Abstract—
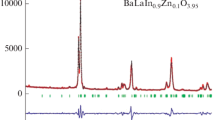
In this paper, we analyze the effect of acceptor doping (Zn2+ and Mg2+) in the indium sublattice on the transport properties of the BaLaInO4 phase with the Ruddlesden–Popper structure. The doping is shown to cause an increase in both the oxygen ion and proton conductivities of the material. The highest oxygen ion and proton conductivities are offered by the BaLaIn0.9Mg0.1O3.95 material.






Similar content being viewed by others
REFERENCES
Campbell-Lendrum, D. and Prüss-Ustün, A., Climate change, air pollution and noncommunicable diseases, Bull. W. H. O., 2019, vol. 97, pp. 160–161.https://doi.org/10.2471/BLT.18.224295
Akadiri, S.S., Alola, A.A., Olasehinde-William, G., and Etokakpan, M.U., The role of electricity consumption, globalization and economic growth in carbon dioxide emissions and its implications for environmental sustainability targets, Sci. Total Environ., 2020, vol. 708, paper 134 653.https://doi.org/10.1016/j.scitotenv.2019.134653
Davis, S.J., Caldeira, K., and Matthew, H.D., Future CO2 emissions and climate change from existing energy infrastructure, Science, 2010, vol. 329, pp. 1330–1333.https://doi.org/10.1126/science.1188566
Höök, M. and Tang, X., Depletion of fossil fuels and anthropogenic climate change – a review, Energy Policy, 2013, vol. 52, pp. 797–809. https://doi.org/10.1016/j.enpol.2012.10.046
Veziroğlu, T.N. and Şahin, S., 21st century’s energy: hydrogen energy system, Energy Convers. Manage., 2008, vol. 49, pp. 1820–1831.https://doi.org/10.1016/j.enconman.2007.08.015
Balat, M., Potential importance of hydrogen as a future solution to environmental and transportation problems, Int. J. Hydrogen Energy, 2008, vol. 33, pp. 4013–4029.https://doi.org/10.1016/j.ijhydene.2008.05.047
Momirlan, M. and Veziroğl, T.N., The properties of hydrogen as fuel tomorrow in sustainable energy system for a cleaner planet, Int. J. Hydrogen Energy, 2005, vol. 30, pp. 795–802.https://doi.org/10.1016/j.ijhydene.2004.10.011
Guangyao, M., Wany, L., and Dingkun, P., New solid state fuel cells – green power source for 21st century, Ionics, 1998, vol. 4, pp. 451–462.https://doi.org/10.1007/BF02375890
Fabbri, E., Bi, L., Pergolesi, D., and Traversa, E., Towards the next generation of solid oxide fuel cells operating below 600°C with chemically stable proton-conducting electrolytes, Adv. Mater., 2002, vol. 24, pp. 195–208.https://doi.org/10.1002/adma.201103102
Medvedev, D., Trends in research and development of protonic ceramic electrolysis cells, Int. J. Hydrogen Energy, 2019, vol. 44, paper 27711.https://doi.org/10.1016/j.ijhydene.2019.08.130
Dai, H., Kou, H., and Wang, H., Bi. l. electrochemical performance of protonic ceramic fuel cells with stable BaZrO3-based electrolyte: a mini-review, Electrochem. Commun., 2018, vol. 96, pp. 11–15.https://doi.org/10.1016/j.elecom.2018.09.001
Loureiro, F.J.A., Nasani, N., Reddy, G.S., Munirathnam, N.R., and Fagg, D.P., A review on sintering technology of proton conducting BaCeO3–BaZrO3 perovskite oxide materials for protonic ceramic fuel cells, J. Power Sources, 2019, vol. 438, paper 226991.https://doi.org/10.1016/j.jpowsour.2019.226991
Choi, S.M., An, H., Yoon, K.J., Kim, B., Lee, H.W., and Son, J.W., Electrochemical analysis of high-performance protonic ceramic fuel cells based on a columnar-structured thin electrolyte, Appl. Energy, 2019, vols. 233–234, pp. 29–36.https://doi.org/10.1016/j.apenergy.2018.10.043
Hideshima, N. and Hashizume, K., Effect of partial substitution of In by Zr, Ti and Hf on protonic conductivity of BaInO2.5, Solid State Ionics, 2010, vol. 181, pp. 1659–1664.https://doi.org/10.1016/j.ssi.2010.09.029
Tarasova, N. and Animitsa, I., The influence of anionic heterovalent doping on transport properties and chemical stability of F-, Cl-doped brownmillerite Ba2In2O5, J. Alloys Compd., 2018, vol. 739, pp. 353–359.https://doi.org/10.1016/j.jallcom.2017.12.317
Fujii, K., Shiraiwa, M., Esaki, Y., Yashima, M., Kim, S.J., and Lee, S., Improved oxide-ion conductivity of NdBaInO4 by Sr doping, J. Mater. Chem. A, 2015, vol. 3, paper 11985.https://doi.org/10.1039/C5TA01336D
Yang, X., Liu, S., Lu, F., Xu, J., and Kuang, X., Acceptor doping and oxygen vacancy migration in layered perovskite NdBaInO4-based mixed conductors, J. Phys. Chem. C, 2016, vol. 120, pp. 6416–6426.https://doi.org/10.1021/acs.jpcc.6b01530
Shiraiwa, M., Fujii, K., Esaki, Y., Kim, S.J., Lee, S., and Yashima, M., Crystal structure and oxide-ion conductivity of Ba1 + xNd1 − xInO4 − x/2, J. Electrochem. Soc., 2017, vol. 164, pp. F1392–F1399.https://doi.org/10.1149/2.0411713jes
Tarasova, N. and Animitsa, I., Protonic transport in oxyfluorides Ba2InO3F and Ba3In2O5F2 with Ruddlesden–Popper structure, Solid State Ionics, 2015, vol. 275, pp. 53–57.https://doi.org/10.1016/j.ssi.2015.03.025
Tarasova, N., Animitsa, I., Galisheva, A., and Korona, D., Incorporation and conduction of protons in Ca, Sr, Ba-Doped BaLaInO4 with Ruddlesden–Popper structure, Materials, 2019, vol. 12, paper 1668.https://doi.org/10.3390/ma12101668
Tarasova, N., Animitsa, I., Galisheva, A., and Pryakhina, V., Protonic transport in the new phases BaLaIn0.9M0.1O4.05 (M = Ti, Zr) with Ruddlesden–Popper structure, Solid State Sci., 2020, vol. 101, paper 106121.https://doi.org/10.1016/j.solidstatesciences.2020.106121
Titov, Yu.A., Belyavina, N.M., and Markiv, V.Ya., Synthesis and crystal structure of BaLaInO4 and SrLnInO4 (Ln−La, Pr), Rep. Natl. Acad. Sci. Ukr., 2009, vol. 10, pp. 160–166.
Shannon, R.D., Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, vol. 32, no. 5, pp. 751–767.https://doi.org/10.1107/S0567739476001551
Kharton, V.V., Marques, F.M.B., and Atkinson, A., Transport properties of solid oxide electrolyte ceramics: a brief review, Solid State Ionics, 2004, vol. 174, pp. 135–149.https://doi.org/10.1016/j.ssi.2004.06.015
Grimaud, A., Bassat, J.M., Mauvy, F., Simon, P., Canizares, A., Rousseau, B., Marrony, M., and Grenier, J.C., Transport properties and in-situ Raman spectroscopy study of BaCe0.9Y0.1O3 − δ as a function of water partial pressures, Solid State Ionics, 2011, vol. 191, pp. 24–31.https://doi.org/10.1016/j.ssi.2011.03.020
Poetzsch, D., Merkle, R., and Maier, J., Proton conductivity in mixed-conducting BSFZ perovskite from thermogravimetric relaxation, Phys. Chem. Chem. Phys., 2014, vol. 16, paper 16446.https://doi.org/10.1039/C4CP00459K
Kochetova, N., Animitsa, I., Medvedev, D., Demin, A., and Tsiakaras, P., Recent activity in the development of proton-conducting oxides for high-temperature applications, RSC Adv., 2014, vol. 6, pp. 73222–73268.https://doi.org/10.1039/c6ra13347a
Kochetova, N.A., Animitsa, I.E., and Neiman, A.Ya., Electric properties of solid solutions based on strontium tantalate with perovskite type structure. protonic conductivity, Russ. J. Electrochem., 2010, vol. 46, pp. 168–174.https://doi.org/10.1134/S1023193510020072
Kreuer, K.D., Proton-conducting oxides, Ann. Rev. Mater. Res., 2003, vol. 33, pp. 333–359.https://doi.org/10.1146/annurev.matsci.33.022802.091825
Haugsrud, R., High temperature proton conductors – fundamentals and functionalities, Diffus. Found., 2016, vol. 8, pp. 31–79.https://doi.org/10.4028/www.scientific.net/DF.8.31
Author information
Authors and Affiliations
Corresponding authors
Additional information
Translated by O. Tsarev
Rights and permissions
About this article
Cite this article
Tarasova, N.A., Galisheva, A.O. & Animitsa, I.E. Electrical Conductivity of BaLaIn0.9M0.1O3.95 (M = Mg, Zn)—New Complex Oxides with the Ruddlesden–Popper Structure. Inorg Mater 57, 60–67 (2021). https://doi.org/10.1134/S0020168521010155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168521010155