Abstract—
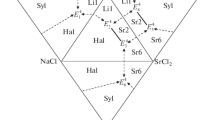
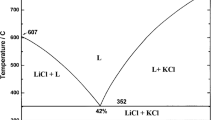
The T–x phase diagrams of the binary system LiCl–V2O5 and ternary system LiCl–LiVO3–V2O5 have been studied by differential, visual, and simultaneous thermal analysis. We have determined characteristics of eutectic and peritectic invariant points and mapped out the phase diagrams of these systems.


Similar content being viewed by others
REFERENCES
Spitsyn, V.I., Oksidnye bronzy (Oxide Bronzes), Moscow: Nauka, 1982.
Fotiev, A.A. and Ivankin, A.A., Vanadievye soedineniya shchelochnykh metallov i usloviya ikh obrazovaniya (Alkali Metal Vanadium Compounds and Conditions of Their Formation), Sverdlovsk: Ural. Otd. Akad. Nauk SSSR, 1970, issue 19.
Garkushin, I.K., Gubanova, T.V., and Frolov, E.I., Fazovye ravnovesiya s uchastiem solei litiya (Phase Equilibria Involving Lithium Salts), Yekaterinburg: Ural. Otd. Ross. Akad. Nauk, 2010.
Egunov, V.P., Vvedenie v termicheskii analiz (Introduction to Thermal Analysis), Samara: PO SamVen, 1996.
Trunov, V.K. and Kovba, L.M., Rentgenofazovyi analiz (Phase Analysis by X-Ray Diffraction), Moscow: Mosk. Gos. Univ., 1976.
Giller, R.A., Tablitsy mezhploskostnykh rasstoyanii (Tables of Interplanar Spacings), Moscow: Nedra, 1966, vol. 2.
Index to the Powder Diffraction File, West Conshohocken: Am. Soc. for Testing Materials, 1975.
Termicheskie konstanty veshchestv. Spravochnik (Thermal Constants of Substances: A Handbook), Glushko, V.P., Ed., Moscow: VINITI, 1981, issue 10, part 1.
Schreinemakers, F.A.H., In-, Mono-, and Divariant Equilibria, New York: Nordemann, 1946.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Tsarev
Rights and permissions
About this article
Cite this article
Gamataeva, B.Y., Kurbanova, S.N., Gasanaliev, A.M. et al. Phase Equilibria in the LiCl–LiVO3–V2O5 System. Inorg Mater 56, 136–141 (2020). https://doi.org/10.1134/S0020168520020053
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168520020053