Abstract
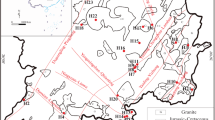
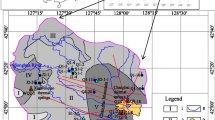
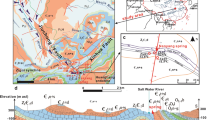
Changbai Mountain in China has been explored as a potential area for enriching mineral fluids including thermal springs and cold springs mineral water. The hydrogeochemical characteristics and reservoir temperatures of the nine thermal springs and fifteen cold springs were evaluated by hydrogeochemical analysis, statistical analysis and geothermometry analysis. The results showed that the concentrations of ions of thermal springs were higher than those of cold springs. \({\text{HCO}}_{3}^{ - }\) and Na+ were the most abundant anion and cation in the thermal springs, and hydrochemical type was Na–HCO3. While the cold springs were enriched in Ca2+, Mg2+, \({\text{HCO}}_{3}^{ - }\) and \({\text{SO}}_{4}^{{2 - }}\), and hydrochemical types were Ca · Mg–HCO3 · SO4, Ca–HCO3, Ca · Mg · Na–HCO3 and Ca · Mg · Na–SO4 · HCO3. The age of thermal springs were larger than 62 years. Olivine and pyroxene were under-saturated for all thermal springs; and olivine, pyroxene, dolomite and fluorite were under-saturated for all cold springs. All the thermal and cold springs samples can be divided into three groups by cluster test, and all the thermal spring samples fell into the part of immature waters, the mixing ration of cold and hot water ranged 12.70 to 36.95%. The reservoir temperatures of WQ1 to WQ9 were between 96.88 and 174.28°C using silica geothermometry.









Similar content being viewed by others
REFERENCES
M. Ahmad, W. Akram, N. Ahmad, et al., “Assessment of reservoir temperatures of thermal springs of the northern areas of Pakistan by chemical and isotope geothermometry,” Geothermics 31 (5), 613–631 (2002).
S. Arab and A. Alshikh, “Estimating of some trace elements in mineral water in the Kingdom of Saudi Arabia,” Life Sci J. 7, 133–137 (2010).
Z. D. Bai, D. B. Xu, and B. L. Zhang, “Study on type and phase of Quaternary explosive volcanism in Longgang volcanic cluster,” Acta Petrologica Sinica 22, 1473–1480 (2006).
R. C. Borden, C. A. Gomez, and M. T Becker, “Geochemical indicators of intrinsic Bioremediation,” Ground Water. 33, 180–189 (1995).
L. Calmbach, Aquachem Computer Code–Version 3.7.42. (Lewis Publishers, New York, 1997).
I. Can, “A new improved Na/K geothermometers by artificial neural networks,” Geothermics 31, 751–760 (2002).
Y. Q. Cao, K. R. Hu, and Z. Y. Hu, “Hydrogeochemical reaction–migration–differentiation model,” J. Changchun Univ. Sci. Technol. 30, 252–256 (2000).
J. V. Cruz, P. Freire, and A. Costa, “Mineral waters characterization in the Azores archipelago (Portugal),” J. Volcanol. Geotherm. Res. 190, 353–364 (2010).
F. Ersoy and S. Calik SÖnmez, “Hydrogeochemical and isotopic characteristics of the Ilica geothermal system (Erzurum, Turkey),” Environ. Earth Sci. 72, 4451–4462 (2014).
R. O. Fournier, “Chemical geothermometers and mixing models for geothermal systems,” Geothermics 5, 41–50 (1977).
R. O. Fournier, “A revised equation for the Na/K geothermometer,” Geotherm. Res Coun Trans. 3, 221–224 (1979).
R. O. Fournier, “Water geothermometers applied to geothermal energy,” in Applications of Geochemistry in Geothermal Reservoir Development, Ed. by F. D’Amore, (A UNITAR/UNDP Centre on Small Resources, Rome, Italy, 1991), pp. 37–69.
R. O. Fournier and A. H. Truesdell, “An empirical Na–K–Ca geothermometer for natural waters,” Geochim. Acta. 37: 1255–1275 (1973).
C. Fouillac and G. Michard, “Sodium/lithium ratio in water applied to geothermometry of geothermal reservoirs,” Geothermics 10, 55–70 (1981).
R. O. Fournier and R. W. Potter II, “A revised and expanded silica (quartz) geothermometer,” Geotherm. Res. Coun. Bull. Acta. 37, 1255–1275 (1982).
W. F. Giggenbach, “Geothermal solute equilibria. Derivation of Na–K–Mg–Ca geoindicators,” Geochim Cosmochim Acta. 52, 2749–2765 (1988).
Q. H. Guo and Y. X. Wang, “Geochemistry of hot springs in the Tengchong hydrothermal areas, Southwestern China,” Geothermics 215, 61–73 (2012).
D. M. Han, X. Liang, and M. G. Jin, “Evaluation of groundwater hydrochemical characteristics and mixing behavior in the Daying and Qicun geothermal systems, Xinzhou Basin,” J. Volcanol. Geotherm. Res. 189, 92–104 (2010).
Y. K. Kharaka and R. H. Mariner, “Chemical geothermometers and their application to formation waters from sedimentary basins,” Thermal History of Sedimentary Basins, Ed. by N. D. Naeser and T. H. McCulloh, (Springer–Verlag, New York, 1989), pp. 99–117.
F. Liu, W. J. Mou, and X. S. Sun, “Application of MATLAB to inverse geochemical simulation,” J. Water Resource Water Engineer. 1, 151–153 (2010).
F. A. Michel, M. D. Allen, and M. B. Grant, “Hydrogeochemistry and geothermal characteristics of the White Lake basin, South–central British Columbia, Canada,” Geothermics. 31, 169–194 (2002).
D. Nieva and R. Nieva, “Developments in geothermal energy in Mexico. Part twelve: a cationic geothermometer for prospecting of geothermal resources,” Heat Recov. Syst. 7, 243–258 (1987).
K. Pachana, P. Zuddas, and P. Censi, “Influence of pH and temperature on the early stage of mica alteration,” Appl. Geochem. 27, 1738–1744 (2012).
K. Pandarinath, “Solute geothermometry of springs and wells of the Los Azufres and Las TresVírgenes geothermal fields, Mexico,” Int. Geol. Rev. 53, 1032–1058 (2014).
D. L. Parkhurst and C. A. J. Appelo, “User’s guide to PHREEQC (version 2) – A computer program for speciation, batch–reaction, one–dimensional transport, and inverse geochemical calculations,” US Geol. Surv. Water–Resources Investigations Report 99–4259, (1999).
S. L. Shvartsev, L. V. Zamana, and A. M. Plyusnin, et al., “Equilibrium of nitrogen–rich spring waters of the Baikal Rift Zone with host rock minerals as a basis for determining mechanisms of their formation,” Geochem. Int. 53 (8), 713–725 (2015).
C. V. Tadros, C. E. Hughes, and J. Crawford, et al., “Tritium in Australian precipitation: a 50 year record,” J. Hydrol. 513, 262–273 (2014).
S. P. Verma, E. Santoyo, “New improved equations for Na/K, Na/Li and SiO2 geothermometers by outlier detection and rejection,” J. Volcanol. Geotherm. Res. 79: 9–23 (1997).
S. P. Verma and E. Santoyo, “New improved equations for Na/K, Na/Li and SiO2 geothermometers by outlier detection and rejection,” J Volcanol. Geotherm. Res. 79, 9–23 (1997).
S. P. Verma, K. Pandarinath, and E. Santoyo, “SolGeo: A new computer program for solute geothermometers and its application to Mexican geothermal fields,” Geothermics 37, 597–621 (2008).
X. Y. Zhang, and G. Z. Li, “The geologic and geochemical characteristics of the Changbai Mountain geothermal field,” JiLin Geology. 1, 25–30 (2006).
ACKNOWLEDGMENTS
This work was financially supported by the Science and Technology Key Research of Jilin Province (20100452), by China’s potential oil and gas resources project (20100331-OSR01-7), Scientific Research Initiation funds for PhD scholars (BQ2017011); Scientific Research Projects of the Higher University in Hebei (ZD2014023); Hebei province water conservancy science and technology plan projects (2017-59) and Youth Foundation of Hebei Province Department (QN2017026), and Natural science fund project in Hebei province (D2018403040).
Author information
Authors and Affiliations
Corresponding authors
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Baizhong Yan, Qiu, S., Xiao, C. et al. Characteristics of Mineral Fluids and Geothermal Reservoir in Changbai Mountain, Northeast of China. Geochem. Int. 57, 83–97 (2019). https://doi.org/10.1134/S0016702919010038
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0016702919010038