Abstract
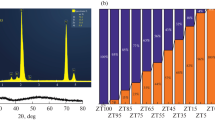
The phase formation and reaction kinetics in the TiO2-Cr2O3 system have been studied by x-ray diffraction and electron microscopy. The Cr2O3 solubility in TiO2 has been accurately determined, and the rate parameters of the formation of solid solutions in this system have been evaluated. The results demonstrate that Cr2O3 dissolves in rutile and not in anatase. Cr2O3 markedly reduces the temperature of the anatase-rutile phase transition.
Similar content being viewed by others
References
Belen’kii, E.V. and Riskin, N.V., Khimiya i tekhnologiya pigmentov (Chemistry and Technology of Pigments), Leningrad: Khimiya, 1974.
Luchinskii, G.P., Khimiya titana (The Chemistry of Titanium), Moscow: Khimiya, 1971.
Khazin, L.G., Dvuokis’ titana (Titanium Dioxide), Leningrad: Khimiya, 1970.
Goroshchenko, Ya.T., Khimiya titana (The Chemistry of Titanium), Kiev: Naukova Dumka, 1970.
Shannon, R.D. and Rask, G.A., Kinetics of the Anatase-Rutile Transformation, J. Am. Ceram. Soc., 1965, vol. 48, no. 8, pp. 391–397.
Sun Yijun and Egawa Takashi, High Anatase-Rutile Transformation Temperature in Anatase Ninania Nanoparticles Prepared by Metalorganic Chemical Vapor Deposition, Jpn. J. Appl. Phys., 2002, vol. 41, no. 8B, pp. 945–948.
Berezhnoi, A.S., Mnogokomponentnye sistemy okislov (Multicomponent Oxide Systems), Kiev: Naukova Dumka, 1970.
Reznichenko, L.A., Shilkina, L.A., Titov, S.V., and Razumovskaya, O.N., Magneli Phases in Ti-Containing Mixed Oxides and Their Solid Solutions, Kristallografiya, 2003, vol. 48, no. 3, pp. 493–427.
Radecka, M. and Zakrzewsra, K., Study of the TiO2-Cr2O3 System for Photoelectrolytic Decomposition of Water, Solid State Ionics, 2003, vol. 157, no. 1, pp. 386–397.
Camara, O.R., Electrochemical Response of Titanium and Chromium Mixed Oxides on Titanium Substrates, J. Electroanal. Chem., 1990, vol. 248, no. 1, pp. 155–172.
Bourikas, K., Fountzoula, Ch., and Kordulis, Ch., Monolayer Transition Metal Supported on Titania Catalysts for the Selective Catalytic Reduction of NO by NH3, Appl. Catal., B: Environmental, 2004, vol. 52, no. 2, pp. 145–153.
Zolotov, Yu.A., Osnovy analiticheskoi khimii (Fundamentals of Analytical Chemistry), Moscow: Vysshaya Shkola, 1999.
Kovel’, N.S., Viktorov, V.V., Evdokimov, I.V., and Fotiev, A.A., Formation of ((CrxAl1 − x )2O3 Solid Solutions during Cocalcination of Aluminum and Chromium Hydroxides, Izv. Akad. Nauk SSSR, Neorg. Mater., 1989, vol. 25, no. 7, pp. 1160–1164.
Morrison, S., The Chemical Physics of Surfaces, New York: Plenum, 1977.
Rao, C.N.R. and Gopalakrishnan, J., New Directions in Solid State Chemistry: Structure, Synthesis, Properties, Reactivity, and Materials Design, Cambridge: Cambridge Univ. Press, 1986.
Tret’yakov, Yu.D., Tverdofaznye reaktsii (Solid-State Reactions), Moscow: Khimiya, 1978.
Viktorov, V.V., Evdokimov, I.V., and Kovel’, M.S., Formation Kinetics of Al2O3-Cr2O3 Solid Solutions, Zh. Fiz. Khim., 1990, vol. 64, no. 7, pp. 1820–1824.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.A. Belaya, V.V. Viktorov, 2008, published in Neorganicheskie Materialy, 2008, Vol. 44, No. 1, pp. 68–72.
Rights and permissions
About this article
Cite this article
Belaya, E.A., Viktorov, V.V. Formation of solid solutions in the TiO2-Cr2O3 system. Inorg Mater 44, 62–66 (2008). https://doi.org/10.1134/S002016850801010X
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002016850801010X