Abstract
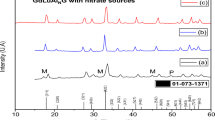
We have synthesized Sr1 − x Pb x FeO3 − δ (x = 0, 0.05, 0.1, 0.15, 0.2, 0.3, 0.5) perovskite-like materials and studied their structure by X-ray diffraction, Mössbauer spectroscopy, and electron microscopy. According to the X-ray diffraction data, the Pb solubility limit in the perovskite structure is x ≈ 0.15. The materials with x = 0.05 and 0.1 contained Pb1.33Sr0.67Fe2O5 inclusions 10–30 nm in size. Using chronopotentiometry and temperature-programmed desorption, we have estimated oxygen mobility in the materials with x = 0.05 and 0.1. The results demonstrate that Pb doping increases oxygen mobility in the strontium-ferrite-based materials.
Similar content being viewed by others
References
Bouwmeester, H.J.M. and Gellings, P.J., Dense Ceramic Membranes for Oxygen Separation, in The CRC Handbook of Solid State Electrochemistry, Enschede: CRC Press, 1996, pp. 482–542.
Kozhevnikov, V.L., Leonidov, I.A., Patrakeev, M.V., et al., Electrical Properties of the Ferrite SrFeOy at High Temperatures, J. Solid State Chem., 2000, no. 158, pp. 320–326.
Grenier, J.C., Ea, N., Pouchard, M., and Hagenmuller, P., Structural Transitions at High Temperature in Sr2Fe2O5, J. Solid State Chem., 1985, no. 58, pp. 243–252.
Nemudry, A. and Uvarov, N., Nanostructuring in Composites and Grossly Nonstoichiometric or Heavily Doped Oxides, Solid State Ionics, 2006, vol. 177, pp. 2491–2494.
Orlovskaya, N., Browning, N., and Nicholls, A., Ferroelasticity in Mixed Conducting LaCoO3 Based Perovskites: A Ferroelastic Phase Transition, Acta Mater., 2003, no. 51, pp. 5063–5071.
Nemudry, A., Goldberg, E.L., Aguirre, M., and Alario-Franco, M.A., Electrochemical Topotactic Oxidation of Nonstoichiometric Perovskites at Ambient Temperature, Solid State Sci., 2002, vol. 4, pp. 677–690.
Moreo, A., Electronic Phase Separation and Giant Cluster Coexistence in Manganites, J. Electron Spectrosc. Relat. Phenom., 2001, vols. 117–118, pp. 251–264.
Pyatiletova, E.B., Nemudry, A.P., and Lyakhov, N.Z., Synthesis and Properties of Sr1 − x MxCo0.8 − y AlyFe0.2O3 − z Perovskite-like Oxides, Inorg. Mater., 2007, vol. 43, no. 10, pp. 1134–1142.
Nemudry, A., Weiss, M., Gainutdinov, I., et al., Room Temperature Electrochemical Redox Reactions of the Defect Perovskite SrFeO2.5 + x , Chem. Mater., 1998, no. 10, pp. 2403–2411.
Abakumov, A.M., Haderman, J., Bals, S., et al., Crystallographic Shear Structures As Route to Anion-Deficient Perovskites, Angew. Chem., Int. Ed., 2006, vol. 45, pp. 6697–6700.
Raynova-Schwarten, V., Massa, W., and Babel, D.Z., A New Lead Strontium Ferrate: The Crystal Structure of the Phase Pb4Sr2Fe6O15, Z. Anorg. Allg. Chem., 1997, vol. 623, pp. 1048–1054.
Wen, C.J., Ho, C., Boukamp, B.A., et al., Use of Electrochemical Methods to Determine Chemical Diffusion Coefficient in Alloys Application to “LiAl”, Int. Met. Rev., 1981, vol. 5, pp. 253–268.
Markov, A.A., Savinskaya, O.A., Patrakeev, M.V., et al., Structural Features, Nonstoichiometry and High-Temperature Transport in SrFe1 − x MoxO3 − δ, J. Solid State Chem., 2009, vol. 182, pp. 799–806.
Falconer, J.L. and Madix, R.J., Desorption Rate Isotherms in Flash Desorption Analysis, J. Catal., 1977, vol. 48, pp. 262–268.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.V. Belen’kaya, O.A. Savinskaya, M.A. Neklyudova, A.P. Nemudry, 2011, published in Neorganicheskie Materialy, 2011, Vol. 47, No. 12, pp. 1476–1485.
Rights and permissions
About this article
Cite this article
Belen’kaya, I.V., Savinskaya, O.A., Neklyudova, M.A. et al. Structure of Sr1 − x Pb x FeO3 − δ (0 < x < 0.5) perovskite-like materials. Inorg Mater 47, 1347–1355 (2011). https://doi.org/10.1134/S002016851111001X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002016851111001X