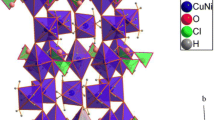
This study explores the oxidation dynamics of “pure” CuCl during prolonged environmental exposure through X-ray diffraction and temperature-dependent magnetization assessments. While CuCl is traditionally considered diamagnetic, our investigation reveals the emergence of an antiferromagnetic transition at 4.7 K. This anomaly is potentially induced by the integration of water molecules into the CuCl matrix. The hydration process initiates a series of oxidation reactions, ultimately transmuting CuCl into Cu2Cl(OH)3. Over time, the distinctive diffractogram peaks corresponding to CuCl diminish, concurrently with the appearance and intensification of those ascribed to Cu2Cl(OH)3, culminating in a complete phase transition as confirmed by X-ray analysis. Correspondingly, magnetization measurements clearly discern magnetic transitions at 6.4 and 16 K, intensifying with the sample’s exposure duration. These findings illuminate the stark potential for probing the magnetic intricacies of Cu2Cl(OH)3, a subject that remains a compelling and unresolved intrigue within the field.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
T. H. Geballe, R. H. Hammond, and P. M. Wu, Physica C: Superconductivity and its Applications 514, 9 (2015).
N. B. Brandt, S. V. Kuvshinnikov, A. P. Rusakov, and V. M. Semenov, Pis’ma ZhETF 27, 37 (1978) [JETP Lett. 27, 33 (1978)].
T. Saerbeck, J. Pereiro, J. Wampler, J. Stanley, J. Wingert, O. G. Shpyrko, and I. K. Schuller, J. Magn. Magn. Mater. 346, 161 (2013).
M. G. Banks, R. K. Kremer, C. Hoch, A. Simon, B. Ouladdiaf, J. M. Broto, and M. H. Whangbo, Phys. Rev. B 80(2), 024404 (2009).
M. Schmitt, O. Janson, M. Schmidt, S. Hoffmann, W. Schnelle, S. L. Drechsler, and H. Rosner, Phys. Rev. B 79(24), 245119 (2009).
H. B. Yi, F. F. Xia, Q. Zhou, and D. Zeng, J. Phys. Chem. A 115(17), 4416 (2011).
K. Miyajima, M. Ashida, and T. Itoh, J. Phys. Condens. Matter 19(44), 445006 (2007).
F. O. Lucas, P. J. McNally, A. Cowley, S. Daniels, L. Bradley, D. Danieluk, and D. M. Taylor, Phys. Status Solidi C 6(S11), S114 (2009).
Q. Li, S. Li, K. Wang, Z. Quan, Y. Meng, and B. Zou, J. Phys. Chem. Lett. 8(2), 500 (2017).
Q. Li, S.W. Zhang, Y. Zhang, and C. Chen, Nanotechnology 17(19), 4981 (2006).
C. Song, Y. You, X. Chen, X. Zhou, Y. Wang, and F. Pan, Nanotechnology 29(11), 112001 (2018).
Zh. Liu, Z. Feng, H. Yan, X. Wang, X. Zhou, P. Qin, H. Guo, R. Yu, and Ch. Jiang, Advanced Electronic Materials 5(7), 1900176 (2019).
R. Rana, P. Pandey, R. P. Singh, and D. S. Rana, Sci. Rep. 4(1), 4138 (2014).
M. Gruyters and D. Schmitz, Phys. Rev. Lett. 100(7), 077205 (2008).
H. Ohldag, A. Scholl, F. Nolting, E. Arenholz, S. Maat, A. T. Young, M. Carey, and J. Stöhr, Phys. Rev. Lett. 91(1), 017203 (2003).
A. V. Mahajan, D. C. Johnston, D. R. Torgeson, and F. Borsa, Phys. Rev. B 46, 10966 (1992).
S. Hashimoto, Y. Matsuda, T. Sato, and S. Anzai, J. Appl. Phys. 98(12), 123903 (2005).
N. Sharma, B. K. Srivastava, A. Krishnamurthy, and A. K. Nigam, J. Alloys Compd. 545, 50 (2012).
Z. P. Li, J. Eisenmenger, C. W. Miller, and I. K. Schuller, Phys. Rev. Lett. 96(13), 137201 (2006).
V. K. Lakhani, B. Zhao, L. Wang, U. N. Trivedi, and K. B. Modi, J. Alloys Compd. 509(14), 4861 (2011).
S. Seki, T. Kurumaji, S. Ishiwata, H. Matsui, H. Murakawa, Y. Tokunaga, Y. Kaneko, T. Hasegawa, and Y. Tokura, Phys. Rev. B 82(6), 064424 (2010).
A. Moskvin, E. Vasinovich, and A. Shadrin, Magnetochemistry 8(4), 45 (2022).
W. He, H. L. Liu, H. Y. Wu, J. W. Cai, and Z. H. Cheng, Appl. Phys. Lett. 106(4), 042401 (2015).
J. Zawadzki, R. Ballou, B. Gorges, and R. Szymczak, Acta Phys. Pol. A 83(2), 209 (1993).
A. Duran, R. Escamilla, R. Escudero, F. Morales, and E. Verdin, Phys. Rev. Materials 2(1), 014409 (2018).
S. Oyama, M. Wakeshima, Y. Hinatsu, and K. Ohoyama, J. Phys. Condens. Matter 16(46), 8429 (2004).
M. Anas, S. Rajput, A. Singh, R. Kumar, T. Maitra, and V. K. Malik, J. Magn. Magn. Mater. 566, 170231 (2023).
R. Pal, B. Pal, S. Mondal, R. O. Sharma, T. Das, P. Mandal, and A. N. Pal, npj 2D Mater. Appl. 8(1), 30 (2024).
Z. D. Zhang, Z. J. Wu, B. H. Zhang, X. Q. Liu, and X. M. Chen, Ceramics International 50(1), 2452 (2024).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Pimentel, D.P. Structural and Magnetic Transformations from CuCl to Cu2Cl(OH)3 Induced by H2O and CuCl2. Jetp Lett. (2024). https://doi.org/10.1134/S0021364024601763
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S0021364024601763