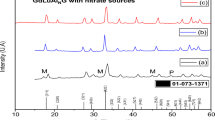
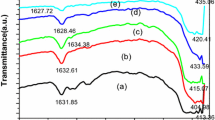
Abstract—
We have studied the kinetics of boehmite nanopowder formation during hydrothermal treatment of γ-Al2O3 nanopowder in a 1.5% HCl solution at 200, 170, and 150°C. The results demonstrate that the temperature-dependent reaction rate constant follows the Arrhenius equation. The Ea of the process has been determined to be 84 kJ/mol. The thermodynamics of γ-Al2O3 nanopowder conversion into boehmite during hydrothermal treatment at 150°C has been studied by differential scanning calorimetry. The heat of vaporization of water from a two-phase nanosystem (γ-Al2O3 + forming boehmite) has been determined to be 8, 16, and 22 kJ/mol H2O, which points to an active role of water with a low heat of vaporization in the initial stages of the hydrothermal treatment of the γ-Al2O3 nanopowder. The heat effect of the АlООН → γ-Al2O3 conversion in the nanopowders is lower than the reference value by 7 kJ/mol AlOOH, which is attributable to the small particle size and low structural perfection of the synthesized boehmite (AlOOH).






Similar content being viewed by others
REFERENCES
Panasyuk, G.P., Kozerozhets, I.V., Semenov, E.A., Azarova, L.A., Belan, V.N., and Danchevskaya, M.N., A new method for producing a nanosized γ-Al2O3 powder, Inorg. Chem., 2018, vol. 63, no. 10, pp. 1303–1308. https://doi.org/10.1134/S0036023618100157
Panasyuk, G.P., Kozerozhets, I.V., Voroshilov, I.L., Belan, V.N., Semenov, E.A., and Luchkov, I.V., The thermodynamic properties and role of water contained in dispersed oxides in precursor-boehmite conversion, based on the example of aluminum hydroxide and oxide under hydrothermal conditions in different environments, J. Phys. Chem. A, 2015, vol. 89, no. 4, pp. 592–597. https://doi.org/10.1134/S0036024415040196
Panasyuk, G.P., Belan, V.N., Voroshilov, I.L., Kozerozhets, I.V., Luchkov, I.V., Kondakov, D.F., and Demina, L.I., The study of hydrargillite and gamma-alumina conversion process in boehmite in different hydrothermal media, Theor. Found. Chem. Eng., 2013, vol. 47, no. 4, pp. 415–421. https://doi.org/10.1134/S0040579513040143
Tso, C.Y. and Chao Christopher, Y.H., Study of enthalpy of evaporation, saturated vapor pressure and evaporation rate of aqueous nanofluids, Int. J. Heat Mass Transfer, 2015, vol. 84, pp. 931–941. https://doi.org/10.1016/j.ijheatmasstransfer.2015.01.090
Panasyuk, G.P., Semenov, E.A., Kozerozhets, I.V., Yorov, Kh.E., Azarova, L.A., and Khol’kin, A.I., A new method of synthesis of nanosized metal oxide powders, Dokl. Chem., 2018, vol. 482, no. 1, pp. 201–203. https://doi.org/10.1134/S0012500818090033
Panasyuk, G.P., Semenov, E.A., Kozerozhets, I.V., Azarova, L.A., Belan, V.N., Danchevskaya, M.N., Nikiforova, G.E., Voroshilov, I.L., and Pershikov, S.A., A new method of synthesis of nanosized boehmite (AlOOH) powders with a low impurity content, Dokl. Chem., 2018, vol. 483, no. 1, pp. 272–274.
Chang Ho and Chang Yu-Chun, Fabrication of Al2O3 nanofluid by a plasma arc nanoparticles synthesis system, J. Mater. Proc. Technol., 2008, vol. 207, nos. 1–3, pp. 193–199. https://doi.org/10.1016/j.jmatprotec.2007.12.070
Chen Ruey-Hung, Phuoc Tran, X., and Martello, D., Effects of nanoparticles on nanofluid droplet evaporation, Int. J. Heat Mass Transfer, 2010, vol. 53, nos. 19–20, pp. 3677–3682. https://doi.org/10.1016/j.ijheatmasstransfer.2010.04.006
Tso, C.Y., Fu, S.C., and Chao Christopher, Y.H., A semi-analytical model for the thermal conductivity of nanofluids and determination of the nanolayer thickness, Int. J. Heat Mass Transfer, 2014, vol. 70, pp. 202–214. https://doi.org/10.1016/j.ijheatmasstransfer.2013.10.077
Sefiane, K. and Bennacer, R., Nanofluids droplets evaporation kinetics and wetting dynamics on rough heated substrates, Adv. Colloid Interface Sci., 2009, vols. 147–48, pp. 263–271. https://doi.org/10.1016/j.cis.2008.09.011
Phuoc Tran, X., Howard Bret, H., and Chyu Minking, K., Synthesis and rheological properties of cation-exchanged laponite suspensions, Colloids Surf., A, 2009, vol. 351, nos. 1–3, pp. 71–77. https://doi.org/10.1016/j.colsurfa.2009.09.039
Sefiane, K., Skilling, J., and MacGillivray, J., Contact line motion and dynamic wetting of nanofluid solutions, Adv. Colloid Interface Sci., 2008, vol. 138, no. 2, pp. 101–120. https://doi.org/10.1016/j.cis.2007.12.003
Wang, X.W., Xu, X.F., and Choi, S.U.S., Thermal conductivity of nanoparticle–fluid mixture, J. Thermophys. Heat Transfer, 1999, vol. 13, no. 4, pp. 474–480. https://doi.org/10.2514/2.6486
Prasher, R., Song, D., Wang, J., and Phelan, P., Measurements of nanofluid viscosity and its implications for thermal applications, Appl. Phys. Lett., 2006, vol. 89, no. 13, paper 133 108. https://doi.org/10.1063/1.2356113
Duangthongsuk, W. and Wongwises, S., Measurement of temperature-dependent thermal conductivity and viscosity of TiO2–water nanofluids, Exp. Therm. Fluid Sci., 2009, vol. 33, no. 4, pp. 706–714. https://doi.org/10.1016/j.expthermflusci.2009.01.005
Lee, S., Choi, S.U.S., Li, S.A., and Eastman, J.A., Measuring thermal conductivity of fluids containing oxide nanoparticles, J. Heat Transfer, 1999, vol. 121, no. 2, pp. 280–289. https://doi.org/10.1115/1.2825978
Das, S.K., Putra, N., Thiesen, P., and Roetzel, W., Temperature dependence of thermal conductivity enhancement for nanofluids, J. Heat Transfer, 2003, vol. 125, no. 4, pp. 567–574. https://doi.org/10.1115/1.1561080
Murshed, S.M.S., Leong, K.C., and Yang, C., A combined model for the effective thermal conductivity of nanofluids, Appl. Therm. Eng., 2009, vol. 29, nos. 11–12, pp. 2477–2483. https://doi.org/10.1016/j.applthermaleng.2008.12.018
Hisatake, K., Tanaka, S., and Aizawa, Y., Evaporation rate of water in a vessel, J. Appl. Phys., 1993, vol. 73, no. 11, pp. 7395–7401. https://doi.org/10.1063/1.354031
Madhusoodanan, M.R., Sajith, V., and Sobhan, C.B., Experimental investigation of phase change phenomena in nanofluids, Proc. Thermal Engineering Heat Transfer Summer Conf., 2007, pp. 859–863.
Barbes, B., Paramo, R., Blanco, E., and Casanova, C., Thermal conductivity and specific heat capacity measurements of CuO nanofluids, J. Therm. Anal. Calorim., 2014, vol. 115, no. 2, pp. 1883–1891. https://doi.org/10.1007/s10973-013-3518-0
Mostafizur, R.M., Bhuiyan, M.H.U., Saidur, R., and Abdul Aziz, A.R., Thermal conductivity variation for methanol based nanofluids, Int. J. Heat Mass Transfer, 2014, vol. 76, pp. 350–356. https://doi.org/10.1016/j.ijheatmasstransfer.2014.04.040
Mostafizur, R.M., Abdul Aziz, A.R., Saidur, R., Bhuiyan, M.H.U., and Mahbubul, I.M., Effect of temperature and volume fraction on rheology of methanol based nanofluids, Int. J. Heat. Mass. Transfer, 2014, vol. 77, pp. 765–769. https://doi.org/10.1016/j.ijheatmasstransfer.2014.05.055
Bhuiyan, M.H.U., Saidur, R., Amalina, M.A., and Mostafizur, R.M., Measurement of latent heat of vaporization of nanofluids using calorimetric technique, J. Therm. Anal. Calorim., 2015, vol. 122, no. 3, pp. 1341–1346. https://doi.org/10.1007/s10973-015-4747-1
Lippincot, E.R., Stromberg, R.R., Grant, W.H., and Cessac, G.L., Polywater, Science, 1969, vol. 164, no. 3887, pp. 1482–1487. https://doi.org/10.1126/science.164.3887.1482
Derjaguin, B.V. and Churaev, N.V., Nature of “anomalous water,” Nature, 1973, vol. 244, no. 5416, pp. 430–431. https://doi.org/10.1038/244430a0
Usov, L.V., Approximate calculation of kinetic parameters of the crystallization of corundum from boehmite under hydrothermal conditions, Russ. J. Inorg. Chem., 1996, vol. 41, no. 9, pp. 1383–1387.
Ivakin, Yu.D., Danchevskaya, M.N., Ovchinnikova, O.G., Murav’eva, G.P., and Kreisberg, V.A., Kinetics and mechanism of doped corundum structure formation in water fluid, Sverkhkrit. Flyuidy: Teor. Praktika, 2008, vol. 3, no. 4, pp. 11–34.
Panasyuk, G.P., Luchkov, I.V., Kozerozhets, I.V., Shabalin, D.G., and Belan, V.N., Effect of pre-heat treatment and cobalt doping of hydrargillite on the kinetics of the hydrargillite–corundum transformation in supercritical water fluid, Inorg. Mater, 2013, vol. 49, no. 9, pp. 899–903. https://doi.org/10.1134/S0020168513090136
Madarasz, J., Pocol, G., Novak, C., Cobos, F.T., and Gal, S., Studies on isothermal kinetics of some reactions of aluminum oxides and hydroxides, J. Therm. Anal., 1992, vol. 38, pp. 445–454. https://doi.org/10.1007/BF01915509
Rivkin, S.L. and Aleksandrov, A.A., Teplofizicheskie svoistva vody i vodyanogo para (Thermophysical Properties of Water and Steam), Moscow: Energiya, 1980.
Bokhimi, X., Toledo-Antonio, J.A., Guzman-Castillo, M.L., Mar-Mar, B., Hernandez-Beltran, F., and Navarrete, J., Dependence of boehmite thermal evolution on its atom bond lengths and crystallite size, J. Solid State Chem., 2001, vol. 161, pp. 319–326. https://doi.org/10.1006/jssc.2001.9320
Funding
This work was supported by the Russian Federation Ministry of Science and Higher Education (state research target for the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, basic research).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panasyuk, G.P., Kozerozhets, I.V., Semenov, E.A. et al. Thermodynamics and Kinetics of γ-Al2O3 and AlOOH Transformations under Hydrothermal Conditions. Inorg Mater 55, 920–928 (2019). https://doi.org/10.1134/S0020168519090127
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168519090127