Abstract
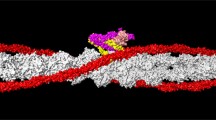
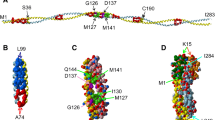
This review concerns the structure and functions of tropomyosin (TM), an actin-binding protein that plays a key role in the regulation of muscle contraction. The TM molecule is a dimer of α-helices, which form a coiled-coil. Recent views on the TM structure are analyzed, and special attention is concentrated on those structural traits of the TM molecule that distinguish it from the other coiled-coil proteins. Modern data are presented on TM functional properties, such as its interaction with actin and ability to move on the surface of actin filaments, which underlies the regulation of the actin-myosin interaction upon contraction of skeletal and cardiac muscles. Also, part of the review is devoted to analysis of the effects of mutations in TM genes associated with muscle diseases (myopathies) on the structure and functions of TM.
Similar content being viewed by others
Abbreviations
- CD:
-
circular dichroism
- DSC:
-
differential scanning calorimetry
- S1:
-
myosin subfragment 1
- TM:
-
tropomyosin
References
Bailey, K. (1948) Biochem. J., 43, 271–279.
Astbury, W. T., Reed, R., and Spark, L. C. (1948) Biochem. J., 43, 282–287.
Crick, F. H. C. (1953) Acta Cryst., 6, 689–697.
Sodek, J., Hodges, R. S., Smillie, L. B., and Jurasek, L. (1972) Proc. Natl. Acad. Sci. USA, 69, 3800–3804.
Stone, D., and Smillie, L. B. (1978) J. Biol. Chem., 253, 1137–1148.
Lehrer, S. S. (1975) Proc. Natl. Acad. Sci. USA, 72, 3377–3381.
Parry, D. A. (1975) J. Mol. Biol., 98, 519–535.
McLachlan, A. D., Stewart, M., and Smillie, L. B. (1975) J. Mol. Biol., 98, 281–291.
O’shea, E. K., Klemm, J. D., Kim, P. S., and Alber, T. (1991) Science, 254, 539–544.
Burkhard, P., Stetefeld, J., and Strelkov, S. V. (2001) Trends Cell Sci., 11, 82–88.
O’shea, E. K., Ruthkowski, R., and Kim, P. (1992) Cell, 68, 699–708.
Arndt, K. M., Pelletier, J. N., Miller, K. M., Plickthun, A., and Alber, T. (2002) Structure, 10, 1235–1248.
Lau, S. Y., Taneja, A. K., and Hodges, R. S. (1984) J. Biol. Chem., 259, 13253–13261.
Wagschal, K., Tripet, B., Lavigne, P., Mant, C., and Hodges, R. S. (1999) Protein Sci., 8, 2312–2329.
Tripet, B., Wagschal, K., Lavigne, P., Mant, C., and Hodges, R. S. (2000) J. Mol. Biol., 300, 377–402.
Tang, Y., and Tirrell, D. A. (2001) J. Am. Chem. Soc., 123, 11089–11090.
Zhou, N. E., Kay, C. M., and Hodges, R. S. (1994) J. Mol. Biol., 237, 500–512.
Mason, J. M., and Arndt, K. M. (2004) Chem. BioChem., 5, 170–176.
Brown, J. H., Cohen, C., and Parry, D. (1996) Proteins, 26, 134–145.
Herrmann, H., Haner, M., Brettel, M., Ku, N.-O., and Aebi, U. (1999) J. Mol. Biol., 286, 1403–1420.
Bullough, P. A., Hughson, F. M., Skehel, J. J., and Wiley, D. C. (1994) Nature, 371, 37–43.
Pace, C. N., and Scholtz, J. M. (1998) Biophys. J., 75, 422–427.
Kwok, S. C., and Hodges, R. S. (2003) J. Biol. Chem., 278, 35248–35254.
Kwok, S. C., and Hodges, R. S. (2004) J. Biol. Chem., 279, 21576–21588.
Perry, S. V. (2001) J. Muscle Res. Cell Motil., 22, 5–49.
Liu, H. P., and Bretscher, A. (1989) Proc. Natl. Acad. Sci. USA, 86, 90–93.
Jung-Ching Lin, J., Eppinga, R. D., Warren, K. S., and McCrae, K. R. (2008) Adv. Exp. Med. Biol., 644, 201–222.
Gunning, P. W., Schevzov, G., Kee, A. J., and Hardeman, E. C. (2005) Trends Cell Biol., 15, 333–341.
Gunning, P., Gordon, M., Wade, R., Gahlmann, R., Lin, C. S., and Hardeman, E. (1990) Dev. Biol., 138, 443–453.
Kee, A. J., and Hardeman, E. C. (2008) Adv. Exp. Med. Biol., 644, 143–157.
Nilsson, J., and Taijsharghi, H. (2008) Eur. J. Neurol., 15, 573–578.
Sanders, C., Burtnick, L. D., and Smillie, L. B. (1986) J. Biol. Chem., 261, 12774–12778.
Strand, J., Nili, M., Homsher, E., and Tobacman, L. S. (2001) J. Biol. Chem., 276, 34832–34839.
Li, X. (E.), Lehman, W., Fischer, S., and Holmes, K. C. (2010) J. Struct. Biol., 170, 307–312.
Lehrer, S. S. (1978) J. Mol. Biol., 118, 209–226.
Yukioka, S., Noda, I., Nagasawa, M., Holtzer, M. E., and Holtzer, A. (1985) Macromolecules, 18, 1083–1086.
Betteridge, D. R., and Lehrer, S. S. (1982) J. Mol. Biol., 167, 481–496.
Greenfield, N., and Hitchcock-DeGregori, S. (1993) Protein Sci., 2, 1263–1273.
Betcher-Lange, S. L., and Lehrer, S. S. (1978) J. Biol. Chem., 253, 3757–3760.
Graceffa, P., and Lehrer, S. S. (1980) J. Biol. Chem., 255, 11296–11300.
Ishii, Y., and Lehrer, S. S. (1991) J. Biol. Chem., 266, 6894–6903.
Ooi, T. (1967) Biochemistry, 6, 2433–2439.
Gorecka, A., and Drabikowski, W. (1977) FEBS Lett., 75, 145–148.
Pato, M. D., Mak, A. S., and Smillie, L. B. (1981) J. Biol. Chem., 256, 593–601.
Ueno, H. (1984) Biochemistry, 23, 4791–4798.
Sumida, J. P., Wu, E., and Lehrer, S. S. (2008) J. Biol. Chem., 283, 6728–6734.
Nevzorov, I. A., Nikolaeva, O. P., Kainov, Y. A., Redwood, C. S., and Levitsky, D. I. (2011) J. Biol. Chem., 286, 15766–15772.
Brown, J. H., Kim, K. H., Jun, G., Greenfield, N. J., Dominguez, R., Volkmann, N., Hitchcock-DeGregori, S. E., and Cohen, C. (2001) Proc. Natl. Acad. Sci. USA, 98, 8496–8501.
Brown, J. H., Zhou, Z., Reshetnikova, L., Robinson, H., Yammani, R. D., Tobacman, L. S., and Cohen, C. (2005) Proc. Natl. Acad. Sci. USA, 102, 18878–18883.
Williams, D. L., Jr., and Swenson, C. A. (1981) Biochemistry, 20, 3856–3864.
Ishii, Y., Hitchcock-DeGregori, S., Mabuchi, K., and Lehrer, S. S. (1992) Protein Sci., 1, 1319–1325.
Potekhin, S. A., and Privalov, P. L. (1982) J. Mol. Biol., 159, 519–535.
Sturtevant, J. M., Holtzer, M. E., and Holtzer, A. (1991) Biopolymers, 31, 489–495.
O’Brien, R., Sturtevant, J. M., Wrabl, J., Holtzer, M. E., and Holtzer, A. (1996) Biophys. J., 70, 2403–2407.
Kremneva, E., Boussouf, S., Nikolaeva, O., Maytum, R., Geeves, M. A., and Levitsky, D. I. (2004) Biophys. J., 87, 3922–3933.
Levitsky, D. I. (2004) Uspekhi Biol. Khim., 44, 133–170.
Levitsky, D. I. (2004) in The Nature of Biological Systems as Revealed by Thermal Methods (Lorinczy, D., ed.) Kluwer Academic Publishers, Dordrecht-Boston-London, pp. 127–158.
Kremneva, E., Nikolaeva, O., Maytum, R., Arutyunyan, A. M., Kleimenov, S. Yu., Geeves, M. A., and Levitsky, D. I. (2006) FEBS J., 273, 588–600.
Levitsky, D. I., Rostkova, E. V., Orlov, V. N., Nikolaeva, O. P., Moiseeva, L. N., Teplova, M. V., and Gusev, N. B. (2000) Eur. J. Biochem., 267, 1869–1877.
Kremneva, E. V., Nikolaeva, O. P., Gusev, N. B., and Levitsky, D. I. (2003) Biochemistry (Moscow), 68, 802–809.
Mirza, M., Robinson, P., Kremneva, E., Copeland, O., Nikolaeva, O., Watkins, H., Levitsky, D., Redwood, C., El-Mezgueldi, M., and Marston, S. (2007) J. Biol. Chem., 282, 13487–13497.
Nevzorov, I. A., Redwood, C. S., and Levitsky, D. I. (2008) Biophysics (Moscow), 53, 479–481.
Nevzorov, I. A., Redwood, C. S., and Levitsky, D. I. (2008) J. Muscle Res. Cell Motil., 29, 173–176.
Whitby, F. G., and Phillips, G. N., Jr. (2000) Proteins, 38, 49–59.
Nitanai, Y., Minakata, S., Maeda, K., Oda, N., and Maeda, Y. (2007) Adv. Exp. Med. Biol., 592, 137–151.
Li, Y., Mui, S., Brown, J. H., Strand, J., Reshetnikova, L., Tobacman, L. S., and Cohen, C. (2002) Proc. Natl. Acad. Sci. USA, 99, 7378–7383.
Minakata, S., Maeda, K., Oda, N., Wakabayashi, K., Nitanai, Y., and Maeda, Y. (2008) Biophys. J., 95, 710–719.
Greenfield, N. J., Palm, T., and Hitchcock-DeGregori, S. E. (2002) Biophys. J., 83, 2754–2766.
Brown, J. H. (2010) Protein Sci., 19, 1366–1375.
Eaton, B. L., Kominz, D. R., and Eizenberg, E. (1975) Biochemistry, 14, 2718–2725.
Yang, Y.-Z., Gordon, D., Korn, E. D., and Eizenberg, E. (1977) J. Biol. Chem., 252, 3374–3378.
Yang, Y.-Z., Korn, E. D., and Eizenberg, E. (1979) J. Biol. Chem., 254, 7137–7140.
Mak, A. S., and Smillie, L. B. (1981) Biochem. Biophys. Res. Commun., 101, 208–214.
Dabrowska, R., Nowak, E., and Drabikowski, W. (1983) J. Muscle Res. Cell Motil., 4, 143–161.
Goonasekara, C. L., Gallivan, L. J., Jackman, D. M., and Heeley, D. H. (2007) J. Muscle Res. Cell Motil., 28, 175–182.
Hitchcock-DeGregori, S. E., and Heald, R. W. (1987) J. Biol. Chem., 262, 9730–9735.
Monteiro, P. B., Lataro, R. C., Ferro, J. A., and Reinach, F. C. (1994) J. Biol. Chem., 269, 10461–10466.
Pittenger, M. F., Kistler, A., and Helfman, D. M. (1995) J. Cell. Sci., 108, 3253–3265.
Urbancikova, M., and Hitchcock-DeGregori, S. E. (1994) J. Biol. Chem., 269, 24310–24315.
Hilarioa, E., Latarob, R. L., Alegriaa, M. C., Lavardaa, S. C. S., Ferrob, J. A., and Bertolini, M. C. (2001) Biochem. Biophys. Res. Commun., 284, 955–960.
Johnson, M., Coulton, A. T., Geeves, M. A., and Mulvihill, D. P. (2010) PLoS One, 5, e15801.
Greenfield, N. J., Huang, Y. J., Swapna, G. V., Bhattacharya, A., Rapp, B., Singh, A., Montelione, G. T., and Hitchcock-DeGregori, S. E. (2006) J. Mol. Biol., 36, 480–496.
Frye, J., Klenchin, V. A., and Rayment, I. (2010) Biochemistry, 49, 4908–4920.
Tobacman, L. S. (2008) Adv. Exp. Med. Biol., 644, 85–94.
Eaton, B. L. (1976) Science, 192, 1337–1339.
Cassell, M., and Tobacman, L. S. (1996) J. Biol. Chem., 271, 12867–12872.
McLachlan, A. D., and Stewart, M. (1976) J. Mol. Biol., 103, 271–298.
Phillips, G. N., Jr. (1986) J. Mol. Biol., 192, 128–131.
Hitchcock-DeGregori, S. E., and An, Y. (1996) J. Biol. Chem., 271, 3600–3603.
Hitchcock-DeGregori, S. E., and Varnell, T. A. (1990) J. Mol. Biol., 214, 885–896.
Hammel, R. L., and Hitchcock-DeGregori, S. E. (1997) J. Biol. Chem., 272, 22409–22416.
Hitchcock-DeGregori, S. E., Song Y., and Greenfield, N. J. (2002) Biochemistry, 41, 15036–15044.
Hitchcock-DeGregori, S. E., and Singh, A. (2006) Structure, 14, 43–50.
Hitchcock-DeGregori, S. E., and Singh, A. (2010) J. Struct. Biol., 170, 319–324.
Singh, A., and Hitchcock-DeGregori, S. E. (2003) Biochemistry, 42, 14114–14121.
Holmes, K. C., and Lehman, W. (2008) J. Muscle Res. Cell Motil., 29, 213–219.
Wegner, A. (1980) FEBS Lett., 119, 245–248.
Lorenz, M., Poole, K. J. V., Popp, D., Rosenbaum, G., and Holmes, K. C. (1995) J. Mol. Biol., 246, 108–119.
Poole, K. J., Lorenz, M., Evans, G., Rosenbaum, G., Pirani, A., Tobacman, L. S., Lehman, W., and Holmes, K. C. (2006) J. Struct. Biol., 155, 273–284.
Li, X., Holmes, K., Lehman, W., Jung, H., and Fischer, S. (2010) J. Mol. Biol., 395, 327–339.
Li, X., Lehman, W., and Fischer, S. (2011) J. Struct. Biol., 170, 313–318.
Li, X., Tobacman, L. S., Mun, J., Craig, R., Fischer, S., and Lehman, W. (2011) Biophys. J., 100, 1005–1013.
Fujime, S., and Ishiwata, S. (1971) J. Mol. Biol., 62, 251–265.
Hitchcock, S. E., Carisson, L., and Lindberg, U. (1976) Cell, 7, 531–542.
Ishikawa, R., Yamashiro, S., and Matsumura, F. (1989) J. Biol. Chem., 264, 7490–7497.
Bamburg, J. R. (1999) Annu. Rev. Cell Dev. Biol., 15, 185–230.
Blanchoin, L., Pollard, T. D., and Hitchcock-DeGregori, S. E. (2001) Curr. Biol., 11, 1300–1304.
Gupton, S. L., Anderson, K. L., Kole, T. P., Fischer, R. S., Ponti, A., Hitchcock-DeGregori, S. E., Danuser, G., Fowler, V. M., Wirtz, D., Hanein, D., and Waterman-Storer, C. M. (2005) J. Cell. Biol., 168, 619–631.
Huxley, H. E. (1957) J. Biophys. Biochem. Cytol., 3, 631–648.
Margossian, S. S., and Lowey, S. (1973) J. Mol. Biol., 74, 313–330.
Engelhardt, W. A., and Liubimova, M. N. (1939) Nature, 144, 688.
Bagshaw, C. R., and Trentham, D. R. (1973) Biochem. J., 133, 323–328.
White, H. D., and Taylor, E. W. (1976) Biochemistry, 15, 5818–5826.
Lymn, R. W., and Taylor, E. W. (1971) Biochemistry, 10, 4617–4624.
Koubassova, N. A., and Tsaturyan, A. K. (2011) Biochemistry (Moscow), 76, 1484–1506.
Huxley, H. E., Simmons, R. M., Faruqi, A. R., Kress, M., Bordas, J., and Koch, M. H. J. (1981) Proc. Natl. Acad. Sci. USA, 78, 2297–2301.
Irving, M., Lombardi, V., Piazzesi, G., and Ferenczi, M. A. (1992) Nature, 357, 156–158.
Holmes, K. C., and Geeves, M. A. (2000) Phil. Trans. R. Soc. Lond. B, 355, 419–431.
Squire, J. M., and Morris, E. P. (1998) FASEB J., 12, 761–771.
Grabarek, Z., Grabarek, J., Leavis, P. C., and Gergely, J. (1983) J. Biol. Chem., 258, 14098–14102.
Greene, L. E., and Eisenberg, E. (1980) Proc. Natl. Acad. Sci. USA, 77, 2616–2620.
Lehrer, S. S., and Morris, E. P. (1982) J. Biol. Chem., 257, 8073–8080.
McKillop, D. F. A., and Geeves, M. A. (1993) Biophys. J., 65, 693–701.
Maytum, R., Lehrer, S. S., and Geeves, M. A. (1999) Biochemistry, 38, 1102–1110.
Lehrer, S. S., Golitsina, N. L., and Geeves, M. A. (1997) Biochemistry, 36, 13449–13455.
Boussouf, S. E., and Geeves, M. A. (2007) Adv. Exp. Med. Biol., 592, 99–109.
Rayment, I., Rypniewski, W. R., Schmidt-Base, K., Smith, R., Tomchick, D. R., Benning, M. M., Winkelmann, D. A., Wesenberg, G., and Holden, H. M. (1993) Science, 261, 50–58.
Holmes, K. C., Popp, D., Gebhard, W., and Kabsch, W. (1990) Nature, 347, 44–49.
Lehman, W., and Craig, R. (2008) Adv. Exp. Med. Biol., 644, 95–109.
Donner, K., Ollikainen, M., Ridanpaa, M., Chricten, H.-J., Goebel, H., Visser, M., de Pelin, K., and Wallgren-Petersson, C. (2002) Neuromuscular Disorders, 12, 151–158.
Robinson, P., Lipscomb, S., Preston, L., Altin, E., Watkins, H., Ashley, C., and Redwood, C. (2007) FASEB J., 21, 896–905.
Seidman, J. G., and Seidman, C. (2001) Cell, 104, 557–567.
Fatkin, D., and Graham, R. M. (2002) Physiol. Rev., 82, 945–980.
Wieczorek, D. F., Jagatheesan, G., and Rajan, S. (2008) Adv. Exp. Med. Biol., 644, 132–142.
Mirza, M., Marston, S., Willott, R., Ashley, C., Mogensen, J., McKenna, W., Robinson, P., Redwood, C., and Watkins, H. (2005) J. Biol. Chem., 280, 28498–28506.
Hitchcock-DeGregori, S. E., and Singh, A. (2006) Structure, 14, 43–50.
Golitsina, N., Yougmi, A., Greenfield, N., Thierfelder, L., Iizuka, K., Seidman, J., Seidman, C., Lehrer, S., and Hitchkock-DeGregori, S. E. (1997) Biochemistry, 36, 4637–4642.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I. A. Nevzorov, D. I. Levitsky, 2011, published in Uspekhi Biologicheskoi Khimii, 2011, Vol. 51, pp. 283–334.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Nevzorov, I.A., Levitsky, D.I. Tropomyosin: Double helix from the protein world. Biochemistry Moscow 76, 1507–1527 (2011). https://doi.org/10.1134/S0006297911130098
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297911130098