Abstract
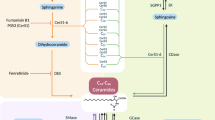
This review presents data on the functional biochemistry of ceramide, one of the key sphingolipids with properties of a secondary messenger. Molecular mechanisms of the involvement of ceramide in apoptosis in pancreatic β-cells and its role in the formation of insulin resistance in pathogenesis of type 2 diabetes are reviewed. One of the main predispositions for the development of insulin resistance and diabetes is obesity, which is associated with ectopic fat deposition and significant increase in intracellular concentrations of cytotoxic ceramides. A possible approach to the restoration of tissue sensitivity to insulin in type 2 diabetes based on selective reduction of the content of cytotoxic ceramides is discussed.
Similar content being viewed by others
Abbreviations
- Apaf-1:
-
apoptotic protease activating factor 1
- Bcl-2:
-
B-cell lymphoma 2 protein
- CAPK:
-
ceramide-activated protein kinase
- CAPP:
-
ceramide-activated protein phosphatase
- c-Jun:
-
c-Jun transcription factor
- CRD:
-
cysteine-rich domain
- DAG:
-
1,2-diacylglycerol
- ER:
-
endoplasmic reticulum
- ERK-1/2:
-
extracellular signal-regulated kinase 1/2
- FA:
-
fatty acid
- FADD:
-
Fas/Apo-1-associated with death domain
- FAN:
-
factor associated with neutral sphingomyelinase activation
- Fas-L:
-
Fas-ligand
- FOXO1:
-
forkhead box protein O1
- GLUT4:
-
glucose transporter 4
- GSH:
-
reduced glutathione
- GSK3:
-
glycogen synthase kinase 3
- IFN-γ:
-
interferon-γ
- IL-1:
-
interleukin-1
- iNOS:
-
inducible nitric oxide synthase
- IRS-1:
-
insulin receptor substrate 1
- JNK:
-
c-Jun N-terminal kinase
- KSR:
-
kinase suppressor of Ras
- MAPK:
-
mitogen-activated protein kinase
- MLK3:
-
mixed lineage kinase 3
- mRNA:
-
messenger RNA
- NF-κB:
-
nuclear factor kappa B
- NO:
-
nitric oxide
- PDK1:
-
3-phosphoinositide-dependent protein kinase 1
- PDX-1:
-
pancreatic and duodenal homeobox gene 1
- PH-domain:
-
pleckstrin homology domain
- PI3K:
-
phosphatidylinositol 3kinase
- PKB/Akt:
-
protein kinase B
- PKC:
-
protein kinase C
- PP1:
-
protein phosphatase 1
- PP2A:
-
protein phosphatase 2A
- PtdIns(4,5)P2 :
-
phosphatidylinositol (4,5)-bisphosphate
- PtdIns(3,4,5)P3 :
-
phosphatidylinositol (3,4,5)-trisphosphate
- Rb:
-
retinoblastoma protein
- RNS:
-
reactive nitrogen species
- ROS:
-
reactive oxygen species
- SAPK:
-
stress-activated protein kinase
- SM:
-
sphingomyelin
- SMase:
-
sphingomyelinase
- S1P:
-
sphingosin-1-phosphate
- TNF-α:
-
tumor necrosis factor-α
- TNFR:
-
tumor necrosis factor receptor
- TRADD:
-
TNF receptor-associated death domain protein
- UPR:
-
unfolded protein response
References
Merrill, A. H., Schmelz, E. M., Wang, E., Dillehay, D. L., Rice, L. G., Meredith, F., and Riley, R. T. (1997) Importance of sphingolipids and inhibitors of sphingolipid metabolism as components of animal diets, J. Nutr., 127, 830S-833S.
Huwiler, A., Kolter, T., Pfeilschifter, J., and Sandhoff, K. (2000) Physiology and pathophysiology of sphingolipid metabolism and signaling, Biochim. Biophys. Acta, 1485, 63–99.
Colombaionia, L., and Garcia-Gilb, M. (2004) Sphingolipid metabolites in neural signaling and function, Brain Res. Rev., 46, 328–355.
Hannun, Y. A., Loomis, C. R., Merrill, A. H., and Bell, R. M. (1986) Sphingosine inhibition of protein kinase C activity and of phorbol dibutyrate binding in vitro and in human platelets, J. Biol. Chem., 261, 12604–12609.
Hannun, Y. A. (1994) The sphingomyelin cycle and the second messenger function of ceramide, J. Biol. Chem., 269, 3125–3128.
Okazaki, T., Bell, R. M., and Hannun, Y. A. (1989) Sphingomyelin turnover induced by vitamin D3 in HL-60 cells. Role in cell differentiation, J. Biol. Chem., 264, 19076–19080.
Ohanian, J., and Ohanian, V. (2001) Sphingolipids in mammalian cell signaling, Cell. Mol. Life Sci., 58, 20532068.
Hannun, Y. A., and Bell, R. M. (1989) Regulation of protein kinase C by sphingosine and lysosphingolipids, Clin. Chim. Acta, 185, 333–345.
Marchesini, N., and Hannun, Y. A. (2004) Acid and neutral sphingomyelinases: roles and mechanisms of regulation, Biochem. Cell Biol., 82, 27–44.
Gault, C. R., Obeid, L. M., and Hannun, Y. A. (2010) An overview of sphingolipid metabolism: from synthesis to breakdown, Adv. Exp. Med. Biol., 688, 1–23.
Pettus, B. J., Chalfant, Ch. E., and Hannun, Y. A. (2002) Ceramide in apoptosis: an overview and current perspectives, Biochim. Biophys. Acta, 1585, 114–125.
Wu, B. X., Rajagopalan, V., Roddy, P. L., Clarke, C. J., and Hannun, Y. A. (2010) Identification and characterization of murine mitochondria-associated neutral sphingomyelinase (MA-nSMase), the mammalian sphingomyelin phosphodiesterase 5, J. Biol. Chem., 285, 17993–18002.
Gatt, S., Barenholz, Y., and Roitman, A. (1966) Isolation of rat brain lecithinase-A, specific for the alpha’-position of lecithin, Biochem. Biophys. Res. Commun., 24, 69–72.
Sawada, M., Nakashima, S., Kiyono, T., Yamada, J., Hara, S., Nakagawa, M., Shinoda, J., and Sakai, N. (2002) Acid sphingomyelinase activation requires caspase8 but not p53 nor reactive oxygen species during Fasinduced apoptosis in human glioma cells, Exp. Cell Res., 273, 157–168.
Rotolo, J., Zhang, J., Donepudi, M., Lee, H., Fuks, Z., and Kolesnick, R. (2005) Caspase-dependent and -independent activation of acid sphingomyelinase signaling, J. Biol. Chem., 280, 26425–26434.
Zeidan, Y. H., and Hannun, Y. A. (2007) Activation of acid sphingomyelinase by protein kinase Cdelta-mediated phosphorylation, J. Biol. Chem., 282, 11549–11561.
Hannun, Y. A., and Obeid, L. M. (2008) Principles of bioactive lipid signaling: lessons from sphingolipids, Nature, 9, 139–150.
Castillo, S. S., Levy, M., Thaikoottathil, J. V., and Goldkorn, T. (2007) Reactive nitrogen and oxygen species activate different sphingomyelinases to induce apoptosis in airway epithelial cells, Exp. Cell Res., 313, 2680–2686.
Parent, N., Scherer, M., Liebisch, G., Schmitz, G., and Bertrand, R. (2011) Protein kinase C-delta isoform mediates lysosome labilization in DNA damage-induced apoptosis, Int. J. Oncol., 38, 313–324.
Santana, P., Pena, L. A., Haimovitz-Friedman, A., Martin, S., Green, D., McLoughlin, M., Cordon-Cardo, C., Schuchman, E. H., Fuks, Z., and Kolesnick, R. (1996) Acid sphingomyelinase-deficient human lymphoblasts and mice are defective in radiation-induced apoptosis, Cell, 86, 189–199.
Paris, F., Grassme, H., Cremesti, A., Zager, J., Fong, Y., Haimovitz-Friedman, A., Fuks, Z., Gulbins, E., and Kolesnick, R. (2001) Natural ceramide reverses Fas resistance of acid sphingomyelinase(–/–) hepatocytes, J. Biol. Chem., 276, 8297–8305.
Gulbins, E., and Grassme, H. (2002) Ceramide and cell death receptor clustering, Biochim. Biophys. Acta, 1585, 139–145.
Tsukamoto, S., Hirotsu, K., Kumazoe, M., Goto, Y., Sugihara, K., Suda, T., Tsurudome, Y., Suzuki, T., Yamashita, S., Kim, Y., Huang, Y., Yamada, K., and Tachibana, H. (2012) Green tea polyphenol EGCG induces lipid-raft clustering and apoptotic cell death by activating protein kinase C delta and acid sphingomyelinase through a 67 kDa laminin receptor in multiple myeloma cells, Biochem. J., 443, 525–534.
Tirodkar, T. S., and Voelkel-Johnson, C. (2012) Sphingolipids in apoptosis, Exp. Oncol., 34, 231–242.
Schneider, P. B., and Kennedy, E. P. (1967) Sphingomyelinase in normal human spleens and in spleens from subjects with Niemann–Pick disease, J. Lipid Res., 8, 202–209.
Tani, M., and Hannun, Y. A. (2007) Analysis of membrane topology of neutral sphingomyelinase 2, FEBS Lett., 581, 1323–1328.
Wu, B. X., Clarke, C. J., and Hannun, Y. A. (2010) Mammalian neutral sphingomyelinases: regulation and roles in cell signaling responses, Neuromol. Med., 12, 320–330.
Hofmann, K., Tomiuk, S., Wolff, G., and Stoffel, W. (2000) Cloning and characterization of the mammalian brain-specific, Mg2+-dependent neutral sphingomyelinase, Proc. Natl. Acad. Sci. USA, 97, 5895–5900.
Marchesini, N., Luberto, C., and Hannun, Y. A. (2003) Biochemical properties of mammalian neutral sphingomyelinase 2 and its role in sphingolipid metabolism, J. Biol. Chem., 278, 13775–13783.
Marchesini, N., Osta, W., Bielawski, J., Luberto, C., Obeid, L. M., and Hannun, Y. A. (2004) Role for mammalian neutral sphingomyelinase 2 in confluence-induced growth arrest of MCF7 cells, J. Biol. Chem., 279, 25101–25111.
Karakashian, A. A., Giltiay, N. V., Smith, G. M., and Nikolova-Karakashian, M. N. (2004) Expreßsion of neutral sphingomyelinase-2 (NSMase-2) in primary rat hepatocytes modulates IL-ß-induced JNK activation, FASEB J., 18, 968–970.
Clarke, C. J., Truong, T. G., and Hannun, Y. A. (2007) Role for neutral sphingomyelinase-2 in tumor necrosis factor a-stimulated expression of vascular cell adhesion molecule-1 (VCAM) and intercellular adhesion molecule-1 (ICAM) in lung epithelial cells: p38 MAPK is an upstream regulator of nSMase2, J. Biol. Chem., 282, 1384–1396.
Adam, D., Wiegmann, K., Adam-Klages, S., Ruff, A., and Kronke, M. (1996) A novel cytoplasmic domain of the p55 tumor necrosis factor receptor initiates the neutral sphingomyelinase pathway, J. Biol. Chem., 271, 14617–14622
Adam-Klages, S., Schwandner, R., Adam, D., Kreder, D., Bernardo, K., and Kronke, M. (1998) Distinct adapter proteins mediate acid versus neutral sphingomyelinase activation through the p55 receptor for tumor necrosis factor, J. Leukoc. Biol., 63, 678–682.
Linardic, L. A., and Hannun, Y. A. (1994) Identification of a distinct pool of sphingomyelin involved in the sphingomyelin cycle, J. Biol. Chem., 269, 23530–23537.
Tepper, A. D., Ruurs, P., Wiedmer, T., Sims, P. J., Borst. J., and Van Blitterswijk, W. J. (2000) Sphingomyelin hydrolysis to ceramide during the execution phase of apoptosis results from phospholipid scrambling and alters cell-surface morphology, J. Cell. Biol., 150, 155–164.
Koval, M., and Pagano, R. E. (1991) Intracellular transport and metabolism of sphingomyelin, Biochim. Biophys. Acta, 1082, 113–125.
Grassme, H., Schwarz, H., and Gulbins, E. (2001) Molecular mechanisms of ceramide-mediated CD95 clustering, Biochem. Biophys. Res. Commun., 284, 1016–1030.
Van Blitterswijk, W., Van der Luit, A., Veldman, R. J., Verheij, M., and Borst, J. (2003) Ceramide: second messenger or modulator of membrane structure and dynamics? Biochem. J., 369, 199–211.
Vance, J. E. (1990) Phospholipid synthesis in a membrane fraction associated with mitochondria, J. Biol. Chem., 265, 7248–7256.
Garcia-Ruiz, C., Colell, A., Mari, M., Morales, A., Calvo, M., Enrich, C., and Fernandez-Checa, J. C. (2003) Defective TNF-mediated hepatocellular apoptosis and liver damage in acidic sphingomyelinase knockout mice, J. Clin. Invest., 111, 197–208.
Bionda, C., Portoukalian, J., Schmitt, D., RodriguezLafrasse, C., and Ardail, D. (2004) Subcellular compartmentalization of ceramide metabolism: MAM (mitochondria-associated membrane) and/or mitochondria? Biochem. J., 382, 527–533.
Ruiz-Arguello, M. B., Basanez, G., Goni, F. M., and Alonso, A. (1996) Different effects of enzyme-generated ceramides and diacylglycerols in phospholipid membrane fusion and leakage, J. Biol. Chem., 271, 26616–26621.
Hannun, Y. A., and Luberto, C. (2000) Ceramide in the eukaryotic stress response, Trends Cell Biol., 10, 73–80.
Ruvolo, P. P. (2003) Intracellular signal transduction pathways activated by ceramide and its metabolites, Pharmacol. Res., 47, 383–392.
Kronke, M. (1999) Biophysics of ceramide signaling: interaction with proteins and phase transition of membranes, Chem. Phys. Lipids, 101, 109–121.
Kolesnick, R., and Fuks, Z. (1995) Ceramide: a signal for apoptosis or mitogenesis? J. Exp. Med., 181, 1949–1952.
Verheij, M., Bose, R., Lin, X. H., Yao, B., Jarvis, W. D., Grant, S., Birrer, M. J., Szabo, E., Zon, L. I., Kyriakis, J. M., Haimovitz-Friedman, A., Fuks, Z., and Kolesnick, R. N. (1996) Requirement for ceramide-initiated SAPK/JNK signaling in stress-induced apoptosis, Nature, 380, 75–79.
Michael, J. M., Lavin, M. F., and Watters, D. J. (1997) Resistance to radiation-induced apoptosis in Burkitt’s lymphoma cells is associated with defective ceramide signaling, Cancer Res., 57, 3600–3605.
Whitman, S., Civoli, F., and Daniel, L. (1997) Protein kinase C beta II activation by 1-beta-D-arabinofuranosylcytosine is antagonistic to stimulation of apoptosis and Bcl2alpha down-regulation, J. Biol. Chem., 272, 23481–23484.
Suzuki, A., Iwasaki, M., Kato, M., and Wagai, N. (1997) Sequential operation of ceramide synthesis and ICE cascade in CPT-11-initiated apoptotic death signaling, Exp. Cell Res., 233, 41–47.
Uchida, Y., Nardo, A. D., Collins, V., Elias, P. M., and Holleran, W. M. (2003) De novo ceramide synthesis participates in the ultraviolet B irradiation-induced apoptosis in undifferentiated cultured human keratinocytes, J. Invest. Dermatol., 120, 662–669.
Kroesen, B. J., Jacobs, S., Pettus, B. J., Sietsma, H., Kok, J. W., Hannun, Y. A., and De Leij, L. F. (2003) BcR-induced apoptosis involves differential regulation of C16and C24ceramide formation and sphingolipid-dependent activation of the proteasome, J. Biol. Chem., 278, 14723–14731.
Mathias, S., Dressler, K. A., and Kolesnick, R. N. (1991) Characterization of a ceramide-activated protein kinase: stimulation by tumor necrosis factor alpha, Proc. Natl. Acad. Sci. USA, 88, 10009–10013.
Zhang, Y., Yao, B., Delikat, S. S., Bayoumy, S., Lin, X. H., Basu, S., McGinley, M., Chan-Hui, P. Y., Lichenstein, H., and Kolesnick, R. (1997) Kinase suppressor of Ras is ceramide-activated protein kinase, Cell, 89, 63–72.
Liu, J., Mathias, S., Yang, Z., and Kolesnick, R. N. (1994) Renaturation and tumor necrosis factor-alpha stimulation of a 97-kDa ceramide-activated protein kinase, J. Biol. Chem., 269, 3047–3052.
Dhillon, A. S., and Kolch, W. (2002) Untying the regulation of the Raf-1 kinase, Arch. Biochem. Biophys., 404, 3–9.
Xing, H. R., and Kolesnick, R. (2001) Kinase suppressor of Ras signals through Thr-269 of c-Raf-1, J. Biol. Chem., 276, 9733–9741.
Basu, S., and Kolesnick, R. (1998) Stress signals for apoptosis: ceramide and c-jun kinase, Oncogene, 17, 3277–3285.
Mathias, Sh., Penna, L. A., and Kolesnick, R. N. (1998) Signal transduction of stress via ceramide, Biochem. J., 335, 465–480.
Kishikawa, K., Chalfant, C. E., Perry, D. K., Bielawska, A., and Hannun, Y. A. (1999) Phosphatidic acid is a potent and selective inhibitor of protein phosphatase 1 and an inhibitor of ceramide-mediated responses, J. Biol. Chem., 274, 21335–21341.
Dobrowsky, R. T., and Hannun, Y. A. (1993) Ceramide-activated protein phosphatase: partial purification and relationship to protein phosphatase 2A, Adv. Lipid Res., 25, 91–104.
Mumby, M. C., and Walter, G. (1993) Protein serine/threonine phosphatases: structure regulation and function in cell growth, Physiol. Rev., 73, 673–699.
Ruvolo, P. P., Deng, X., Ito, T., Carr, B. K., and May, W. S. (1999) Ceramide induces Bcl2 dephosphorylation via a mechanism involving mitochondrial PP2A, J. Biol. Chem., 274, 20296–20300.
Ruvolo, P. P., Clark W., Mumby, M., Gao, F., and May, W. S. (2002) A functional role for the B56 alpha-subunit of protein phosphatase 2A in ceramide-mediated regulation of Bcl2 phosphorylation status and function, J. Biol. Chem., 277, 22847–22852.
Reyes, J. G., Robayna, I. G., Delgado, P. S., Gonzalez, I. H., Aguiar, J. Q., Rosas, F. E., Fanjul, L. F., and Galarreta, C. M. (1996) C-jun is a downstream target for ceramideactivated protein phosphatase in a431 cells, J. Biol. Chem., 271, 21375–21380.
Nikolova-Karakashian, M., Russell, R., Booth, R., Jenden, D., and Merrill, A. J. (1997) Sphingomyelin metabolism in rat liver after chronic dietary replacement of choline by N-aminodeanol, J. Lipid Res., 38, 1764–1770.
Chiang, C.-W., Harris, G., Ellig, C., Masters, S. C., Subramanian, R., Shenolikar, S., Wadzinski, B. E., and Yang, E. (2001) Protein phosphatase 2A activates the proapoptotic function of BAD in interleukin-3-dependent lymphoid cells by a mechanism requiring 14-3-3 dissociation, Blood, 97, 1289–1297.
Schubert, K. M., Scheid, M. P., and Duronio, V. (2000) Ceramide inhibits protein kinase B/Akt by promoting dephosphorylation of serine 473, J. Biol. Chem., 275, 13330–13335.
Salinas, M., Lopez-Valdaliso, R., Martin, D., Alvarez, A., and Cuadrado, A. (2000) Inhibition of PKB/Akt1 by C2ceramide involves activation of ceramide-activated protein phosphatase in PC12 cells, Mol. Cell. Neurosci., 15, 156–169.
Basu, S., Bayoumy, S., Zhang, Y., Lozano, J., and Kolesnick, R. (1998) BAD enables ceramide to signal apoptosis via Ras and Raf-1, J. Biol. Chem., 273, 3041930426.
Cardone, M. H., Roy, N., Stennicke, H. R., Salvesen, G. S., Franke, T. F., Stanbridge, E., Frisch, S., and Reed, J. C. (1998) Regulation of cell death protease caspase-9 by phosphorylation, Science, 282, 1318–1321.
Dbaibo, G. S., Pushkareva, M. Y., Jayadev, S., Schwarz, J. K., Horowitz, J. M., Obeid, L. M., and Hannun, Y. A. (1995) Retinoblastoma gene product as a downstream target for a ceramide-dependent pathway of growth arrest, Proc. Natl. Acad. Sci. USA, 92, 1347–1351.
Hurley, J. H., Newton, A. C., Parker, P. J., Blumberg, P. M., and Nishizuka, Y. (1997) Taxonomy and function of C1 protein kinase C homology domains, Protein Sci., 6, 477–480.
Bourbon, N. A., Yun, J., and Kester, M. (2000) Ceramide directly activates protein kinase C to regulate a stress-activated protein kinase signaling complex, J. Biol. Chem., 275, 35617–35623.
Doornbos, R. P., Theelen, M., Van der Hoeven, P. C., Van Blitterswijk, W. J., Verkleij, A. J., and Van Bergen en Henegouwen, P. M. (1999) Protein kinase C zeta is a negative regulator of protein kinase B activity, J. Biol. Chem., 274, 8589–8596.
Brenner, B., Koppenhoefer, U., Weinstock, C., Linderkamp, O., Lang, F., and Gulbins, E. (1997) Fasor ceramide-induced apoptosis is mediated by a Rac1-regulated activation of Jun N-terminal kinase/p38 kinases and GADD153, J. Biol. Chem., 272, 22173–22181.
Haldar, S., Basu, A., and Croce, C. M. (1998) Serine-70 is one of the critical sites for drug-induced Bcl 2 phosphorylation in cancer cells, Cancer Res., 58, 1609–1615.
Yamamoto, K., Ihijo, H., and Korsmeyer, S. (1999) Bcl2 is phosphorylated and inactivated by an ASK1/Jun N-terminal protein kinase pathway normally activated at G2/M, Mol. Cell. Biol., 19, 8469–8478.
Mechta-Grigoriou, F., Gerald, D., and Yaniv, M. (2001) The mammalian Jun proteins: redundancy and specificity, Oncogene, 20, 2378–2389.
Foghsgaard, L., Lademann, U., Wissing, D., Poulsen, B., and Jaattela, M. (2002) Cathepsin B mediates tumor necrosis factor-induced arachidonic acid release in tumor cells, J. Biol. Chem., 277, 39499–39506.
Wu, G. S., Saftig, P., Peters C., and El-Deiry, W. S. (1998) Potential role for cathepsin D in p53-dependent tumor suppression and chemosensitivity, Oncogene, 16, 2177–2183.
Heinrich, M., Neumeyer, J., Jakob, M., Hallas, C., Tchikov, V., Winoto-Morbach, S., Wickel, M., SchneiderBrachert, W., Trauzold, A., Hethke, A., and Schutze, S. (2004) Cathepsin D links TNF-induced acid sphingomyelinase to Bid-mediated caspase-9 and -3 activation, Cell Death Differ., 11, 550–563.
Heinrich, M., Wickel, M., Winoto-Morbach, S., Schneider-Brachert, W., Weber, T., Brunner, J., Saftig, P., Peters, C., Kronke, M., and Schutze, S. (2000) Ceramide as an activator lipid of cathepsin D, Adv. Exp. Med. Biol., 477, 305–331.
Dumitru, C. A., Sandalcioglu, I. E., Wagner, M., Weller, M., and Gulbins, E. (2009) Lysosomal ceramide mediates gemcitabine-induced death of glioma cells, J. Mol. Med. (Berlin), 87, 1123–1132.
Liu, B., and Hannun, Y. A. (1997) Inhibition of the neutral magnesium-dependent sphingomyelinase by glutathione, J. Biol. Chem., 272, 16281–16287.
Gulbins, E., and Li, P. L. (2006) Physiological and pathophysiological aspects of ceramide, Am. J. Physiol. Regul. Integr. Comp. Physiol., 290, R11–R26.
Taha, T. A., Mullen, T. D., and Obeid, L. M. (2006) A house divided: ceramide, sphingosine, and sphingosine-1phosphate in programmed cell death, Biochim. Biophys. Acta, 1758, 2027–2036.
Zheng, W., Kollmeyer, J., Symolon, H., Momin, A., Munter, E., Wang, E., Kelly, S., Allegood, J. C., Liu, Y., Peng, Q., Ramaraju, H., Sullards, M. C., Cabot, M., and Merrill, A. H., Jr. (2006) Ceramides and other bioactive sphingolipid backbones in health and disease: lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy, Biochim. Biophys. Acta, 1758, 1864–1884.
Kerr, J. F. R., Wyllie, A. H., and Currie, A. R. (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics, Br. J. Cancer, 26, 239–257.
Kiechle, F. L., and Zhang, X. (2002) Apoptosis: biochemical aspects and clinical implications, Clin. Chim. Acta, 326, 27–45.
Riedl, S. J., and Shi, Y. (2004) Molecular mechanisms of caspase regulation during apoptosis, Nat. Rev. Mol. Cell Biol., 5, 897–907.
Szegezdi, E., Fitzgerald, U., and Samali, A. (2003) Caspase-12 and ER-stress-mediated apoptosis: the story so far, Ann. N. Y. Acad. Sci., 1010, 186–194.
Wiegmann, K., Schutze, S., Machleidt, T., Witte, D., and Kronke, M. (1994) Functional dichotomy of neutral and acidic sphingomyelinases in tumor necrosis factor signaling, Cell, 78, 1005–1015.
Dbaibo, G. S., Perry, D. K., Gamard, C. J., Platt, R., Poirier, G. G., Obeid, L. M., and Hannun, Y. A. (1997) Cytokine response modifier A (CrmA) inhibits ceramide formation in response to tumor necrosis factor (TNF)alpha: CrmA and Bcl-2 target distinct components in the apoptotic pathway, J. Exp. Med., 185, 481–490.
Schwandner, R., Wiegmann, K., Bernardo, K., Kreder, D., and Kronke, M. (1998) TNF receptor death domainassociated proteins TRADD and FADD signal activation of acid sphingomyelinase, J. Biol. Chem., 273, 5916–5922.
De Maria, R., Rippo, M., Schuchman, E., and Testi, R. (1998) Acidic sphingomyelinase (ASM) is necessary for fas-induced GD3 ganglioside accumulation and efficient apoptosis of lymphoid cells, J. Exp. Med., 187, 897–902.
Salvesen, G., and Dixit, V. (1997) Caspases: intracellular signaling by proteolysis, Cell, 91, 443–446.
Cifone, M. G., Roncaioli, P., De Maria, R., Camarda, G., Santoni, A., Ruberti, G., and Testi, R. (1995) Multiple pathways originate at the Fas/APO-1 (CD95) receptor: sequential involvement of phosphatidylcholine-specific phospholipase C and acidic sphingomyelinase in the propagation of the apoptotic signal, EMBO J., 14, 5859–5868.
Chinnaiyan, A. M., Tepper, C. G., Seldin, M. F., O’Rourke, K., Kischkel, F. C., Hellbardt, S., Krammer, P. H., Peter, M. E., and Dixit, V. M. (1996) FADD/MORT1 is a common mediator of CD95 (Fas/APO-1) and tumor necrosis factor receptor-induced apoptosis, J. Biol. Chem., 271, 4961–4965.
Keane, R. W., Srinivasan, A., Foster, L. M., Testa, M. P., Ord, T., Nonner, D., Wang, H. G., Reed, J. C., Bredesen, D. E., and Kayalar, C. (1997) Activation of CPP32 during apoptosis of neurons and astrocytes, J. Neurosci. Res., 48, 168–180.
Summers, S. A., and Nelson, D. H. (2005) A role for sphingolipids in producing the common features of type 2 diabetes, metabolic syndrome X, and Cushing’s syndrome, Diabetes, 54, 591–602.
Turinsky, J., O’ Sullivan, D. M., and Bayly, B. P. (1990) 1,2-Diacylglycerol and ceramide levels in insulin-resistant tissues of the rat in vivo, J. Biol. Chem., 265, 16880–16885.
Gorska, M., Dobrzyn, A., Zendzian-Piotrowska, M., and Gorski, J. (2004) Effect of streptozotocin-diabetes on the functioning of the sphingomyelin-signaling pathway in skeletal muscles of the rat, Horm. Metab. Res., 36, 14–21.
Adams, J. M., Pratipanawatr, T., Berria, R., Wang, E., DeFronzo, R. A., Sullards, M. C., and Mandarino, L. J. (2004) Ceramide content is increased in skeletal muscle from obese insulin-resistant humans, Diabetes, 53, 25–31.
Straczkowski, M., Kowalska, I., Nikolajuk, A., DzienisStraczkowska, S., Kinalska, I., Baranowski, M., ZendzianPiotrowska, M., Brzezinska, Z., and Gorski, J. (2004) Relationship between insulin sensitivity and sphingomyelin signaling pathway in human skeletal muscle, Diabetes, 53, 1215–1221.
Holland, W. L., and Summers, S. A. (2008) Sphingolipids, insulin resistance, and metabolic disease: new insights from in vivo manipulation of sphingolipid metabolism, Endocrin. Rev., 29, 381–402.
Hajduch, E., Balendran, A., Batty, I. H., Litherland, G. J., Blair, A. S., Downes, C. P., and Hundal, H. S. (2001) Ceramide impairs the insulin-dependent membrane recruitment of protein kinase B leading to a loss in downstream signaling in L6 skeletal muscle cells, Diabetologia, 44, 173–183.
Kralik, S. F., Liu, P., Leffler, B. J., and Elmendorf, J. S. (2002) Ceramide and glucosamine antagonism of alternate signaling pathways regulating insulinand osmotic shockinduced glucose transporter 4 translocation, Endocrinology, 143, 37–46.
Powell, D. J., Turban, S., Gray, A., Hajduch, E., and Hundal, H. S. (2004) Intracellular ceramide synthesis and protein kinase Czeta activation play an essential role in palmitate-induced insulin resistance in rat L6 skeletal muscle cells, Biochem. J., 382, 619–629.
Watson, M. L., Coghlan, M., and Hundal, H. S. (2009) Modulating serine palmitoyl transferase (SPT) expression and activity unveils a crucial role in lipid-induced insulin resistance in rat skeletal muscle cells, Biochem. J., 417, 791–801.
Hu, W., Ross, J., Geng, T., Brice, S. E., and Cowart, L. A. (2011) Differential regulation of dihydroceramide desaturase by palmitate versus monounsaturated fatty acids: implications for insulin resistance, J. Biol. Chem., 286, 16596–16605.
Kanety, H., Hemi, R., Papa, M. Z., and Karasik, A. (1996) Sphingomyelinase and ceramide suppress insulin-induced tyrosine phosphorylation of the insulin receptor substrate1, J. Biol. Chem., 271, 9895–9897.
Paz, K., Hemi, R., LeRoith, D., Karasik, A., Elhanany, E., Kanety, H., and Zick, Y. (1997) A molecular basis for insulin resistance. Elevated serine/threonine phosphorylation of IRS-1 and IRS-2 inhibits their binding to the juxtamembrane region of the insulin receptor and impairs their ability to undergo insulin-induced tyrosine phosphorylation, J. Biol. Chem., 272, 29911–29918.
Sathyanarayana, P., Barthwal, M. K., Kundu, C. N., Lane, M. E., Bergmann, A., Tzivion, G., and Rana, A. (2002) Activation of the Drosophila MLK by ceramide reveals TNFalpha and ceramide as agonists of mammalian MLK3, Mol. Cell, 10, 1527–1533.
Kim, K. Y., Kim, B. C., Xu, Z., and Kim, S. J. (2004) Mixed lineage kinase 3 (MLK3)-activated p38 MAP kinase mediates transforming growth factor-beta-induced apoptosis in hepatoma cells, J. Biol. Chem., 279, 2947829484.
Aguirre, V., Uchida, T., Yenush, L., Davis, R., and White, M. F. (2000) The c-Jun NH(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of Ser(307), J. Biol. Chem., 275, 9047–9054.
Hirosumi, J., Tuncman, G., Chang, L., Gorgun, C. Z., Uysal, K. T., Maeda, K., Karin, M., and Hotamisligil, G. S. (2002) A central role for JNK in obesity and insulin resistance, Nature, 420, 333–336.
Teruel, T., Hernandez, R., and Lorenzo, M. (2001) Ceramide mediates insulin resistance by tumor necrosis factor-alpha in brown adipocytes by maintaining Akt in an inactive dephosphorylated state, Diabetes, 50, 2563–2571.
Zinda, M. J., Vlahos, C. J., and Lai, M. T. (2001) Ceramide induces the dephosphorylation and inhibition of constitutively activated Akt in PTEN negative U87MG cells, Biochem. Biophys. Res. Commun., 280, 1107–1115.
Stratford, S., DeWald, D. B., and Summers, S. A. (2001) Ceramide dissociates 3’-phosphoinositide production from pleckstrin homology domain translocation, Biochem. J., 354, 359–368.
Bourbon, N. A., Sandirasegarane, L., and Kester, M. (2002) Ceramide-induced inhibition of Akt is mediated through protein kinase C zeta: implications for growth arrest, J. Biol. Chem., 277, 3286–3292.
Powell, D. J., Hajduch, E., Kular, G., and Hundal, H. S. (2003) Ceramide disables 3-phosphoinositide binding to the pleckstrin homology domain of protein kinase B (PKB)/Akt by a PKCzeta-dependent mechanism, Mol. Cell. Biol., 23, 7794–7808.
Hajduch, E., Turban, S., Le Liepvre, X., Le Lay, S., Lipina, C., Dimopoulos, N., Dugail, I., and Hundal, H. S. (2008) Targeting of PKCzeta and PKB to caveolinenriched microdomains represents a crucial step underpinning the disruption in PKB-directed signaling by ceramide, Biochem. J., 410, 369–379.
Lipina, C., and Hundal, H. S. (2011) Sphingolipids: agents provocateurs in the pathogenesis of insulin resistance, Diabetologia, 54, 1596–1607.
Van Epps-Fung, M., Williford, J., Wells, A., and Hardy, R. W. (1997) Fatty acid-induced insulin resistance in adipocytes, Endocrinology, 138, 4338–4345.
Mathis, D., Vence, L., and Benoist, C. (2001) Beta-cell death during progression to diabetes, Nature, 414, 792–798.
Chandra, J., Zhivotovsky, B., Zaitsev, V., Juntti-Berggren, L., Berggren, P. O., and Orrenius, S. (2001) Role of apoptosis in pancreatic beta-cell death in diabetes, Diabetes, 50, S44–S47.
Kim, K. A., and Lee, M. S. (2009) Recent progress in research on beta-cell apoptosis by cytokines, Front. Biosci., 14, 657–664.
Ishizuka, N., Yagui, K., Tokuyama, Y., Yamada, K., Suzuki, Y., Miyazaki, J., Hashimoto, N., Makino, H., Saito, Y., and Kanatsuka, A. (1999) Tumor necrosis factor alpha signaling pathway and apoptosis in pancreatic beta cells, Metabolism, 48, 1485–1492.
Welsh, N. (1996) Interleukin-1 beta-induced ceramide and diacylglycerol generation may lead to activation of the c-Jun NH2-terminal kinase and the transcription factor ATF2 in the insulin-producing cell line RINm5F, J. Biol. Chem., 271, 8307–8312.
Lupi, R., Dotta, F., Marselli, L., Del Guerra, S., Masini, M., Santangelo, C., Patane, G., Boggi, U., Piro, S., Anello, M., Bergamini, E., Mosca, F., Di Mario, U., Del Prato, S., and Marchetti, P. (2002) Prolonged exposure to free fatty acids has cytostatic and proapoptotic effects on human pancreatic islets: evidence that beta cell death is caspase mediated, partially dependent on ceramide pathway, and Bcl-2 regulated, Diabetes, 51, 1437–1442.
Shimabukuro, M., Higa, M., Zhou, Y. T., Wang, M. Y., Newgard, C. B., and Unger, R. H. (1998) Lipoapoptosis in beta cells of obese prediabetic fa/fa rats. Role of serine palmitoyltransferase overexpression, J. Biol. Chem., 273, 32487–32490.
Maedler, K., Spinas, G. A., Dyntar, D., Moritz, W., Kaiser, N., and Donath, M. Y. (2001) Distinct effects of saturated and monounsaturated fatty acids on beta-cell turnover and function, Diabetes, 50, 69–76.
Lupi, R., Del Guerra, S., Fierabracci, V., Marselli, L., Novelli, M., Patane, G., Boggi, U., Mosca, F., Piro, S., Del Prato, S., and Marchetti, P. (2002) Lipotoxicity in human pancreatic islets and the protective effect of metformin, Diabetes, 51, S134–S137.
Veret, J., Coant, N., Berdyshev, E. V., Skobeleva, A., Therville, N., Bailbe, D., Gorshkova, I., Natarajan, V., Portha, B., and Le Stunff, H. (2011) Ceramide synthase 4 and de novo production of ceramides with specific N-acyl chain lengths are involved in glucolipotoxicity-induced apoptosis of INS-1 ß-cells, Biochem. J., 438, 177–189.
Thomas, H. E., McKenzie, M. D., Angstetra, E., Campbell, P. D., and Kay, T. W. (2009) Beta cell apoptosis in diabetes, Apoptosis, 14, 1389–1404.
Liadis, N., Salmena, L., Kwan, E., Tajmir, P., Schroer, S. A., Radziszewska, A., Li, X., Sheu, L., Eweida, M., Xu, S., Gaisano, H. Y., Hakem, R., and Woo, M. (2007) Distinct in vivo roles of caspase-8 in beta-cells in physiological and diabetes models, Diabetes, 56, 2302–2311.
Tait, S. W., and Green, D. R. (2010) Mitochondria and cell death: outer membrane permeabilization and beyond, Nat. Rev. Mol. Cell Biol., 11, 621–632.
Birbes, H., El Bawab, S., and Hannun, Y. A. (2001) Mitochondria and ceramide: intertwined roles in regulation of apoptosis, FASEB J., 15, 669–2679.
Taylor, R. C., Cullen, S. P., and Martin, S. J. (2008) Apoptosis: controlled demolition at the cellular level, Nat. Rev. Mol. Cell Biol., 9, 231–241.
Birbes, H., Luberto, C., Hsu, Y. T., El Bawab, S., Hannun, Y. A., and Obeid, L. M. (2005) A mitochondrial pool of sphingomyelin is involved in TNFalpha-induced Bax translocation to mitochondria, Biochem. J., 386, 445–451.
Birbes, H., El Bawab, S., Obeid, L. M., and Hannun, Y. A. (2002) Mitochondria and ceramide: intertwined roles in regulation of apoptosis, Adv. Enzyme Regul., 42, 113–129.
Maestre, I., Jordan, J., Calvo, S., Reig, J. A., Cena, V., Soria, B., Prentki, M., and Roche, E. (2003) Mitochondrial dysfunction is involved in apoptosis induced by serum withdrawal and fatty acids in the betacell line INS-1, Endocrinology, 144, 335–345.
Siskind, L. J., Kolesnick, R. N., and Colombini, M. (2006) Ceramide forms channels in mitochondrial outer membranes at physiologically relevant concentrations, Mitochondrion, 6, 118–125.
Navarro, P., Valverde, A. M., Rohn, J. L., Benito, M., and Lorenzo, M. (2000) Akt mediates insulin rescue from apoptosis in brown adipocytes: effect of ceramide, Growth Horm. IGF Res., 10, 256–266.
Garcia-Ruiz, C., Colell, A., Mari, M., Morales, A., and Fernandez-Checa, J. C. (1997) Direct effect of ceramide on the mitochondrial electron transport chain leads to generation of reactive oxygen species. Role of mitochondrial glutathione, J. Biol. Chem., 272, 11369–11377.
Voehringer, D. W., McConkey, D. J., McDonnell, T. J., Brisbay, S., and Meyn, R. E. (1998) Bcl-2 expression causes redistribution of glutathione to the nucleus, Proc. Natl. Acad. Sci. USA, 95, 2956–2960.
Zhang, A. Y., Teggatz, E. G., Zou, A. P., Campbell, W. B., and Li, P. L. (2005) Endostatin uncouples NOand Ca2+ response to bradykinin through enhanced O2 ·–production in the intact coronary endothelium, Am. J. Physiol. Heart Circ. Physiol., 288, H686–H694.
Hatanaka, Y., Fujii, J., Fukutomi, T., Watanabe, T., Che, W., Sanada, Y., Igarashi, Y., and Taniguchi, N. (1998) Reactive oxygen species enhances the induction of inducible nitric oxide synthase by sphingomyelinase in RAW264.7 cells, Biochim. Biophys. Acta, 1393, 203–210.
Pilane, C. M., and LaBelle, E. F. (2004) NO induced apoptosis of vascular smooth muscle cells accompanied by ceramide increase, J. Cell. Physiol., 199, 310–315.
Franzen, R., Fabbro, D., Aschrafi, A., Pfeilschifter, J., and Huwiler, A. (2002) Nitric oxide induces degradation of the neutral ceramidase in rat renal mesangial cells and is counterregulated by protein kinase C, J.Biol. Chem., 277, 46184–46190.
Di Paola, M., Cocco, T., and Lorusso, M. (2000) Ceramide interaction with the respiratory chain of heart mitochondria, Biochemistry, 39, 6660–6668.
Gudz, T. I., Tserng, K. Y., and Hoppel, C. L. (1997) Direct inhibition of mitochondrial respiratory chain complex III by cell-permeable ceramide, J. Biol. Chem., 272, 2415424158.
Morgan, D., Rebelato, E., Abdulkader, F., Graciano, M. F., Oliveira-Emilio, H. R., Hirata, A. E., Rocha, M. S., Bordin, S., Curi, R., and Carpinelli, A. R. (2009) Association of NAD(P)H oxidase with glucose-induced insulin secretion by pancreatic beta-cells, Endocrinology, 150, 2197–2201.
Won, J. S., Im, Y. B., Khan, M., Singh, A. K., and Singh, I. (2004) The role of neutral sphingomyelinase produced ceramide in lipopolysaccharide-mediated expression of inducible nitric oxide synthase, J. Neurochem., 88, 583–593.
Shimabukuro, M., Zhou, Y. T., Levi, M., and Unger, R. H. (1998) Fatty acid-induced beta cell apoptosis: a link between obesity and diabetes, Proc. Natl. Acad. Sci. USA, 95, 2498–2502.
Cnop, M., Foufelle, F., and Velloso, L. A. (2012) Endoplasmic reticulum stress, obesity and diabetes, Trends Mol. Med., 18, 59–68.
Socha, L., Silva, D., Lesage, S., Goodnow, C., and Petrovsky, N. (2003) The role of endoplasmic reticulum stress in nonimmune diabetes: NOD.k iHEL, a novel model of beta cell death, Ann. N. Y. Acad. Sci., 1005, 178183.
Laybutt, D. R., Preston, A. M., Akerfeldt, M. C., Kench, J. G., Busch, A. K., Biankin, A. V., and Biden, T. J. (2007) Endoplasmic reticulum stress contributes to beta cell apoptosis in type 2 diabetes, Diabetologia, 50, 752–763.
Lai, E., Bikopoulos, G., Wheeler, M. B., Rozakis-Adcock, M., and Volchuk, A. (2008) Differential activation of ER stress and apoptosis in response to chronically elevated free fatty acids in pancreatic beta-cells, Am. J. Physiol. Endocrinol. Metab., 294, e540-E550.
Riemer, J., Bulleid, N., and Herrmann, J. M. (2009) Disulfide formation in the ER and mitochondria: two solutions to a common process, Science, 324, 1284–1287.
Briaud, I., Harmon, J. S., Kelpe, C. L., Segu, V. B., and Poitout, V. (2001) Lipotoxicity of the pancreatic beta cell is associated with glucose-dependent esterification of fatty acids into neutral lipids, Diabetes, 50, 315–321.
Kelpe, C. L., Moore, P. C., Parazzoli, S. D., Wicksteed, B., Rhodes, C. J., and Poitout, V. (2003) Palmitate inhibition of insulin gene expression is mediated at the transcriptional level via ceramide synthesis, J. Biol. Chem., 278, 30015–30021.
Guo, J., Qian, Y., Xi, X., Hu, X., Zhu, J., and Han, X. (2010) Blockage of ceramide metabolism exacerbates palmitate inhibition of pro-insulin gene expression in pancreatic beta cells, Mol. Cell. Biochem., 338, 283–290.
Henderson, E., and Stein, R. (1994) c-Jun inhibits transcriptional activation by the insulin enhancer, and the insulin control element is the target of control, Mol. Cell. Biol., 14, 655–662.
Kaneto, H., Xu, G., Fujii, N., Kim, S., Bonner-Weir, S., and Weir, G. C. (2002) Involvement of c-Jun N-terminal kinase in oxidative stress-mediated suppression of insulin gene expression, J. Biol. Chem., 277, 30010–30018.
Bourbon, N. A., Yun, J., and Kester, M. (2002) Ceramide directly activates protein kinase C zeta to regulate a stressactivated protein kinase signaling complex, J. Biol. Chem., 275, 35617–35623.
Furukawa, N., Shirotani, T., Araki, E., Kaneko, K., Todaka, M., Matsumoto, K., Tsuruzoe, K., Motoshima, H., Yoshizato, K., Kishikawa, H., and Shichiri, M. (1999) Possible involvement of atypical protein kinase C (PKC) in glucose-sensitive expression of the human insulin gene: DNA binding activity and transcriptional activity of pancreatic and duodenal homeobox gene-1 (PDX-1) are enhanced via calphostin C-sensitive but phorbol 12-myristate 13-acetate (PMA) and Go 6976-insensitive pathway, Endocrin. J., 46, 43–58.
Sano, H., Kane, S., Sano, E., Miinea, C. P., Asara, J. M., Lane, W. S., Garner, Ch. W., and Lienhard, G. E. (2003) Insulin-stimulated phosphorylation of a Rab GTPaseactivating protein regulates GLUT4 translocation, J. Biol. Chem., 278, 14599–14602.
Veret, J., Coant, N., Berdyshev, E. V., Skobeleva, A., Therville, N., Bailbe, D., Gorshkova, I., Natarajan, V., Portha, B., and Le Stunff, H. (2011) Ceramide synthase 4 and de novo production of ceramides with specific N-acyl chain lengths are involved in glucolipotoxicity-induced apoptosis of INS-1 ß-cells, Biochem. J., 438, 177–189.
Cinar, R., Godlewski, G., Liu, J., Tam, J., Jourdan, T., Mukhopadhyay, B., Harvey-White, J., and Kunos, G. (2014) Hepatic cannabinoid-1 receptors mediate dietinduced insulin resistance by increasing de novo synthesis of long-chain ceramides, Hepatology, 59, 143–153.
Park, J. W., Park, W. J., Kuperman, Y., Boura-Halfon, S., Pewzner-Jung, Y., and Futerman, A. H. (2013) Ablation of very long acyl chain sphingolipids causes hepatic insulin resistance in mice due to altered detergent-resistant membranes, Hepatology, 57, 525–532.
Bajpeyi, S., Myrland, C. K., Covingto, J. D., Obanda, D., Cefalu, W. T., Smith, S. R., Rustan, A. C., and Ravussin, E. (2014) Lipid in skeletal muscle myotubes is associated to the donors’ insulin sensitivity and physical activity phenotypes, Obesity (Silver Spring), 22, 426–434.
Frangioudakis, G., Diakanastasis, B., Liao, B. Q., Saville, J. T., Hoffman, N. J., Mitchell, T. W., and Schmitz-Peiffer, C. (2013) Ceramide accumulation in L6 skeletal muscle cells due to increased activity of ceramide synthase isoforms has opposing effects on insulin action to those caused by palmitate treatment, Diabetologia, 56, 26972701.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D. I. Kuzmenko, T. K. Klimentyeva, 2016, published in Biokhimiya, 2016, Vol. 81, No. 9, pp. 1155-1171.
Rights and permissions
About this article
Cite this article
Kuzmenko, D.I., Klimentyeva, T.K. Role of ceramide in apoptosis and development of insulin resistance. Biochemistry Moscow 81, 913–927 (2016). https://doi.org/10.1134/S0006297916090017
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297916090017