Abstract
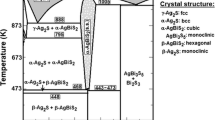
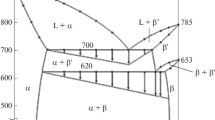
The standard thermodynamic properties of the intermetallic compounds Ag3Sb and Ag6Sb (the minerals dyscrasite and allargentum, respectively) in the Ag-Sb system have been determined in the temperature range 350–500 K at atmospheric pressure using a solid-state galvanic cell (emf method). The results have been used to propose recommended standard thermodynamic functions of formation at 298.15 K and 105 Pa. The Gibbs energy, entropy, and enthalpy of Ag3Sb(cr) are −5934 ± 1277 J/mol, 195.89 ± 2.54 J/(mol K), and 841 ± 1046 J/mol, respectively, and those of Ag6Sb(cr) are −6637 ± 3064 J/mol, 332.40 ± 5.99 J/(mol K), and 2778 ± 2491 J/mol, respectively.
Similar content being viewed by others
References
Wachtler, M., Winter, M., and Besenhard, J.O., Anodic materials for rechargeable Li-batteries, J. Power Sources, 2002, vol. 105, pp. 151–160.
Vaughey, J.T., Fransson, L., Swinger, H.A., et al., Alternative anode materials for lithium-ion batteries: A study of Ag3Sb, J. Power Sources, 2003, vol. 119–121, pp. 64–68.
Wang, F., Yao, G., Xu, M., et al., Ag-Sb composite prepared by chemical reduction method as new anode materials for lithium-ion batteries, Mater. Sci. Eng., B, 2011, vol. 176, pp. 442–445.
Masson, D.B. and Kirkpatrick, B.K., Equilibrium solidification solder alloys of Sn-Ag-Sb thermal fatigueresistant, J. Electron. Mater., 1986, vol. 15, no. 6, pp. 349–353.
Lee, H.-T., Chen, M.-H., Jao, H.-M., and Hsu, C.-J., Effect of adding Sb on microstructure and adhesive strength of Sn-Ag solder joints, J. Electron. Mater., 2004, vol. 33, no. 9, pp. 1048–1054.
Weibke, F. and Efinger, I., Elektrochemische Untersuchungen am System Silber-Antimon, Z. Elektrochem., 1940, vol. 46, no. 2, pp. 61–69.
Verduch, A.G. and Wagner, C., Contributions to the thermodynamics of the systems PbS-Sb2S3, Cu2S-Sb2S3, Ag2S-Sb2S3 and Ag-Sb, J. Phys. Chem., 1957, vol. 61, pp. 558–562.
Birks, N., Rickert, H., and Wagner, C., Berechnung von Metall-Schlacken-Gleichgewichten sowie Gleichgewichten zwischen binären flässigen oder festen Legierungen und festen Oxyden oder Sulfiden mit einer Anwendung auf das System Ag-Sb-S, Z. Elektrochem., 1962, vol. 66, no. 3, pp. 266–269.
Lukashenko, G.M., Eremenko, V.N., and Sidorko, V.R., Thermodynamic study of the silver-antimony system, Zh. Neorg. Khim., 1964, no. 9, pp. 220–221.
Osadchii, E., Boström, D., Lunin, S., and Rosén, E., Thermodynamic properties of dyscrasite (Ag3Sb) at isostatic pressures 4000–8000 bar, Phys. Chem. Miner., 1998, vol. 25, pp. 288–291.
Wallbrecht, P.C., Blachnik, R., and Mills, K.C., The heat capacity and enthalpy of some Hume-Rothery phases formed by copper, silver and gold. Part I. Cu + Sb, Ag + Sb, Au + Sb, Au + Bi systems, Thermochim. Acta, 1981, vol. 45, pp. 189–198.
Kleppa, O.J., Heat of formation of solid and liquid alloys in the systems silver-cadmium, silver-indium and silver-antimony at 450°, J. Phys. Chem., 1956, vol. 60, no. 7, pp. 846–852.
Weibke, F. and Efinger, I., Der Aufbau der Legierungen des Systems Silber-Antimon, Z. Elektrochem., 1940, vol. 46, no. 2, pp. 53–60.
Feschotte, P., Monachon, F., and Durussel, Ph., The binary system Sb-Ag: A revision of the Ag3Sb phase boundaries, J. Alloys Compd., 1992, vol. 186, no. 2, pp. L17–L18.
Cipriani, C., Corazza, M., and Mazzetti, G., Reinvestigation of natural silver antimonides, Eur. J. Mineral., 1996, vol. 8, pp. 1347–1350.
Hassam, S., Bahari, Z., and Legendre, B., Phase diagrams of the Ag-Bi-Sb ternary system, J. Alloys Compd., 2001, vol. 315, pp. 211–217.
Voronin, M.V. and Osadchii, E.G., Determination of thermodynamic properties of silver selenide by the galvanic cell method with solid and liquid electrolytes, Russ. J. Electrochem., 2011, vol. 47, no. 4, pp. 420–426.
Osadchii, E.G. and Rappo, O.A., Determination of standard thermodynamic properties of sulfides in the Ag-Au-S system by means of a solid-state galvanic cell, Am. Mineral., 2004, vol. 89, pp. 1405–1410.
Osadchii, E.G. and Echmaeva, E.A., The system Ag-Au-Se: Phase relations below 405 K and determination of standard thermodynamic properties of selenides by solid-state galvanic cell technique, Am. Mineral., 2007, vol. 92, pp. 640–647.
Zhdanov, N.N., Osadchii, E.G., and Zotov, A.V., Multipurpose system for electrochemical measurements in hydrothermal and condensed media, Sbornik materialov XV Rossiiskogo soveshchaniya po eksperimental’noi mineralogii (Proc. XV Russ. Conf. on Experimental Mineralogy), Syktyvkar: Geoprint, 2005, pp. 166–168.
Robie, R.A. and Hemingway, B.S., Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar (105 pascals) pressure and at high temperatures, U.S. Geol. Surv. Bull., 1995, no. 2131.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.V. Voronin, E.G. Osadchii, 2013, published in Neorganicheskie Materialy, 2013, Vol. 49, No. 6, pp. 585–589.
Rights and permissions
About this article
Cite this article
Voronin, M.V., Osadchii, E.G. Standard thermodynamic properties of Ag3Sb and Ag6Sb evaluated by EMF measurements. Inorg Mater 49, 550–554 (2013). https://doi.org/10.1134/S0020168513060186
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168513060186