysm on the Biaxial Mechanical Behavior of Human Abdominal Aorta," J. Biomech. 39 (7), 1324–1334 (2006).
E. S. Di Martino, A. Bohra, J. P. V. Geest, et al., “Biomechanical Properties of Ruptured Versus Electively Repaired Abdominal Aortic Aneurysm Wall Tissue," J. Vascular Surgery 43 (3), 570–576 (2006).
S. Laurent, P. Boutouyrie, and P. Lacolley, “Structural and Genetic Bases of Arterial Stiffness," Hypertension 45 (6), 1050–1055 (2005).
J. J. Oliver and D. J. Webb, “Noninvasive Assessment of Arterial Stiffness and Risk of Atherosclerotic Events," Arteriosclerosis, Thrombosis, Vascular Biology 23 (4), 554–566 (2003).
J. G. Logan, M. B. Engler, and H. Kim, “Genetic Determinants of Arterial Stiffness," J. Cardiovascular Translate. Res. 8 (1), 23–43 (2015).
W. P. Robinson, A. Schanzer, Y. Li, et al., “Derivation and Validation of a Practical Risk Score for Prediction of Mortality after Open Repair of Ruptured Abdominal Aortic Aneurysms in a US Regional Cohort and Comparison to Existing Scoring Systems," J. Vascular Surgery 57 (2), 354–361 (2013).
A. Samy, G. Murray, and G. MacBain, “Glasgow Aneurysm Score," Cardiovascular Surgery 2 (1), 41–44 (1994).
A. Tambyraja, J. Murie, and R. Chalmers, “Predictors of Outcome after Abdominal Aortic Aneurysm Rupture: Edinburgh Ruptured Aneurysm Score," World J. Surgery 31 (11), 2243–2247 (2007).
D. Backes, M. D. Vergouwen, A. T. Tiel Groenestege, et al., “Phases Score for Prediction of Intracranial Aneurysm Growth," Stroke 46 (5), 1221–1226 (2015).
B. D. Stemper, N. Yoganandan, M. R. Stineman, et al., “Mechanics of Fresh, Refrigerated, and Frozen Arterial Tissue," J. Surgical Res. 139 (2), 236–242 (2007).
T. C. Gasser, S. Gallinetti, X. Xing, et al., “Spatial Orientation of Collagen Fibers in the Abdominal Aortic Aneurysm’s Wall and Its Relation to Wall Mechanics," Acta Biomaterialia 8 (8), 3091–3103 (2012).
A. E. Medvedev, V. I. Samsonov, and V. M. Fomin, “Rational Structure of Blood Vessels," Prikl. Mekh. Tekh. Fiz. 47 (3), 24–30 (2006) [J. Appl. Mech. Tech. Phys. 47 (3), 324–329 (2006); DOI: 10.1007/s10808-006-0059-3].
D. V. Parshin, A. I. Lipovka, A. V. Dubovoy, et al., “Different Stages of the Evolution of Cerebral Aneurysms: Joint Analysis of Mechanical Test Data and Histological Analysis of Aneurysm Tissue," EPJ. Web Conf. EDP Sci. 221, 01028 (2019).
L. Cardamone, A. Valentin, J. Eberth, and J. Humphrey, “Origin of Axial Prestretch and Residual Stress in Arteries," Biomech. Model. Mechanobiol. 8 (6), 431–446 (2009).
I. Tagiltsev and A. Shutov, “Geometrically Nonlinear Modeling of Pre-Stressed Viscoelastic Fiber-Reinforced Composites with Application to Arteries," Biomech. Model. Mechanobiol. 20 (1), 323–337 (2021).
W. Jin and J. Alastruey, “Arterial Pulse Wave Propagation across Stenoses and Aneurysms: Assessment of One-Dimensional Simulations against Three-Dimensional Simulations and In Vitro Measurements," J. Roy. Soc. Interface. 18 (177), 20200881 (2021).
M. Shmurak, A. Kuchumov, and N. Voronova, “Analysis of Hyperelastic Models for Describing Soft Human Body Behavior," Master’s J., No. 1, 230–243 (2017).
A. Liogky, P. Karavaikin, and V. Salamatova, “Impact of Material Stiffness and Anisotropy on Coaptation Characteristics for Aortic Valve Cusps Reconstructed from Pericardium," Mathematics. 9 (18), 2193 (2021).
D. Parshin, A. Lipovka, A. Yunoshev, et al., “On the Optimal Choice of a Hyperelastic Model of Ruptured and Unruptured Cerebral Aneurysm," Scientific Rep. 9 (1), 1–11 (2019).
R. Teixeira, M. J. Vieira, A. Gonçalves, et al., “Ultrasonographic Vascular Mechanics to Assess Arterial Stiffness: a Review," Europ. Heart J. Cardiovascular Imaging. 17 (3), 233–246 (2016).
B. J. Doyle, A. Callanan, P. A. Grace, and E. G. Kavanagh, “On the Influence of Patient-Specific Material Properties in Computational Simulations: A Case Study of a Large Ruptured Abdominal Aortic Aneurysm," Intern. J. Numer. Methods Biomed. Engng. 29 (2), 150–164 (2013).
D. H. Wang, M. Makaroun, M. W. Webster, and D. A. Vorp, “Mechanical Properties and Microstructure of Intraluminal Thrombus from Abdominal Aortic Aneurysm," J. Biomech. engng. 123 (6), 536–539 (2001).
M. Bukač and S. C. Shadden, “Quantifying the Effects of Intraluminal Thrombi and Their Poroelastic Properties on Abdominal Aortic Aneurysms," Arch. Appl. Mech. 92, 435–466 (2021).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Translated from Prikladnaya Mekhanika i Tekhnicheskaya Fizika, 2021, Vol. 63, No. 2, pp. 84-93. https://doi.org/10.15372/PMTF20220208.
Rights and permissions
About this article
Cite this article
Lipovka, A.I., Karpenko, A.A., Chupakhin, A.P. et al. STRENGTH PROPERTIES OF ABDOMINAL AORTIC VESSELS: EXPERIMENTAL RESULTS AND PERSPECTIVES. J Appl Mech Tech Phy 63, 251–258 (2022). https://doi.org/10.1134/S0021894422020080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0021894422020080