Abstract
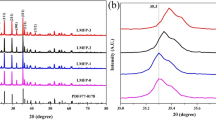
We have investigated LiNi1 − x − y Co x Mn y O2 (x = y = 0.1, 0.2, 0.33) cathode materials synthesized from mechanically activated mixtures of lithium hydroxide and nickel cobalt manganese hydroxide. The materials have a layered structure (sp. gr. \(R\bar 3m\)). Their unit-cell volume and the degree of disordering in their structure decrease with decreasing nickel content. According to x-ray photoelectron spectroscopy data, the major states of the transition-metal ions in the surface layer of the materials are Ni2+, Co3+, and Mn4+. With increasing nickel content, the Ni 2p 3/2 and Co 2p 3/2 binding energies increase, attesting to changes in M-O bond covalence. The highest specific electrochemical capacity, ∼170 mA h/g, is offered by LiNi0.6Co0.2Mn0.2O2. The position of redox peaks in the differential capacity curves of the three materials depends on composition: with increasing nickel content, the peaks shift to lower voltages.
Similar content being viewed by others
References
Yabuuchi, N. and Ohzuku, T., Novel Lithium Insertion Material of LiCo1/3Ni1/3Mn1/3O2 for Advanced Lithium-Ion Batteries, J. Power Sources, 2003, vols. 119–121, pp. 171–174.
Koyama, Y., Makimura, Y., Tanaka, I., et al., Systematic Research on Insertion Materials Based on Superlattice Models in a Phase Triangle of LiCoO2-LiNiO2-LiMnO2: I. First-Principles Calculation on Electronic and Crystal Structures, Phase Stability and New LiNi1/2Mn1/2O2 Material, J. Electrochem. Soc., 2004, vol. 151, no. 9, pp. A1499–A1506.
Koyama, Y., Yabuuchi, N., Tanaka, I., et al., Solid-State Chemistry and Electrochemistry of LiCo1/3Ni1/3Mn1/3O2 for Advanced Lithium-Ion Batteries: I. First-Principles Calculation on the Crystal and Electronic Structures, J. Electrochem. Soc., 2004, vol. 151, no. 10, pp. A1545–A1551.
Yabuuchi, N., Koyama, Y., Nakayama, N., and Ohzuku, T., Solid-State Chemistry and Electrochemistry of LiCo1/3Ni1/3Mn1/3O2 for Advanced Lithium-Ion Batteries: II. Preparation and Characterization, J. Electrochem. Soc., 2005, vol. 152, no. 7, pp. A1434–A1440.
Jiang, J., Eberman, K.W., Krause, L.J., and Dahn, J.R., Structure, Electrochemical Properties, and Thermal Stability Studies of Cathode Materials in the xLi[Mn1/2Ni1/2]O2 · yLiCoO2 · zLi[Li1/3Mn2/3]O2 Pseudoternary System (x + y + z = 1), J. Electrochem. Soc., 2005, vol. 152, no. 9, pp. A1879–A1889.
Ngala, J.K., Chernova, N.A., Ma, M., et al., The Synthesis, Characterization, and Electrochemical Behavior of the Layered LiNi0.4Mn0.4Co0.2O2, J. Mater. Chem., 2004, vol. 14, pp. 214–220.
Tran, N., Croguennec, L., Jordy, C., et al., Influence of the Synthesis Route on the Electrochemical Properties of LiNi0.425Mn0.425Co0.15O2, Solis State Ionics, 2005, vol. 176, pp. 1539–1547.
Liao, P.Y., Duh, J.G., and Sheen, S.R., Effect of Mn Content on the Microstructure and Electrochemical Performance of LiNi0.75 − x Co0.25Mn x O2 Cathode Materials, J. Electrochem. Soc., 2005, vol. 152, no. 9, pp. A1695–A1700.
Cao, H., Zhang, Y., Zhang, J., and Xia, B., Synthesis and Electrochemical Characteristics of Layered LiNi0.6Co0.2Mn0.2O2 Cathode Material for Lithium Ion Batteries, Solid State Ionics, 2005, vol. 176, pp. 1207–1211.
Liu, Z., Yu, A., and Lee, J.Y., Synthesis and Characterization of LiNi1 − x − y Co x Mn y O2 as the Cathode Materials of Secondary Lithium Batteries, J. Power Sources, 1999, vols. 81–82, pp. 416–419.
Cho, J., Kim, T.-J., Kim, J., et al., Synthesis, Thermal, and Electrochemical Properties of AlPO4-Coated LiNi0.8Co0.1Mn0.1O2 Cathode Materials for a Li-Ion Cell, J. Electrochem. Soc., 2004, vol. 151, no. 11, pp. A1899–A1904.
Avvakumov, E., Senna, M., and Kosova, N., Soft Mechanochemical Synthesis. A Basics for New Chemical Technologies, Dordrecht: Kluwer, 2001.
Guerlou-Demourgues, L., Denage, C., and Delmas, C., New Manganese-Substituted Nickel Hydroxides: Part 1. Crystal Chemistry and Physical Characterization, J. Power Sources, 1994, vol. 52, pp. 269–274.
Eikhler, V.V., Lukashenko, A.I., Makukha, V.K., and Kosova, N.V., Automatic System for Cycling Electrochemical Cells, Elektrokhim. Energ., 2005, vol. 5, no. 2, pp. 135–138.
Julien, C., Local Structure and Electrochemistry of Lithium Cobalt Oxides and Their Doped Compounds, Solid State Ionics, 2003, vol. 157, pp. 57–71.
Kosova, N., Devyatkina, E., and Kaichev, V., Structural and Electronic Properties of Layered LiNi1 − x Mn x O2 Prepared Using Mechanical Activation, in Lithium Battery Discussion: Electrode Materials, Bordeaux: Arcachon, 2005, pp. 50–51.
McIntyre, N.S. and Cook, M.G., X-ray Photoelectron Studies on Some Oxides and Hydroxides of Cobalt, Nickel, and Copper, Anal. Chem., 1975, vol. 47, pp. 2208–2213.
Li, C.P., Proctor, A., and Hercules, D.M., Curve Fitting Analysis of ESCA Ni 2p Spectra of Nickel-Oxygen Compounds and Ni/Al2O3 Catalysts, Appl. Spectrosc., 1984, vol. 38, pp. 880–886.
Shaju, K.M., Subba Rao, G.V., and Chowdari, B.V.R., X-ray Photoelectron Spectroscopy and Electrochemical Behaviour of 4 V Cathode, Li(Ni1/2Mn1/2)O2, Electrochim. Acta, 2003, vol. 48, pp. 1505–1514.
Carley, A.F., Jackson, S.D., Shea, J.N., and Roberts, M.W., The Formation and Characterization of Ni3+—An X-ray Photoelectron Spectroscopic Investigation of Potassium-Doped Ni(110)-O, Surf. Sci., 1999, vol. 440, pp. L868–L874.
Amine, K., Tukamoto, H., Yasuda, H., and Fujita, Y., A New Three-Volt Spinel Li1 + x Mn1.5Ni0.5O4 for Secondary Lithium Batteries, J. Electrochem. Soc., 1996, vol. 143, pp. 1607–1613.
Montoro, L.A., Abbate, M., Almeida, E.C., and Rosolen, J.M., Electronic Structure of the Transition Metal Ions in LiCoO2, LiNiO2, and LiCo0.5Ni0.5O2, Chem. Phys. Lett., 1999, vol. 309, pp. 14–18.
Shaju, K.M., Subba Rao, G.V., and Chowdari, B.V.R., Performance of Layered Li(Ni1/3Co1/3Mn1/3)O2 as Cathode for Li-Ion Batteries, Electrochim. Acta, 2003, vol. 48, pp. 145–151.
Oku, M., Hirokawa, K., and Ikeda, S., X-ray Photoelectron Spectroscopy of Manganese-Oxygen Systems, J. Electron Spectrosc. Relat. Phenom., 1975, vol. 7, pp. 465–473.
Kosova, N.V., Kaichev, V.V., Bukhtiyarov, V.I., et al., Electronic State of Cobalt and Oxygen Ions in Stoichiometric and Nonstoichiometric Li1 + x CoO2 before and after Delithiation according to XPS and DRS, J. Power Sources, 2003, vols. 119–121, pp. 669–673.
Sun, Y., Ouyang, C., Wang, Z., et al., Effect of Co Content on Rate Performance of LiMn0.5 − x Co x Ni0.5 − x O2 Cathode Materials for Lithium-Ion Batteries, J. Electrochem. Soc., 2004, vol. 151, pp. A504–A508.
Hwang, B.J., Tsai, Y.W., Chen, C.H., and Santhanam, R., Influence of Mn Content on the Morphology and Electrochemical Performance of LiNi1 − x − y Co x Mn y O2 Cathode Materials, J. Mater. Chem., 2003, vol. 13, pp. 1962–1968.
Kuiper, P., Kruizinga, G., Ghijsen, J., et al., Character of Holes in Li x Ni1 − x O2 and Their Magnetic Behavior, Phys. Rev. Lett., 1989, vol. 62, pp. 221–224.
Bukhtiyarov, V.I., Hävecker, M., Kaichev, V.V., et al., The Nature of Atomic Oxygen Species on Silver: Photoelectron Spectroscopy and X-ray Absorption Studies, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, vol. 67, no. 23, p. 235422.
Author information
Authors and Affiliations
Additional information
Original Russian Text © N.V. Kosova, E.T. Devyatkina, V.V. Kaichev, 2007, published in Neorganicheskie Materialy, 2007, Vol. 43, No. 2, pp. 227–235.
Rights and permissions
About this article
Cite this article
Kosova, N.V., Devyatkina, E.T. & Kaichev, V.V. LiNi1 − x − y Co x Mn y O2 (x = y = 0.1, 0.2, 0.33) cathode materials prepared using mechanical activation: Structure, state of ions, and electrochemical performance. Inorg Mater 43, 185–193 (2007). https://doi.org/10.1134/S0020168507020161
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0020168507020161