Abstract
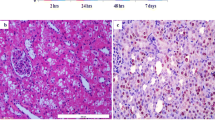
One of the most important pathological consequences of renal ischemia/reperfusion (I/R) is kidney malfunctioning. I/R leads to oxidative stress, which affects not only nephron cells but also cells of the vascular wall, especially endothelium, resulting in its damage. Assessment of endothelial damage, its role in pathological changes in organ functioning, and approaches to normalization of endothelial and renal functions are vital problems that need to be resolved. The goal of this study was to examine functional and morphological impairments occurring in the endothelium of renal vessels after I/R and to explore the possibility of alleviation of the severity of these changes using mitochondria-targeted antioxidant 10-(6′-plastoquinonyl)decylrhodamine 19 (SkQR1). Here we demonstrate that 40-min ischemia with 10-min reperfusion results in a profound change in the structure of endothelial cells mitochondria, accompanied by vasoconstriction of renal blood vessels, reduced renal blood flow, and increased number of endothelial cells circulating in the blood. Permeability of the kidney vascular wall increased 48 h after I/R. Injection of SkQR1 improves recovery of renal blood flow and reduces vascular resistance of the kidney in the first minutes of reperfusion; it also reduces the severity of renal insufficiency and normalizes permeability of renal endothelium 48 h after I/R. In in vitro experiments, SkQR1 provided protection of endothelial cells from death provoked by oxygen–glucose deprivation. On the other hand, an inhibitor of NO-synthases, L-nitroarginine, abolished the positive effects of SkQR1 on hemodynamics and protection from renal failure. Thus, dysfunction and death of endothelial cells play an important role in the development of reperfusion injury of renal tissues. Our results indicate that the major pathogenic factors in the endothelial damage are oxidative stress and mitochondrial damage within endothelial cells, while mitochondria-targeted antioxidants could be an effective tool for the protection of tissue from negative effects of ischemia.
Similar content being viewed by others
Abbreviations
- AKI:
-
acute kidney injury
- DCF:
-
2,7-dichlorodihydrofluorescein
- I/R:
-
ischemia/reperfusion
- IREC:
-
isolated renal epithelial cells
- OGD:
-
oxygen–glucose deprivation
- ROS:
-
reactive oxygen species
- SkQR1:
-
10-(6′-plastoquinonyl)decylrhodamine 19
References
Ajis, A., Bagnall, N. M., Collis, M. G., and Johns, E. J. (2003) Effect of endothelin antagonists on the renal hemodynamic and tubular responses to ischemia-reperfusion injury in anesthetized rats, Exp. Physiol., 88, 483–490.
Olof, P., Hellberg, A., Kallskog, O., and Wolgast, M. (1991) Red cell trapping and postischemic renal blood flow. Differences between the cortex, outer and inner medulla, Kidney Int., 40, 625–631.
Adams, P. L., Adams, F. F., Bell, P. D., and Navar, L. G. (1980) Impaired renal blood flow autoregulation in ischemic acute renal failure, Kidney Int., 18, 68–76.
Kelleher, S. P., Robinette, J. B., and Conger, J. D. (1984) Sympathetic nervous system in the loss of autoregulation in acute renal failure, Am. J. Physiol., 246, F379–F386.
De Greef, K. E., Ysebaert, D. K., Dauwe, S., Persy, V., Vercauteren, S. R., Mey, D., and De Broe, M. E. (2001) Anti-B7-1 blocks mononuclear cell adherence in vasa recta after ischemia, Kidney Int., 60, 1415–1427.
Takada, M., Nadeau, K. C., Shaw, G. D., Marquette, K. A., and Tilney, N. L. (1997) The cytokine-adhesion molecule cascade in ischemia/reperfusion injury of the rat kidney. Inhibition by a soluble P-selectin ligand, J. Clin. Invest., 99, 2682–2690.
Kwon, O., Phillips, C. L., and Molitoris, B. A. (2002) Ischemia induces alterations in actin filaments in renal vascular smooth muscle cells, Am. J. Physiol. Renal Physiol., 282, F1012–F1019.
Kuhne, W., Besselmann, M., Noll, T., Muhs, A., Watanabe, H., and Piper, H. M. (1993) Disintegration of cytoskeletal in energy-depleted endothelial structure cells of actin filaments in energy-depleted endothelial cells, Am. J. Physiol. Heart Circ. Physiol., 264, H1599–1608.
Sutton, T. A., Mang, H. E., Campos, S. B., Sandoval, R. M., Yoder, M. C., and Molitoris, B. A. (2003) Injury of the renal microvascular endothelium alters barrier function after ischemia, Am. J. Physiol. Renal Physiol., 285, F191–198.
Kevil, C. G., Oshima, T., Alexander, B., Coe, L. L., and Alexander, J. S. (2000) H(2)O(2)-mediated permeability: role of MAPK and occludin, Am. J. Physiol. Cell Physiol., 279, C21–C30.
Kwon, O., Hong, S.-M., Sutton, T. A., and Temm, C. J. (2008) Preservation of peritubular capillary endothelial integrity and increasing pericytes may be critical to recovery from postischemic acute kidney injury, Am. J. Physiol. Renal Physiol., 295, F351–F359.
Cantaluppi, V., Gatti, S., Medica, D., Figliolini, F., Bruno, S., Deregibus, M. C., Sordi, A., Biancone, L., Tetta, C., and Camussi, G. (2012) Microvesicles derived from endothelial progenitor cells protect the kidney from ischemia-reperfusion injury by microRNA-dependent reprogramming of resident renal cells, Kidney Int., 82, 412–427.
Basile, D. P., Friedrich, J. L., Spahic, J., Knipe, N., Mang, H., Leonard, E. C., Changizi-Ashtiyani, S., Bacallao, R. L., Molitoris, B. A., and Sutton, T. A. (2011) Impaired endothelial proliferation and mesenchymal transition contribute to vascular rarefaction following acute kidney injury, Am. J. Physiol. Renal Physiol., 300, F721–F733.
Zorov, D. B., Juhaszova, M., and Sollott, S. J. (2014) Mitochondrial reactive oxygen species (ROS) and ROSinduced ROS release, Physiol. Rev., 94, 909–950.
Silachev, D. N., Plotnikov, E. Y., Pevzner, I. B., Zorova, L. D., Babenko, V. A., Zorov, S. D., Popkov, V. A., Jankauskas, S. S., Zinchenko, V. P., Sukhikh, G. T., and Zorov, D. B. (2014) The mitochondrion as a key regulator of ischaemic tolerance and injury, Heart Lung Circ., 10, 897–904.
Ahmeda, A. F., and Johns, E. J. (2012) The regulation of blood perfusion in the renal cortex and medulla by reactive oxygen species and nitric oxide in the anesthetized rat, Acta Physiol., 204, 443–450.
Majid, D. S. A., and Nishiyama, A. (2002) Nitric oxide blockade enhances renal responses to superoxide dismutase inhibition in dogs, Hypertension, 39, 293–297.
Liu, R., Ren, Y., Garvin, J. L., and Carretero, O. A. (2004) Superoxide enhances tubuloglomerular feedback by constricting the afferent arteriole, Kidney Int., 66, 268–274.
Zhang, H., Schmeisser, A., Garlichs, C. D., Plotze, K., Damme, U., Mugge, A., and Daniel, W. G. (1999) Angiotensin II-induced superoxide anion generation in human vascular endothelial cells: role of membrane-bound NADH-/NADPH-oxidases, Cardiovasc. Res., 44, 215–222.
Dikalova, A. E., Bikineyeva, A. T., Budzyn, K., Nazarewicz, R. R., McCann, L., Lewis, W., Harrison, D. G., and Dikalov, S. I. (2010) Therapeutic targeting of mitochondrial superoxide in hypertension, Circ. Res., 107, 106–116.
Plotnikov, E. Y., Silachev, D. N., Chupyrkina, A. A., Danshina, M. I., Jankauskas, S. S., Morosanova, M. A., Stelmashook, E. V., Vasileva, A. K., Goryacheva, E. S., Pirogov, Y. A., Isaev, N. K., and Zorov, D. B. (2010) Newgeneration Skulachev ions exhibiting nephroprotective and neuroprotective properties, Biochemistry (Moscow), 75, 145–150.
Plotnikov, E. Y., Chupyrkina, A. A., Jankauskas, S. S., Pevzner, I. B., Silachev, D. N., Skulachev, V. P., and Zorov, D. B. (2011) Mechanisms of nephroprotective effect of mitochondria-targeted antioxidants under rhabdomyolysis and ischemia/reperfusion, Biochim. Biophys. Acta, 1812, 77–86.
Plotnikov, E. Y., Silachev, D. N., Jankauskas, S. S., Rokitskaya, T. I., Chupyrkina, A. A., Pevzner, I. B., Zorova, L. D., Isaev, N. K., Antonenko, Y. N., Skulachev, V. P., and Zorov, D. B. (2012) Mild uncoupling of respiration and phosphorylation as a mechanism providing nephro- and neuroprotective effects of penetrating cations of the SkQ family, Biochemistry (Moscow), 77, 1029–1037.
Jankauskas, S. S., Plotnikov, E. Y., Morosanova, M. A., Pevzner, I. B., Zorova, L. D., Skulachev, V. P., and Zorov, D. B. (2012) Mitochondria-targeted antioxidant SkQR1 ameliorates gentamycin-induced renal failure and hearing loss, Biochemistry (Moscow), 77, 666–670.
Plotnikov, E. Y., Morosanova, M. A., Pevzner, I. B., Zorova, L. D., Manskikh, V. N., Pulkova, N. V., Galkina, S. I., Skulachev, V. P., and Zorov, D. B. (2013) Protective effect of mitochondria-targeted antioxidants in an acute bacterial infection, Proc. Natl. Acad. Sci. USA, 110, E3100–E3108.
Bakeeva, L. E., Barskov, I. V., Egorov, M. V., Isaev, N. K., Kapelko, V. I., Kazachenko, A. V., Kirpatovsky, V. I., Kozlovsky, S. V., Lakomkin, V. L., Levina, S. B., Pisarenko, O. I., Plotnikov, E. Y., Saprunova, V. B., Serebryakova, L. I., Skulachev, M. V., Stelmashook, E. V., Studneva, I. M., Tskitishvili, O. V., Vasilyeva, A. K., Victorov, I. V., Zorov, D. B., and Skulachev, V. P. (2008) Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 2. Treatment of some ROS- and age-related diseases (heart arrhythmia, heart infarctions, kidney ischemia, and stroke), Biochemistry (Moscow), 73, 1288–1299.
Mosmann, T. (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays, J. Immunol. Methods, 65, 55–63.
Jankauskas, S. S., Matsievsky, D. D., Plotnikov, E. Y., and Zorov, D. B. (2014) High-frequency ultrasound Doppler system for study of renal blood flow during ischemia/reperfusion of kidney, Nephrol. Dial., 16, 169–175.
Plotnikov, E. Y., Kazachenko, A. V., Vyssokikh, M. Y., Vasileva, A. K., Tcvirkun, D. V., Isaev, N. K., Kirpatovsky, V. I., and Zorov, D. B. (2007) The role of mitochondria in oxidative and nitrosative stress during ischemia/reperfusion in the rat kidney, Kidney Int., 72, 1493–1502.
Nazarewicz, R. R., Dikalova, A. E., Bikineyeva, A., and Dikalov, S. I. (2013) Nox2 as a potential target of mitochondrial superoxide and its role in endothelial oxidative stress, Am. J. Physiol. Heart Circ. Physiol., 305, H1131–1140.
Zorov, D. B., Filburn, C. R., Klotz, L. O., Zweier, J. L., and Sollott, S. J. (2000) Reactive oxygen species (ROS)induced ROS release: a new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes, J. Exp. Med., 192, 1001–1014.
Dikalov, S. (2011) Cross talk between mitochondria and NADPH oxidases, Free Radic. Biol. Med., 51, 1289–1301.
Woywodt, A., Kirsch, T., and Haubitz, M. (2008) Circulating endothelial cells in renal disease: markers and mediators of vascular damage, Nephrol. Dial. Transplant., 23, 7–10.
Zinovkin, R. A., Romaschenko, V. P., Galkin, I. I., Zakharova, V. V., Pletjushkina, O. Y., Chernyak, B. V., and Popova, E. N. (2014) Role of mitochondrial reactive oxygen species in age-related inflammatory activation of endothelium, Aging (Albany N. Y.), 6, 661–674.
Demyanenko, I. A., Popova, E. N., Zakharova, V. V., Ilyinskaya, O. P., Vasilieva, T. V., Romashchenko, V. P., Fedorov, A. V., Manskikh, V. N., Skulachev, M. V., Zinovkin, R. A., Pletjushkina, O. Y., Skulachev, V. P., and Chernyak, B. V. (2015) Mitochondria-targeted antioxidant SkQ1 improves impaired dermal wound healing in old mice, Aging (Albany N. Y.), 7, 475–485.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM16-208, October 24, 2016.
Rights and permissions
About this article
Cite this article
Jankauskas, S.S., Andrianova, N.V., Alieva, I.B. et al. Dysfunction of kidney endothelium after ischemia/reperfusion and its prevention by mitochondria-targeted antioxidant. Biochemistry Moscow 81, 1538–1548 (2016). https://doi.org/10.1134/S0006297916120154
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297916120154