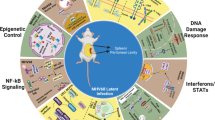
Abstract
The genomes of certain types of human and primate herpesviruses contain functional homologs of important host cytokines (IL-6, IL-17, and IL-10), or so-called virokines. Virokines can interact with immune cell receptors, transmit a signal to them, and thus switch the type of immune response that facilitates viral infection development. In this work, we have summarized possible ways of virokine origin and proposed an evolutionary scenario of virokine acquisition with involvement of retroviral coinfection of the host. This scenario is probably valid for vIL-6 of HHV-8 and MRV-5 viruses, vIL-17 of HVS virus, and vIL-10 of HHV-4, Bonobo-HV, RhLCV, and BaLCV viruses. The ability to acquire cytokine genes allows herpesviruses to implement unique strategies of avoiding the immune response and provides them an evolutionary advantage: more than 90% of the host population can be chronically infected with different herpesviruses. It is possible that the biological success of herpesviruses can be partially due to their cooperation with another group of viruses. This hypothesis emphasizes the importance of studies on the reciprocal influence of pathogens on their coinfection, as well as their impact on the host organism.
Similar content being viewed by others
Abbreviations
- BaLCV:
-
baboon lymphocryptovirus
- BonoboHV:
-
bonobo herpesvirus
- EBV:
-
Epstein–Barr virus
- HHV:
-
human herpesvirus
- HVS:
-
herpesvirus saimiri
- IFN:
-
interferon
- IL:
-
interleukin
- MRV:
-
rhesus macaque rhadinovirus
- RhCMV:
-
rhesus macaque cytomegalovirus
- RhLCV:
-
rhesus macaque lymphocryptovirus
- SaHV-2:
-
Saimiriine herpesvirus 2
- vIL:
-
virokine, interleukin homolog
References
Kunin, E. V. (2014) Logic of Case. On the Nature and Origin of Biological Evolution [in Russian], Tsentrolitograf, Moscow.
Kotwal, G. J. (1999) Virokines: mediators of virus-host interaction and future immunomodulators in medicine, Arch. Immunol. Ther. Exp. (Warsz.), 47, 135–138.
Alcami, A. (2003) Viral mimicry of cytokines, chemokines and their receptors, Nat. Rev. Immunol., 3, 36–50.
Brown, J. C. (2014) The role of DNA repair in herpesvirus pathogenesis, Genomics, 104, 287–294.
Zheng, Z.-M. (2003) Split genes and their expression in Kaposi’s sarcoma-associated herpesvirus, Rev. Med. Virol., 13, 173–184.
Farlow, A., Meduri, E., and Schlotterer, C. (2011) DNA double-strand break repair and the evolution of intron density, Trends Genet., 27, 1–6.
Rice, P., Longden, I., and Bleasby, A. (2000) EMBOSS: the European molecular biology open software suite, Trends Genet., 16, 276–277.
Katoh, K., Misawa, K., Kuma, K. I., and Miyata, T. (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform, Nucleic Acids Res., 30, 3059–3066.
Katoh, K., Kuma, K., Toh, H., and Miyata, T. (2005) MAFFT version 5: improvement in accuracy of multiple sequence alignment, Nucleic Acids Res., 33, 511–518.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., and Kumar, S. (2013) MEGA6: Molecular Evolutionary Genetics Analysis version 6.0, Mol. Biol. Evol., 30, 27252729.
Drummond, A. J., and Rambaut, A. (2007) BEAST: Bayesian evolutionary analysis by sampling trees, BMC Evol. Biol., 7, 1.
Kent, W. J. (2002) BLAT–the BLAST-like alignment tool, Genome Res., 12, 656–664.
Hedges, S. B., Dudley, J., and Kumar, S. (2006) TimeTree: a public knowledge base of divergence times among organisms, Bioinformatics, 22, 2971–2972.
Maul, D. H., Zaiss, C. P., MacKenzie, M. R., Shiigi, S. M., Marx, P. A., and Gardner, M. B. (1988) Simian retrovirus D serogroup 1 has a broad cellular tropism for lymphoid and nonlymphoid cells, J. Virol., 62, 1768–1773.
Rogers, D. L., McClure, G. B., Ruiz, J. C., Abee, C. R., and Vanchiere, J. A. (2015) Endemic viruses of squirrel monkeys (Saimiri spp.), Comp. Med., 65, 232–240.
Ouyang, P., Rakus, K., Van Beurden, S. J., Westphal, A. H., Davison, A. J., Gatherer, D., and Vanderplasschen, A. F. (2014) IL-10 encoded by viruses: a remarkable example of independent acquisition of a cellular gene by viruses and its subsequent evolution in the viral genome, J. Gen. Virol., 95, 245–262.
Bruce, A. G., Thouless, M. E., Haines, A. S., Pallen, M. J., Grundhoff, A., and Rose, T. M. (2015) Complete genome sequence of pig-tailed macaque rhadinovirus 2 and its evolutionary relationship with rhesus macaque rhadinovirus and human herpesvirus 8/Kaposi’s sarcoma-associated herpesvirus, J. Virol., 89, 3888–3909.
Sin, S. H., and Dittmer, D. P. (2012) Cytokine homologs of human gammaherpesviruses, J. Interferon Cytokine Res., 32, 53–59.
Chatterjee, M., Osborne, J., Bestetti, G., Chang, Y., and Moore, P. S. (2002) Viral IL-6-induced cell proliferation and immune evasion of interferon activity, Science, 298, 1432–1435.
Polizzotto, M. N., Uldrick, T. S., Wyvill, K. M., Aleman, K., Marshall, V., Wang, V., Whitby, D., Pittaluga, S., Jaffe, E. S., Millo, C., Tosato, G., Little, R. F., Steinberg, S. M., Sereti, I., and Yarchoan, R. (2016) Clinical features and outcomes of patients with symptomatic Kaposi sarcoma herpesvirus (KSHV)-associated inflammation: prospective characterization of KSHV inflammatory cytokine syndrome (KICS), Clin. Infect. Dis., 62, 730–738.
Damania, B., and Desrosiers, R. C. (2001) Simian homologs of human herpesvirus 8, Philos. Trans. R Soc. Lond. B: Biol. Sci., 356, 535–543.
Estep, R. D., and Wong, S. W. (2013). Rhesus macaque rhadinovirus-associated disease, Curr. Opin. Virol., 3, 245–250.
Gorshkova, E. A., Nedospasov, S. A., and Shilov, E. S. (2016) Evolutionary plasticity of the IL-6 family cytokines, Mol. Biol., 50, in press.
Boulanger, M. J., Chow, D. C., Brevnova, E. E., and Garcia, K. C. (2003) Hexameric structure and assembly of the interleukin-6/IL-6 a-receptor/gp130 complex, Science, 300, 2101–2104.
Chow, D. C., Brevnova, L., He, X. L., Martick, M. M., Bankovich, A., and Garcia, K. C. (2002) A structural template for gp130-cytokine signaling assemblies, Biochim. Biophys. Acta, 1592, 225–235.
Sakakibara, S., and Tosato, G. (2011) Viral interleukin-6: role in Kaposi’s sarcoma-associated herpesvirus-associated malignancies, J. Interferon Cytokine Res., 31, 791–801.
McGeoch, D. J., Gatherer, D., and Dolan, A. (2005) On phylogenetic relationships among major lineages of the Gammaherpesvirinae, J. Gen. Virol., 86, 307–316.
Ensser, A., Thurau, M., Wittmann, S., and Fickenscher, H. (2003) The genome of herpesvirus saimiri C488 which is capable of transforming human T-cells, Virology, 314, 471487.
Folcik, V. A., Garofalo, M., Coleman, J., Donegan, J. J., Rabbani, E., Suster, S., Nuovo, A., Magro, C. M., Di Leva, G., and Nuovo, G. J. (2014) Idiopathic pulmonary fibrosis is strongly associated with productive infection by herpesvirus saimiri, Modern Pathol., 27, 851–862.
Yao, Z., Fanslow, W. C., Seldin, M. F., Rousseau, A. M., Painter, S. L., Comeau, M. R., and Spriggs, M. K. (1995) Herpesvirus Saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor, Immunity, 3, 811–821.
Gasse, P., Riteau, N., Vacher, R., Michel, M. L., Fautrel, A., Di Padova, F., Fick, L., Charron, S., Lagente, V., Eberl, G., and Le Bert, M. (2011) IL-1 and IL-23 mediate early IL-17A production in pulmonary inflammation leading to late fibrosis, PLoS One, 6, e23185.
Ouyang, P., Rakus, K., Van Beurden, S. J., Westphal, A. H., Davison, A. J., Gatherer, D., and Vanderplasschen, A. F. (2014) IL-10 encoded by viruses: a remarkable example of independent acquisition of a cellular gene by viruses and its subsequent evolution in the viral genome, J. Gen. Virol., 95, 245–262.
Nicholas, J., Zong, J. C., Alcendor, D. J., Ciufo, D. M., Poole, L. J., Sarisky, R. T., Chiou, C. J., Zhang, X., Wan, X., Guo, H. G., Reitz, M.S., and Hayward, G. S. (1998) Novel organizational features, captured cellular genes, and strain variability within the genome of KSHV/HHV8, J. Natl. Cancer Inst. Monogr., 23, 79–88.
Brinzevich, D., Young, G. R., Sebra, R., Ayllon, J., Maio, S. M., Deikus, G., Chen, B. K., Fernandez-Sesma, A., Simon, V., and Mulder, L. C. (2014) HIV-1 interacts with HERV-K (HML-2) envelopes derived from human primary lymphocytes, J. Virol., 88, 6213–6223.
Aswad, A., and Katzourakis, A. (2015) Convergent capture of retroviral superantigens by mammalian herpesviruses, Nat. Commun., 6, 8299.
Kimura, A., and Kishimoto, T. (2010) IL-6: regulator of Treg/Th17 balance, Eur. J. Immunol., 40, 1830–1835.
Rossi, J. F., Lu, Z. Y., Jourdan, M., and Klein, B. (2015) Interleukin-6 as a therapeutic target, Clin. Cancer Res., 6, 1248–1257.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E. A. Gorshkova, E. S. Shilov, 2016, published in Biokhimiya, 2016, Vol. 81, No. 11, pp. 1604–1613.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gorshkova, E.A., Shilov, E.S. Possible mechanisms of acquisition of herpesvirus virokines. Biochemistry Moscow 81, 1350–1357 (2016). https://doi.org/10.1134/S0006297916110122
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297916110122