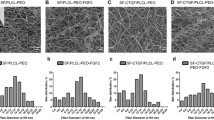
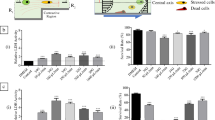
Abstract
The effect of bioresorbable materials on aging in cultured mouse NIH 3T3 fibroblasts treated with elevated glucose concentration was investigated. The cells were grown on films produced from the silkworm fibroin and rS1/9, a recombinant analog of Nephila clavipes spidroin 1. Exposure to 50 mM glucose of the cells grown on uncoated glass support resulted in the cell growth retardation. The average areas of the cells and nuclei and the percentage of apoptotic cells increased, whereas the amount of soluble collagen decreased. In contrast, on the fibroin and spidroin films, the cell density and the percentage of 5-bromo-2′-deoxyuridine (BrdU)-positive cells were higher vs. the cells grown on the glass support. The films protected NIH 3T3 fibroblasts from the glucose-induced death. The most prominent effects on the cell density, BrdU incorporation, and apoptosis prevention were observed in the cells cultured on spidroin films. Unlike the cells grown on glass support (decrease in the soluble collagen production) or fibroin (no effect), production of soluble collagen by the cells grown on spidroin films increased after cell exposure to 50 mM glucose. Molecular analysis demonstrated that 50 mM glucose upregulated phosphorylation of the NFκB heterodimer p65 subunit in the cells grown on the glass support. The treatment of cells grown on fibroin films with 5.5 mM or 50 mM glucose had no effect on p65 phosphorylation. The same treatment decreased p65 phosphorylation in the cells on the spidroin films. These results demonstrate the anti-aging efficacy of biomaterials derived from the silk proteins and suggest that spidroin is more advantageous for tissue engineering and therapy than fibroin.







Similar content being viewed by others
Abbreviations
- BrdU:
-
5-bromo-2′-deoxyuridine
- DMSO:
-
dimethyl sulfoxide
- ECM:
-
extracellular matrix
- HFIP:
-
1,1,1,3,3,3-hexafluoro-2-propanol
REFERENCES
West, M. D., Pereira-Smith, O. M., and Smith, J. R. (1989) Replicative senescence of human skin fibroblasts correlates with a loss of regulation and overexpression of collagenase activity, Exp. Cell Res., 184, 138-147.
Stadelmann, W. K., Digenis, A. G., and Tobin, G. R. (1998) Physiology and healing dynamics of chronic cutaneous wounds, Am. J. Surg., 176, 26S-38S.
Mekkes, J. R., Loots, M. A. M., Van Der Wal, A. C., and Bos, J. D. (2003) Causes, investigation and treatment of leg ulceration, Br. J. Dermatol., 148, 388-401.
Bellin, C., De Wiza, D. H., Wiernsperger, N. F., and Rösen, P. (2006) Generation of reactive oxygen species by endothelial and smooth muscle cells: Influence of hyperglycemia and metformin, Horm. Metab. Res., 38, 732-739.
Beauchef, G., Bigot, N., Kypriotou, M., Renard, E., Porée, B., Widom, R., Dompmartin-Blanchere, A., Oddos, T., Maquart, F. X., Demoor ,M., Boumediene, K., and Galera, P. (2012) The p65 subunit of NF-κB inhibits COL1A1 gene transcription in human dermal and scleroderma fibroblasts through its recruitment on promoter by protein interaction with transcriptional activators (c-Krox, Sp1, and Sp3), J. Biol. Chem., 287, 3462-3478.
Fisher, G. J., Kang, S., Varani, J., Bata-Csorgo, Z., Wan, Y., Datta, S., and Voorhees, J. J. (2002) Mechanisms of photoaging and chronological skin aging, Arch. Dermatol., 138, 1462-1470.
Fisher, G. J., Quan, T., Purohit, T., Shao, Y., Moon, K. C., He, T., Varani, J., Kang, S., and Voorhees, J. J. (2009) Collagen fragmentation promotes oxidative stress and elevates matrix metalloproteinase-1 in fibroblasts in aged human skin, Am. J. Pathol., 174, 101-114.
Cole, M. A., Quan, T., Voorhees, J. J., and Fisher, G. J. (2018) Extracellular matrix regulation of fibroblast function: redefining our perspective on skin aging, J. Cell Commun. Signal., 12, 35-43.
Dyer, D. G., Dunn, J. A., Thorpe, S. R., Bailie, K. E., Lyons, T. J., McCance, D. R., and Baynes, J. W. (1993) Accumulation of Maillard reaction products in skin collagen in diabetes and aging, J. Clin. Invest., 91, 2463-2469.
Hadley, J. C., Meek, K. M., and Malik, N. S. (1998) Glycation changes the charge distribution of type I collagen fibrils, Glycoconj. J., 15, 835-840.
Phillip, J. M., Aifuwa, I., Walston, J., and Wirtz, D. (2015) The mechanobiology of aging, Annu. Rev. Biomed. Eng., 17, 113-141.
Shanbhag, S., Nayak, A., Narayan, R., and Nayak, U. Y. (2019) Anti-aging and sunscreens: paradigm shift in cosmetics, Adv. Pharm. Bull., 9, 348-359.
Matsuyama, M., WuWong, D. J., Horvath, S., and Matsuyama, S. (2019) Epigenetic clock analysis of human fibroblasts in vitro: effects of hypoxia, donor age, and expression of hTERT and SV40 largeT, Aging (Albany, NY), 11, 3012-3022.
Yan, L.-J. (2014) Pathogenesis of chronic hyperglycemia: from reductive stress to oxidative stress, J. Diabetes Res., 2014, 1-11.
Campos, C. (2012) Chronic hyperglycemia and glucose toxicity: pathology and clinical sequelae, Postgrad. Med., 124, 90-97.
Safi, S. Z., Qvist, R., Kumar, S., Batumalaie, K., and Ismail, I. S. B. (2014) Molecular mechanisms of diabetic retinopathy, general preventive strategies, and novel therapeutic targets, Biomed Res. Int., 2014, 801269.
Sun, Q., Li, J., and Gao, F. (2014) New insights into insulin: the anti-inflammatory effect and its clinical relevance, World J. Diabetes, 5, 89-96.
Geraldes, P., and King, G. L. (2010) Activation of protein kinase C isoforms and its impact on diabetic complications, Circ. Res., 106, 1319-1331.
Groop, P.-H., Forsblom, C., and Thomas, M. C. (2005) Mechanisms of disease: pathway-selective insulin resistance and microvascular complications of diabetes, Nat. Clin. Pract. Endocrinol. Metab., 1, 100-110.
Soydas, T., Sarac, E. Y., Cinar, S., Dogan, S., Solakoglu, S., Tuncdemir, M., Sultuybek, G. K. (2018) The protective effects of metformin in an in vitro model of aging 3T3 fibroblast under the high glucose conditions, J. Physiol. Biochem., 74, 273-281.
Li, B., Bian, X., Hu, W., Wang, X., Li, Q., Wang, F., Sun, M., Ma, K., Zhang, C., Chang, J., and Fu, X. (2020) Regenerative and protective effects of calcium silicate on senescent fibroblasts induced by high glucose, Wound Repair Regen., 28, 315-325.
Maeda, M., Hayashi, T., Mizuno, N., Hattori, Y., and Kuzuya, M. (2015) Intermittent high glucose implements stress-induced senescence in human vascular endothelial cells: role of superoxide production by NADPH oxidase, PLoS One, 10, e0123169.
Christian, F., Smith, E., and Carmody, R. (2016) The regulation of NF-κB subunits by phosphorylation, Cells, 5, 12.
Wei, W., and Ji, S. (2018) Cellular senescence: molecular mechanisms and pathogenicity, J. Cell. Physiol., 233, 9121-9135.
Zhang, L., Yousefzadeh, M. J., Suh, Y., Niedernhofer, L. J., and Robbins, P. D. (2019) Signal transduction, ageing and disease, Subcell Biochem., 91, 227-247.
Zhang, W.-S., Pan, A., Zhang, X., Ying, A., Ma, G., Liu, B.-L., Qi, L.-W., Liu, Q., and Li, P. (2019) Inactivation of NF-κB2 (p52) restrains hepatic glucagon response via preserving PDE4B induction, Nat. Commun., 10, 4303.
Soria-Valles, C., López-Soto, A., Osorio, F. G., and López-Otín, C. (2017) Immune and inflammatory responses to DNA damage in cancer and aging, Mech. Ageing Dev., 165, 10-16.
Tilstra, J. S., Robinson, A. R., Wang, J., Gregg, S. Q., Clauson, C. L., Reay, D. P., Nasto, L. A., St Croix, C. M., Usas, A., Vo, N., Huard, J., Clemens, P. R., Stolz, D. B., Guttridge, D. C., Watkins, S. C., Garinis, G. A., Wang, Y., Niedernhofer, L. J., and Robbins, P. D. (2012) NF-κB inhibition delays DNA damage-induced senescence and aging in mice, J. Clin. Invest., 122, 2601-2612.
Sheikh, F. A., Ju, H. W., Lee, J. M., Moon, B. M., Park, H. J., Lee, O. J., Kim, J. H., Kim, D. K., and Park, C. H. (2015) 3D electrospun silk fibroin nanofibers for fabrication of artificial skin, Nanomedicine, 11, 681-691.
Teplenin, A., Krasheninnikova, A., Agladze, N., Sidoruk, K., Agapova, O., Agapov, I., Bogush, V., and Agladze, K. (2015) Functional analysis of the engineered cardiac tissue grown on recombinant spidroin fiber meshes, PLoS One, 10, e0121155.
Sidoruk, K. V., Davydova, L. I., Kozlov, D. G., Gubaidullin, D. G., Glazunov, A. V., Bogush, V. G., and Debabov, V. G. (2015) Fermentation optimization of a Saccharomyces cerevisiae strain producing 1F9 recombinant spidroin, Appl. Biochem. Microbiol., 51, 766-773.
Bogush, V. G., Sidoruk, K. V, Molchan, O. K., Ptitsyn, L. R., Altman, I. B., Kozlov, D. G., Efremov, B. D., Benevolenskii, S. V., Agapov, I. I., and Mashko, S. V. (2001) Molecular cloning and expression in yeast of synthetic genes encoding protein analogues of the dragline silk protein spidroin 1, Russ. J. Biotechnol., 2, 11-22.
Moisenovich, M. M., Pustovalova, O. L., Yu Arhipova, A., Vasiljeva, T. V., Sokolova, O. S., Bogush, V. G., Debabov, V. G., Sevastianov, V. I., Kirpichnikov, M. P., and Agapov, I. I. (2011) In vitro and in vivo biocompatibility studies of a recombinant analogue of spidroin 1 scaffolds, J. Biomed. Mater. Res. A, 96 A, 125-131.
Moisenovich, M. M., Pustovalova, O., Shackelford, J., Vasiljeva, T. V., Druzhinina, T. V., Kamenchuk, Y. A., Guzeev, V. V., Sokolova, O. S., Bogush, V. G., Debabov, V. G., Kirpichnikov, M. P., and Agapov, I. I. (2012) Tissue regeneration in vivo within recombinant spidroin 1 scaffolds, Biomaterials, 33, 3887-3898.
Moisenovich, M. M., Malyuchenko, N. V., Arkhipova, A. Y., Kotlyarova, M. S., Davydova, L. I., Goncharenko, A. V., Agapova, O. I., Drutskaya, M. S., Bogush, V. G., Agapov, I. I., Debabov, V. G., Kirpichnikov, M. P. (2015) Novel 3D-microcarriers from recombinant spidroin for regenerative medicine, Dokl. Biochem. Biophys., 463, 232-235.
Nosenko, M. A., Moysenovich, A. M., Zvartsev, R. V., Arkhipova, A. Y., Zhdanova, A. S., Agapov, I. I., Vasilieva, T. V., Bogush, V. G., Debabov, V. G., Nedospasov, S. A., Moisenovich, M. M., and Drutskaya, M. S. (2018) Novel biodegradable polymeric microparticles facilitate scarless wound healing by promoting re-epithelialization and inhibiting fibrosis, Front. Immunol., 9, 1-11.
Bessonov, I., Moysenovich, A., Arkhipova, A., Ezernitskaya, M., Efremov, Y., Solodilov, V., Timashev, P., Shaytan, K., Shtil, A., and Moisenovich, M. (2020) The mechanical properties, secondary structure, and osteogenic activity of photopolymerized fibroin, Polymers, 12, 646.
Bessonov, I. V., Rochev, Y. A., Arkhipova, A. Y., Kopitsyna, M. N., Bagrov, D. V., Karpushkin, E. A., Bibikova, T. N., Moysenovich, A. M., Soldatenko, A. S., Nikishin, I. I., Kotliarova, M. S., Bogush, V. G., Shaitan, K. V., and Moisenovich, M. M. (2019) Fabrication of hydrogel scaffolds via photocrosslinking of methacrylated silk fibroin, Biomed. Mater., 14, 034102.
Baker, B. M., Trappmann, B., Wang, W. Y., Sakar, M. S., Kim, I. L., Shenoy, V. B., Burdick, J. A., and Chen, C. S. (2015) Cell-mediated fibre recruitment drives extracellular matrix mechanosensing in engineered fibrillar microenvironments, Nat. Mater., 14, 1262-1268.
Lodder, E. M., Scicluna, B. P., Beekman, L., Arends, D., Moerland, P. D., Tanck, M. W. T., Adriaens, M. E., and Bezzina, C. R. (2014) Integrative genomic approach identifies multiple genes involved in cardiac collagen deposition, Circ. Cardiovasc. Genet., 7, 790-798.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding, Anal. Biochem., 72, 248-254.
Zhang, Q., Lenardo, M. J., and Baltimore, D. (2017) 30 Years of NF-κB: a blossoming of relevance to human pathobiology, Cell, 168, 37-57.
World Health Organization and International Diabetes Federation (2006) Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation, World Health Organization.
Zhang, D., Lu, H., Chen, Z., Wang, Y., Lin, J., Xu, S., Zhang, C., Wang, B., Yuan, Z., Feng, X., Jiang, X., and Pan, J. (2017) High glucose induces the aging of mesenchymal stem cells via Akt/mTOR signaling, Mol. Med. Rep., 16, 1685-1690.
Bian, X., Li, B., Yang, J., Ma, K., Sun, M., Zhang, C., and Fu, X. (2020) Regenerative and protective effects of dMSC-sEVs on high-glucose-induced senescent fibroblasts by suppressing RAGE pathway and activating Smad pathway, Stem Cell Res. Ther., 11, 166.
Khemais-Benkhiat, S., Belcastro, E., Idris-Khodja, N., Park, S. H., Amoura, L., Abbas, M., Auger, C., Kessler, L., Mayoux, E., Toti, F., and Schini-Kerth, V. B. (2020) Angiotensin II-induced redox-sensitive SGLT1 and 2 expression promotes high glucose-induced endothelial cell senescence, J. Cell. Mol. Med., 24, 2109-2122.
Stirban, A., Gawlowski, T., and Roden, M. (2013) Vascular effects of advanced glycation endproducts: clinical effects and molecular mechanisms, Mol. Metab., 3, 94-108.
Chen, J. H., Lin, X., Bu, C., and Zhang, X. (2018) Role of advanced glycation end products in mobility and considerations in possible dietary and nutritional intervention strategies, Nutr. Metab., 15, 72.
Bogush, V. G., Sokolova, O. S., Davydova, L. I., Klinov, D. V., Sidoruk, K. V., Esipova, N. G., Neretina, T. V., Orchanskyi, I. A., Makeev, V. Y., Tumanyan, V. G., Shaitan, K. V., Debabov, V. G., and Kirpichnikov, M. P. (2009) A novel model system for design of biomaterials based on recombinant analogs of spider silk proteins, J. Neuroimmune Pharmacol., 4, 17-27.
Schmidt, D. R., Waldeck, H., and Kao, W. J. (2009) Protein adsorption to biomaterials, in Biological Interactions on Materials Surfaces (Puleo, D., and Bizios, R., eds.) Springer, New York, NY.
Gao, C., Peng, S., Feng, P., and Shuai, C. (2017) Bone biomaterials and interactions with stem cells, Bone Res., 5, 17059.
Haugh, M. G., Murphy, C. M., and O’Brien, F. J. (2010) Novel freeze-drying methods to produce a range of collagen-glycosaminoglycan scaffolds with tailored mean pore sizes, Tissue Eng. Part C Methods, 16, 887-894.
Sai, K. P., and Babu, M. (2000) Collagen based dressings – a review, Burns, 26, 54-62.
Tu, Y., and Quan, T. (2016) Oxidative stress and human skin connective tissue aging, Cosmetics, 3, 1-12.
Hang, W., He, B., Chen, J., Xia, L., Wen, B., Liang, T., Wang, X., Zhang, Q., Wu, Y., Chen, Q., and Chen, J. (2018) Berberine ameliorates high glucose-induced cardiomyocyte injury via AMPK signaling activation to stimulate mitochondrial biogenesis and restore autophagic flux, Front. Pharmacol., 9, 1121.
Chen, W., Kang, J., Xia, J., Li, Y., Yang, B., Chen, B., Sun, W., Song, X., Xiang, W., Wang, X., Wang, F., Wan, Y., and Bi, Z. (2008) p53-related apoptosis resistance and tumor suppression activity in UVB-induced premature senescent human skin fibroblasts, Int. J. Mol. Med., 21, 645-653.
Chen, Q. M., Tu, V. C., Catania, J., Burton, M., Toussaint, O., and Dilley, T. (2000) Involvement of Rb family proteins, focal adhesion proteins and protein synthesis in senescent morphogenesis induced by hydrogen peroxide, J. Cell Sci., 113, 4087-4097.
Bentov, I., Damodarasamy, M., Plymate, S., and Reed, M. J. (2014) Decreased proliferative capacity of aged dermal fibroblasts in a three dimensional matrix is associated with reduced IGF1R expression and activation, Biogerontology, 15, 329-337.
Beausejour, C. M. (2003) Reversal of human cellular senescence: roles of the p53 and p16 pathways, EMBO J., 22, 4212-4222.
Mehta, I. S., Figgitt, M., Clements, C. S., Kill, I. R., and Bridger, J. M. (2007) Alterations to nuclear architecture and genome behavior in senescent cells, Ann. N. Y. Acad. Sci., 1100, 250-263.
Zhang, R., Chen, W., and Adams, P. D. (2007) Molecular dissection of formation of senescence-associated heterochromatin foci, Mol. Cell. Biol., 27, 2343-2358.
Chujo, S., Shirasaki, F., Kondo-Miyazaki, M., Ikawa, Y., and Takehara, K. (2009) Role of connective tissue growth factor and its interaction with basic fibroblast growth factor and macrophage chemoattractant protein-1 in skin fibrosis, J. Cell. Physiol., 220, 189-195.
Arkhipova, A. Y., Nosenko, M. A., Malyuchenko, N. V., Zvartsev, R. V., Moisenovich, A. M., Zhdanova, A. S., Vasil’eva, T. V., Gorshkova, E. A., Agapov, I. I., Drutskaya, M. S., Nedospasov, S. A., and Moisenovich, M. M. (2016) Effects of fibroin microcarriers on inflammation and regeneration of deep skin wounds in mice, Biochemistry (Moscow), 81, 1251-1260.
Chien, Y., Scuoppo, C., Wang, X., Fang, X., Balgley, B., Bolden, J. E., Premsrirut, P., Luo, W., Chicas, A., Lee, C. S., Kogan, S. C., and Lowe, S. W. (2011) Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity, Genes Dev., 25, 2125-2136.
Funding
This work was supported by the Russian Foundation for Basic Research [projects Nos. 17-00-00359 (17-00-00356, 17-00-00357, 17-00-00358) and 18-34-00875].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest. This article does not contain any studies involving human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Moysenovich, A., Moisenovich, M., Sudina, A. et al. Recombinant Spidroin Films Attenuate Individual Markers of Glucose Induced Aging in NIH 3T3 Fibroblasts. Biochemistry Moscow 85, 808–819 (2020). https://doi.org/10.1134/S0006297920070093
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297920070093