Abstract
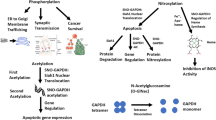
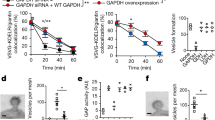
Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is a well-studied glycolytic protein with energy production as its implied occupation. It has established itself lately as a multifunctional protein. Recent studies have found GAPDH to be involved in a variety of nuclear and cytosolic pathways ranging from its role in apoptosis and regulation of gene expression to its involvement in regulation of Ca2+ influx from endoplasmic reticulum. Numerous studies also indicate that GAPDH interacts with microtubules and participates in cell membrane fusion. This review is focused on the cytosolic functions of the protein related to vesicular transport. Suggestions for future directions as well as the model of protein polymer structure and possible post-translational modifications as a basis for its multifunctional activities in the early secretory pathway are given.
Similar content being viewed by others
Abbreviations
- GAPDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- VTC:
-
vesicular tubular cluster
References
Jeffery, C. J. (1999) Trends Biochem. Sci., 24, 8–11.
Eisenstein, R. S. (2000) Annu. Rev. Nutr., 20, 627–662.
Cairo, G., Recalcati, S., Pietrangelo, A., and Minotti, G. (2002) Free Rad. Biol. Med., 32, 1237–1243.
Harris, J. I., and Waters, M. (1976) in The Enzymes (Boyer, P. D., ed.) Academic Press, New York, pp. 12968–12976.
Sirover, M. A. (1999) Biochim. Biophys. Acta, 1432, 159–184.
Singh, R., and Green, M. R. (1993) Science, 259, 365–368.
Carmona, P., Rodriguez-Casado, A., and Molina, M. (1999) Biochim. Biophys. Acta, 1432, 222–233.
Engel, M., Seifert, M., Theisinger, B., Seyfert, U., and Welter, C. (1998) J. Biol. Chem., 273, 20058–20065.
Meyer-Siegler, K., Mauro, D. J., Seal, G., Wurzer, J., deRiel, J. K., and Sirover, M. A. (1991) Proc. Natl. Acad. Sci. USA, 88, 8460–8464.
Mansur, N. R., Meyer-Siegler, K., Wurzer, J. C., and Sirover, M. A. (1993) Nucleic Acids Res., 21, 993–998.
Zheng, L., Roeder, R. G., and Luo, Y. (2003) Cell, 114, 255–266.
Nakagawa, T., Hirano, Y., Inomata, A., Yokota, S., Miyachi, K., Kaneda, M., Umeda, M., Furukawa, K., Omata, S., and Horigome, T. (2003) J. Biol. Chem., 278, 20395–20404.
Kumagai, H., and Sakai, H. (1983) J. Biochem. (Tokyo), 93, 1259–1269.
Walsh, J. L., Keith, T. J., and Knull, H. R. (1989) Biochim. Biophys. Acta, 999, 64–70.
Volker, K. W., and Knull, H. R. (1993) J. Mol. Recognit., 6, 167–177.
Volker, K. W., Reinitz, C. A., and Knull, H. R. (1995) Comp. Biochem. Physiol. Biochem. Mol. Biol., 112, 503–514.
Tisdale, E. J. (2002) J. Biol. Chem., 277, 3334–3341.
Hara, M. R., and Snyder, S. H. (2006) Cell Mol. Neurobiol., 26, 527–538.
Jahn, R., Lang, T., and Sudhof, T. C. (2003) Cell, 112, 519–533.
De, D. C., Wattiaux, R., and Bauhuin, P. (1962) Adv. Enzyme Regul., 24, 291–358.
Wooster, M. S., and Wrigglesworth, J. M. (1976) Biochem. J., 153, 93–100.
Wooster, M. S., and Wrigglesworth, J. M. (1976) Biochem. J., 159, 627–631.
Morero, R. D., Vinals, A. L., Bloj, B., and Farias, R. N. (1985) Biochemistry, 24, 1904–1909.
Lopez Vinals, A. E., Farias, R. N., and Morero, R. D. (1987) Biochem. Biophys. Res. Commun., 143, 403–409.
Hessler, R. J., Blackwood, R. A., Brock, T. G., Francis, J. W., Harsh, D. M., and Smolen, J. E. (1998) J. Leukoc. Biol., 63, 331–336.
Dowhan, W., and Bogdanov, M. (2002) in Biochemistry of Lipids, Lipoproteins, and Membranes (Vance, D. E., and Vance, J. E., eds.) Elsevier.
Glaser, P. E., and Gross, R. W. (1995) Biochemistry, 34, 12193–12203.
Glaser, P. E., and Gross, R. W. (1994) Biochemistry, 33, 5805–5812.
Glaser, P. E., Han, X., and Gross, R. W. (2002) Proc. Natl. Acad. Sci. USA, 99, 14104–14109.
Volker, K. W., and Knull, H. R. (1997) Arch. Biochem. Biophys., 338, 237–243.
Tisdale, E. J. (2001) J. Biol. Chem., 276, 2480–2486.
Tisdale, E. J., Kelly, C., and Artalejo, C. R. (2004) J. Biol. Chem., 279, 54046–54052.
Springer, S., Spang, A., and Schekman, R. (1999) Cell, 97, 145–148.
Bi, X., Corpina, R. A., and Goldberg, J. (2002) Nature, 419, 271–277.
Grosshans, B. L., Ortiz, D., and Novick, P. (2006) Proc. Natl. Acad. Sci. USA, 103, 11821–11827.
Presley, J. F., Cole, N. B., Schroer, T. A., Hirschberg, K., Zaal, K. J., and Lippincott-Schwartz, J. (1997) Nature, 389, 81–85.
Beard, M., Satoh, A., Shorter, J., and Warren, G. (2005) J. Biol. Chem., 280, 25840–25848.
Sonnichsen, B., Lowe, M., Levine, T., Jamsa, E., Svejstrup, B., and Warren, G. (1998) J. Cell Biol., 140, 1013–1021.
Bethune, J., Wieland, F., and Moelleken, J. (2006) J. Membr. Biol., 211, 65–79.
Tisdale, E. J. (1999) Mol. Biol. Cell, 10, 1837–1849.
Tisdale, E. J., Bourne, J. R., Khosravi-Far, R., Der, C. J., and Balch, W. E. (1992) J. Cell Biol., 119, 749–761.
Gallione, C. J., and Rose, J. K. (1985) J. Virol., 54, 374–382.
Tisdale, E. J., and Jackson, M. R. (1998) J. Biol. Chem., 273, 17269–17277.
Tisdale, E. J. (2000) Traffic., 1, 702–712.
Han, X., Ramanadham, S., Turk, J., and Gross, R. W. (1998) Biochim. Biophys. Acta, 1414, 95–107.
Forster, R., Weiss, M., Zimmermann, T., Reynaud, E. G., Verissimo, F., Stephens, D. J., and Pepperkok, R. (2006) Curr. Biol., 16, 173–179.
Barry, M., and Fruh, K. (2006) Sci. STKE, 2006, e21.
Tisdale, E. J., and Artalejo, C. R. (2006) J. Biol. Chem., 281, 8436–8442.
Robbins, A. R., Ward, R. D., and Oliver, C. (1995) J. Cell Biol., 130, 1093–1104.
Appenzeller-Herzog, C., and Hauri, H. P. (2006) J. Cell Sci., 119, 2173–2183.
Tisdale, E. J. (2005) Meth. Enzymol., 403, 381–391.
Lippincott-Schwartz, J., Cole, N. B., Marotta, A., Conrad, P.A., and Bloom, G. S. (1995) J. Cell Biol., 128, 293–306.
Roghi, C., and Allan, V. J. (1999) J. Cell Sci., 112 (Pt. 24), 4673–4685.
Patterson, R. L., van Rossum, D. B., Kaplin, A. I., Barrow, R. K., and Snyder, S. H. (2005) Proc. Natl. Acad. Sci. USA, 102, 1357–1359.
Burri, L., Varlamov, O., Doege, C. A., Hofmann, K., Beilharz, T., Rothman, J. E., Sollner, T. H., and Lithgow, T. (2003) Proc. Natl. Acad. Sci. USA, 100, 9873–9877.
Dilcher, M., Veith, B., Chidambaram, S., Hartmann, E., Schmitt, H. D., and Fischer von Mollard, G. (2003) EMBO J., 22, 3664–3674.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A. V. Bryksin, P. P. Laktionov, 2008, published in Biokhimiya, 2008, Vol. 73, No. 6, pp. 773–781.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bryksin, A.V., Laktionov, P.P. Role of glyceraldehyde-3-phosphate dehydrogenase in vesicular transport from Golgi apparatus to endoplasmic reticulum. Biochemistry Moscow 73, 619–625 (2008). https://doi.org/10.1134/S0006297908060011
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297908060011