Abstract—
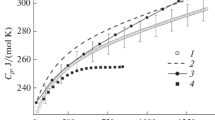
This paper presents temperature dependences of the molar heat capacity in the range 310–1360 K and lattice parameter in the range 298–1273 K for terbium hafnate with the pyrochlore structure, which has been characterized by X-ray diffraction, scanning electron microscopy, and chemical analysis.




Similar content being viewed by others
REFERENCES
Andrievskaya, E.R., Phase equilibria in the refractory oxide systems of zirconia, hafnia and yttria with rare-earth oxides, J. Eur. Ceram. Soc., 2008, vol. 28, pp. 2363–2388.https://doi.org/10.1016/jeurceramsoc.2008.01.009
Arsen’ev, P.A., Glushkova, V.B., Evdokimov, A.A., et al., Soedineniya redkozemel’nykh elementov. Tsirkonaty, gafnaty, niobaty, tantalaty, antimonaty (Rare-Earth Compounds: Zirconates, Hafnates, Niobates, Tantalates, and Antimonates), Moscow: Nauka, 1985.
Blanchard, P.E.R., Lio, S., Kennedy, B.J., Ling, C.D., Avdeev, M., Aitken, J.B., Cowie, B.C.C., and Tadish, A., Investigating the local structure of lanthanoid hafnates Ln2Hf2O7 via diffraction and spectroscopy, J. Phys. Chem. C, 2013, vol. 117, pp. 2266–2273.https://doi.org/10.1021/jp311329q
Popov, V.V., Menushenkov, A.P., Yaroslavtsev, A.A., Zubavichus, Ya.V., Gayanov, B.R., Jastrebov, A.A., Leshchev, D.S., and Chernikov, R.V., Fluorite–pyrochlore phase transition in nanostructured Ln2Hf2O7 (Ln = La – Lu), J. Alloys Compd., 2016, vol. 689, pp. 669–679. https://doi.org/101016/j.jallcom.2016.08.019
Menushenkov, A.P., Popov, V.V., Zubavichus, Ya.V., and Yaroslavtsev, A.A., Local peculiarities of the nanocrystalline structure of ternary oxides Ln2Hf2O7 (Ln = Gd, Tb, Dy), J. Struct. Chem., 2016, vol. 57, pp. 1450–1458.https://doi.org/10.1134/s0022476616070210
Rushton, M.J.D., Grimes, R.W., Stanek, C.R., and Owens, S., Predicted pyrochlore to fluorite disorder temperature for A2Zr2O7 compositions, J. Mater. Res., 2004, vol. 19, pp. 1603–1604.https://doi.org/10.1557/JMR.2004.0231
Subramanian, M.A., Aravamudan, G., and Subba Rao, G.V., Oxide pyrochlores – a review, Prog. Solid State Chem., 1983, vol. 15, pp. 55–143.https://doi.org/10.1016/0079-6786(83)90001-8
Kabanova, V.A., Popov, V.V., Zubavichus, Ya.V., Kulik, E.S., Yaroslavtsev, A.A., Chernikov, R.V., and Menushenkov, A.P., High local disorder in Tb2Hf2O7 pyrochlore oxide nanocrystals, J. Phys.: Conf. Ser., 2016, vol. 712, paper 012113.https://doi.org/10.1088/1742-6596/712/1/012113
Costa, G., Harder, B.J., Wiesner, V.L., Zhu, D.M., Bansal, N., Lee, K.N., Jacobson, N.S., Kapush, D., Ushakov, S.V., and Navrotsky, A., Thermodynamics of reaction between gas-turbine ceramic coatings and ingested CMAS corrodents, J. Am. Ceram. Soc., 2019, vol. 102, pp. 2948–2964.https://doi.org/10.1111/jace.16113
Fabrichnaya, O. and Seifert, H.J., Up-date of a thermodynamic database of the ZrO2–Gd2O3–Y2O3–Al2O3 system for TBC applications, J. Phase Equilib. Diffus., 2010, vol. 32, pp. 2–16.https://doi.org/10.1007/s11669-010-9815-4
Anand, V.K., Opherden, L., Xu, J., Adroja, D.T., Hillier, A.D., Biswas, P.K., Herrmannsdörfer, T., Uhlarz, M., Hornung, J., Wosnitza, J., Canévet, E., and Lake, B., Evidence for a dynamical ground state in the frustrated pyrohafnate Tb2Hf2O7, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, vol. 97, paper 094402.https://doi.org/10.1103/physrevb.97.094402
Guskov, V.N., Tyurin, A.V., Guskov, A.V., Gagarin, P.G., Khoroshilov, A.V., and Gavrichev, K.S., Thermal expansion and thermodynamic properties of gadolinium hafnate ceramics, Ceram. Int., 2020, vol. 46, pp. 12822–12829.https://doi.org/10.1016/j.ceramint.2020.02.052
Ryumin, M.A., Nikiforova, G.E., Tyurin, A.V., Khoroshilov, A.V., Kondrat’eva, O.N., Guskov, V.N., and Gavrichev, K.S., Heat capacity and thermodynamic functions of La2Sn2O7, Inorg. Mater., 2020, vol. 56, no. 1, pp. 97–104.https://doi.org/10.1134/S0020168520010148
Wieser, M.E., Atomic weights of the elements 2005 (IUPAC technical report), Pure Appl. Chem., 2006, vol. 78, pp. 2051–2066.https://doi.org/10.1351/pac200678112051
Gagarin, P.G., Guskov, A.V., Guskov, V.N., Tyurin, A.V., Khoroshilov, A.V., and Gavrichev, K.S., Dysprosium orthotantalate ceramics: thermal expansion and heat capacity, Ceram. Int., 2021, vol. 47, pp. 2892–2896.https://doi.org/10.1016/j.ceramint.2020.09072
Popov, V.V., Zubavichus, Y.V., Menushenkov, A.P., et al., Lanthanide effect on the formation and evolution of nanocrystalline structures in Ln2Hf2O7 compounds (Ln = Sm–Dy), Russ. J. Inorg. Chem., 2015, vol. 60, no. 1, pp. 18–25.https://doi.org/10.1134/s003602361501009x
Maier, C.G. and Kelley, K.K., An equation for representation of high temperature heat content data, J. Am. Chem. Soc., 1932, vol. 54, pp. 3243–3246.https://doi.org/10.1021/ja01347a029
Konings, R.J.M., Benes, O., Kovacs, O.A., Manara, D., Sedmidubsky, D., Gorokhov, L.N., Iorish, V.S., Yungman, V., and Shenyavskaya, O.E., The thermodynamic properties of the f-elements and their compounds: Part 2. The lanthanide and actinide oxides, J. Phys. Chem. Ref. Data, 2014, vol. 43, no. 14, paper 013101.https://doi.org/10.1063/1.4825256
Pankratz, L.B., Thermodynamic properties of elements and oxides, U.S. Bur. Mines Bull., 1982, vol. 672, p. 188.
ACKNOWLEDGEMENTS
This work was carried out using equipment of the JRC PMR IGIC RAS. The assistance of PhD A.A. Ashmarin in HTXRD studies is kindly appreciated.
Funding
This work was supported by the Russian Science Foundation grant no. 18-13-00025, https://rscf.ru/en/project/18-13-00025/.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
About this article
Cite this article
Guskov, A.V., Gagarin, P.G., Guskov, V.N. et al. Heat Capacity and Thermal Expansion of Terbium Hafnate. Inorg Mater 57, 710–713 (2021). https://doi.org/10.1134/S0020168521070074
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168521070074