Abstract
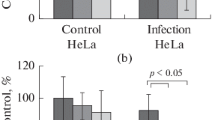
Human lung cancer cells (Calu-3 line) were studied for the development of apoptosis, necrosis, and autophagy in response to infection with orthoand paramyxoviruses. Biochemical pathways underlying various mechanisms of cell death differed for different viruses. When infected with murine Sendai paramyxovirus, Calu-3 cells demonstrated typical necrotic features such as cell swelling (but not shrinkage), lack of chromatin DNA laddering, of caspase 3 and 8 activation, and of apoptotic cleavage of poly(ADP-ribose) polymerase (PARP) protein; an activation of antiapoptotic protein kinase Akt was also revealed. In contrast, infection with avian influenza virus A/FPV/Rostock/34 (H7N1 subtype) or Newcastle disease virus (NDV, avian paramyxovirus) caused the development of typical apoptotic markers such as cell shrinkage, ladder-type chromosomal DNA fragmentation, caspase 3 and 8 activation, and proteolytic cleavage of PARP in the absence of Akt activation. Notably, no upregulation of p53 protein phosphorylation was observed in all infected cells, which indicates that p53 is not involved in the virus-induced death of Calu-3 cells. Cell death caused by the influenza virus was accompanied by overstimulation of autophagy, whereas no stimulation of autophagy was observed in the NDV-infected cells. Infection with Sendai virus caused moderate stimulation of autophagy, which suggests that the mechanism of the virus-induced cell death and the balance between autophagy and cell death in infected cancer cells depend on the virus type and might significantly differ even for closely related viruses. Therefore, an optimal strategy for oncolytic virus-mediated destruction of tumor cells in cancer patients requires selection of the most appropriate oncolytic virus based on the mechanism of its cytolytic action in a particular type of tumor.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhirnov, O., and Klenk, H. D. (2003) Human influenza A viruses are proteolytically activated and do not induce apoptosis in CACO-2 cells, Virology, 313, 198–212; Erratum in: Virology (2003) 317, 383.
Zhirnov, O. P., and Klenk, H. D. (2007) Control of apoptosis in influenza virus-infected cells by up-regulation of Akt and p53 signaling, Apoptosis, 12, 1419–1432.
Zhirnov, O. P., and Klenk, H. D. (2013) Influenza A virus proteins NS1 and hemagglutinin along with M2 are involved in stimulation of autophagy in infected cells, J. Virol., 87, 13107-13014.
Kondylis, V., Kumari, S., Vlantis, K., and Pasparakis, M. (2017) The interplay of IKK, NF-κB and RIPK1 signaling in the regulation of cell death, tissue homeostasis and inflammation, Immunol. Rev., 277, 113–127.
Pasparakis, M., and Vandenabeele, P. (2015) Necroptosis and its role in inflammation, Nature, 517, 311–320.
Davidovich, P., Kearney, C. J., and Martin, S. J. (2014) Inflammatory outcomes of apoptosis, necrosis and necroptosis, Biol. Chem., 395, 1163–1171.
Creagh, E. M. (2014) Caspase crosstalk: integration of apoptotic and innate immune signaling pathways, Trends Immunol., 35, 631–640.
Man, S. M., and Kanneganti, T. D. (2016) Converging roles of caspases in inflammasome activation, cell death and innate immunity, Nat. Rev. Immunol., 16, 7–21.
Cuadrado-Castano, S., Sanchez-Aparicio, M. T., GarciaSastre, A., and Villar, E. (2015) The therapeutic effect of death: Newcastle disease virus and its antitumor potential, Virus Res., 209, 56–66.
Saga, K., and Kaneda, Y. (2015) Oncolytic Sendai virusbased virotherapy for cancer: recent advances, Oncolyt. Virother., 4, 141–147.
Matveeva, O. V., Guo, Z. S., Shabalina, S. A., and Chumakov, P. M. (2015) Oncolysis by paramyxoviruses: multiple mechanisms contribute to therapeutic efficiency, Mol. Ther. Oncolyt., 2, 15011–15021.
Chen, D., Yu, J., and Zhang, L. (2016) Necroptosis: an alternative cell death program defending against cancer, Biochim. Biophys. Acta, 1865, 228–236.
Zhirnov, O. P. (2017) Paramyxoviruses activation by host proteases in cultures of normal and cancer cells, Vopr. Virusol., 62, 65–72.
Zhirnov, O. P., Vorob’eva, I. V., Safonova, O. A., Malyshev, N. A., Schwalm, F., and Klenk, H.-D. (2013) Pathogenic effect of pandemic influenza virus H1N1 under replication in cultures of human cells, Vopr. Virusol., 58, 20–28.
Liu, G., Pei, F., Yang, F., Li, L., Amin, A. D., Liu, S., Buchan, J. R., and Cho, W. C. (2017) Role of autophagy and apoptosis in non-small-cell lung cancer, Int. J. Mol. Sci., 18, E367.
Saitoh, M., Pullen, N., Brennan, P., Cantrell, D., Dennis, P. B., and Thomas, G. (2002) Regulation of an activated S6 kinase 1 variant reveals a novel mammalian target of rapamycin phosphorylation site, J. Biol. Chem., 277, 20104–20112.
Fumarola, C., Bonelli, M. A., Petronini, P. G., and Alfieri, R. R. (2014) Targeting PI3K/AKT/mTOR pathway in nonsmall cell lung cancer, Biochem. Pharmacol., 90, 197–207.
Shimobayashi, M., and Hall, M. N. (2014) Making new contacts: the mTOR network in metabolism and signaling crosstalk, Nat. Rev. Mol. Cell Biol., 15, 155–162.
Fogh, J., Fogh, J. M., and Orfeo, T. (1977) One hundred and twenty-seven cultured human tumor cell lines producing tumors in nude mice, J. Natl. Cancer Inst., 59, 221–226.
Shen, B. Q., Finkbeiner, W. E., Wine, J. J., Mrsny, R. J., and Widdicombe, J. H. (1994) Calu-3: a human airway epithelial cell line that shows cAMP-dependent Cl− secretion, Am. J. Physiol. Lung Cell Mol. Physiol., 266, L493–L501.
Levine, B., and Kroemer, G. (2008) Autophagy in the pathogenesis of disease, Cell, 132, 27–42.
Marino, G., Niso-Santano, M., Baehrecke, E. H., and Kroemer, G. (2014) Self-consumption: the interplay of autophagy and apoptosis, Nat. Rev. Mol. Cell Biol., 15, 81–94.
Rossman, J. S., and Lamb, R. A. (2009) Autophagy, apoptosis, and the influenza virus M2 protein, Cell Host Microbe, 6, 299–300.
Wei, B., Cui, Y., Huang, Y., Liu, H., Li, L., Li, M., Ruan, K. C., Zhou, Q., and Wang, C. (2015) Tom70 mediates Sendai virus-induced apoptosis on mitochondria, J. Virol., 89, 3804–3818.
Chattopadhyay, S., Fensterl, V., Zhang, Y., Veleeparambil, M., Yamashita, M., and Sen, G. C. (2013) Role of interferon regulatory factor 3-mediated apoptosis in the establishment and maintenance of persistent infection by Sendai virus, J. Virol., 87, 16–24.
Koks, C. A., Garg, A. D., Ehrhardt, M., Riva, M., Vandenberk, L., Boon, L., De Vleeschouwer, S., Agostinis, P., Graf, N., and Van Gool, S. W. (2015) Newcastle disease virotherapy induces long-term survival and tumor-specific immune memory in orthotopic glioma through the induction of immunogenic cell death, Int. J. Cancer, 136, E313–325.
Nomura, M., Ueno, A., Saga, K., Fukuzawa, M., and Kaneda, Y. (2014) Accumulation of cytosolic calcium induces necroptotic cell death in human neuroblastoma, Cancer Res., 74, 1056–1066.
O’Donnell, M. A., Perez-Jimenez, E., Oberst, A., Ng, A., Massoumi, R., Xavier, R., Green, D. R., and Ting, A. T. (2011) Caspase 8 inhibits programmed necrosis by processing CYLD, Nat. Cell Biol., 13, 1437–1442.
Feng, S., Yang, Y., Mei, Y., Ma, L., Zhu, D. E., Hoti, N., Castanares, M., and Wu, M. (2007) Cleavage of RIP3 inactivates its caspase-independent apoptosis pathway by removal of kinase domain, Cell Signal., 19, 2056–2067.
Man, S. M., Karki, R., and Kanneganti, T. D. (2017) Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases, Immunol. Rev., 277, 61–75.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O. P. Zhirnov, 2017, published in Biokhimiya, 2017, Vol. 82, No. 9, pp. 1345-1353.
Rights and permissions
About this article
Cite this article
Zhirnov, O.P. Biochemical variations in cytolytic activity of ortho- and paramyxoviruses in human lung tumor cell culture. Biochemistry Moscow 82, 1048–1054 (2017). https://doi.org/10.1134/S0006297917090085
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297917090085