Abstract
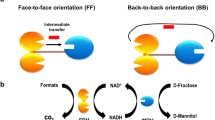
Trehalose has been utilized in the food, cosmetic, and pharmaceutical industries due to its ability to stabilize biomolecules. In this study, we developed a strategy of constructing a self-assembling supramolecular multienzyme complex for the production of trehalose using SpyCatcher and SpyTag. Maltooligosyl trehalose synthase (MTS) and maltooligosyl trehalose trehalohydrolase (MTH) were fused to SpyCatcher and its corresponding ligand, SpyTag. The fused MTS-SpyCatcher (MTS-SC) and MTH-SpyTag (MTH-ST) self-assembled into a supramolecular multienzyme complex (MTS-MTH) via the isopeptide bonds formed between SpyCatcher and SpyTag. MTS-MTH showed the faster reaction rate, which reached the peak of trehalose conversion rate 10 h earlier than the free enzymes. The results demonstrate that the multienzyme self-assembly via the protein-peptide interactions of SpyCatcher and SpyTag can increase the catalytic efficiency of sequential reactions.






Similar content being viewed by others
REFERENCES
Ohtake, S. and Wang, Y.J., J. Pharm. Sci., 2011, vol. 100, no. 6, pp. 2020–2053.
Sola-Penna, M. and Meyer-Fernandes, J.R., Arch. Biochem. Biophys., 1998, vol. 360, no. 1, pp. 10–14.
Elbein, A.D., Pan, Y.T., Pastuszak, I., and Carroll, D., Glycobiology, 2003, vol. 13, no. 4, pp. 17–27.
Crowe, J.H., Crowe, L.M., and Chapman, D., Science, 1984, vol. 223, no. 4637, pp. 701–703.
Richards, A.B., Krakowka, S., Dexter, L.B., Schmid, H., Wolterbeek, A.P.M., Waalkens-Berendsen D.H., et al., Food Chem. Toxicol., 2002, vol. 40, no. 7, pp. 871–898.
Feofilova, E.P., Usov, A.I., Mysyakina, I.S., and Kochkina, G.A., Microbiology, 2014, vol. 83, no. 3, pp. 184–194.
Eastmond, P.J. and Graham I.A., Curr. Opin. Plant Bio-l., 2003, vol. 6, no. 3, pp. 231–235.
Nishimoto, T., Nakano, M., Nakada, T., Chaen, H., Fukuda, S., Sugimoto, T., et al., Biosci. Biotech. Bioc., 1996, vol. 60, no. 4, pp. 640–644.
Maruta, K., Mitsuzumi, H., Nakada, T., Kubota, M., Chaen, H., Fukuda, S., et al., Biochim. Biophys. Acta, 1996, vol. 1291, no. 3, pp. 1–181.
Nakada, T., Maruta, K., Mitsuzumi, H., Kubota, M., Chaen, H., Sugimoto, T., et al., Biosci. Biotech. Biochem., 1996, vol. 59, no. 12, pp. 2215–2218.
Wheeldon, I., Minteer, S.D., Banta, S., Barton, S.C., Atanassov, P., and Sigman, M., Nat. Chem., 2016, vol. 8, no. 4, pp. 299–309.
Yang, Z., Gao, X., Xie, H., Wang, F., Ren, Y., and Wei, D., Biotechnol. Bioeng., 2017, vol. 114, no. 2, pp. 457–462.
Gao, X., Yang, S., Zhao, C., Ren, Y., and Wei, D., Angew. Chem. Int. Ed. Engl., 2014, vol. 53, no. 51, pp. 14 027–14 030.
Kim, S. and Hahn, J.S., J. Biotechnol., 2014, vol. 192, no. 20, pp. 192–196.
Reddington, S.C., and Howarth, M., Curr. Opin. Chem. Biol., 2015, vol. 29, pp. 94–99.
Pessino, V., Citron, R., Feng, S., and Huang, B., ChemBioChem, 2017, vol. 18, no. 15, pp. 1492–1495.
Veggiani, G., Zakeri, B., and Howarth, M., Trends. Biotechnol., 2014, vol. 32, no. 10, pp. 506–512.
Zakeri, B., Fierer, J.O., Celik, E., Chittock, E.C., Schwarz-linek, U., Moy, V.T., and Howarth, M., Proc. Natl. Acad. Sci. U.S.A., 2012, vol. 109, no. 12, pp. 4347–4348.
Zakeri, B., and Howarth, M., J. Am. Chem. Soc., 2010, vol. 132, no. 13, pp. 4526–4527.
Xu, C., Xu, Q., Huang, H., and Jiang, L., Biosci. Biotechnol. Biochem., 2018, vol. 82, no. 9, pp. 1473–1479.
Si, M., Xu, Q., and Jiang, L., Plos One, 2016, vol. 11, no. 9.
Bradford, M.M., Anal. Biochem., 1976, vol. 72, nos. 1–2, pp. 248–254.
Su, L., Wu, S., Feng, J., and Wu, J., Bioproc. Biosyst. Eng., 2019, vol. 42, no. 3, pp. 345–354.
Bao, J., Liu, N., Xu, Q., Zhu, L., Huang, H., and J-iang, L., J. Agr. Food. Chem., 2018, no. 66, pp. 8061–8068.
Gilbert, C., Howarth, M., Harwood, C.R., and Ellis, T., ACS. Synth. Biol., 2017, vol. 6, no. 6, pp. 957–967.
Veggiani, G., Nakamura, T., Brenner, M.D., Gayet R.V., Yan, J., Robinson, C.V., and Howarth, M., Proc. Natl. Acad. Sci. U.S.A., 2016, vol. 113, no. 5, pp. 1202–1207.
Kobayashi, K., Komeda, T., Miura, Y., Kettoku, M., and Kato, M., J. Ferment. Bioeng., 1997, vol. 83, no. 3, pp. 296–298.
Kim, Y.H., Kwon, T.K., Park, S., Seo, H.S., Cheong, J.J., Kim, C.H., et al., Appl. Environ. Microbiol., 2000, vol. 66, no. 11, pp. 4620–4624.
Mukai, K., Tabuchi, A., Nakada, T., Shibuya, T., Chaen, H., Fukuda, S., et al., Starch-Starke, 1997, no. 49, pp. 26–30.
Rehman, H.U., Aman, A., Nawaz, M.A., and Qader, S.A.U., Food Hydrocolloids, 2015, no. 43, pp. 819–824.
Szasz, G., Eur. J. Clin. Chem. Clin. Biochem, 1974, vol. 12, no. 4, pp. 166–170.
Hu, Y., Tang, S., Jiang, L., Zou, B., Yang, J., and Huang, H. Process Biochem., 2012, vol. 47, no. 12, pp. 2291–2299.
Pascale, D.D., Lernia, I.D., Sasso, M.P., Furia, A., Rosa, M.D., and Rossi, M., Extremophiles, 2002, vol. 6, no. 6, pp. 463–468.
Wang, J.H., Tsai, M.Y., Lee, G.C., and Shaw, J.F., J. Agr. Food Chem., 2007, vol. 55, no. 4, pp. 1256–1263.
Iturrate, L., Sánchez-Moreno, I., Doyagüez, E.G., and García-Junceda, E., Chem. Commun., 2009, no. 13, pp. 1721–1723.
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (no. 21606129, no. 31922070) and the Natural Science Fund for Colleges and Universities in Jiangsu Province (no. 17KJB416005).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Shen, X., Tang, S., Xu, Q. et al. SpyCatcher/SpyTag-Mediated Self-Assembly of a Supramolecular Complex for Improved Biocatalytic Production of Trehalose. Appl Biochem Microbiol 55, 596–602 (2019). https://doi.org/10.1134/S0003683819060115
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683819060115