Abstract
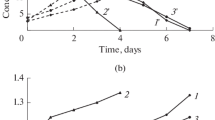
A comparative study of the oxidation of ferrous iron ions by various cultures of acidophilic chemolithotrophic microorganisms in solutions obtained after ferric leaching of copper-zinc concentrate at 80°C has been carried out. It was shown that the use of a moderately thermophilic culture for bioregeneration of leaching solutions was preferable. At the same time, the oxidation rate of Fe2+ ions reached 0.88 g/(L h), or 21.1 g/(L day). We propose that the activity of the moderately thermophilic culture was due to the presence of the mixotrophic bacteria Sulfobacillus spp., which used organic products of the microbial lysis for their growth. These products were formed during high-temperature ferric leaching of the copper-zinc concentrate with the biosolution.
Similar content being viewed by others
References
Fomchenko, N.V. and Muravyov, M.I., Appl. Biochem. Microbiol., 2017, vol. 53, no. 1, pp. 73–77.
Fomchenko, N.V. and Muravyov, M.I., Appl. Biochem. Microbiol., 2017, vol. 53, no. 4, pp. 448–452.
Fomchenko, N.V. and Muravyov, M.I., Appl. Biochem. Microbiol., 2017, vol. 53, no. 6, pp. 715–718.
Fomchenko, N.V., Muravyov, M.I., and Kondrat’eva, T.F., Hydrometallurgy, 2010, vol. 101, nos. 1–2, pp. 28–34.
Muravyov, M.I. and Bulaev, A.G., Miner. Eng., 2013, vol. 45, pp. 108–114.
Fomchenko, N.V. and Muravyov, M.I., Int. J. Miner. Proc., 2014, vol. 133, pp. 112–118.
Fomchenko, N.V., Kondrat’eva, T.F., and Muravyov, M.I., Hydrometallurgy, 2016, vol. 64, pp. 78–82.
Smalley, N. and Davis, G., Miner. Eng., 2000, vol. 13, no. 6, pp. 599–608.
Patel, B.C., Sinha, M.K., Tipre, D.R., Pillai, A., and Dave, S.R., Biores. Technol., 2014, vol. 157, pp. 310–315.
Fomchenko, N.V. and Biryukov, V.V., Appl. Biochem. Microbiol., 2009, vol. 45, no. 1, pp. 56–60.
Muravyov, M.I., Fomchenko, N.V., and Kondrat’eva, T.F., Adv. Mater. Res., 2015, vol. 1130, pp. 359–362.
Muravyov, M.I., Fomchenko, N.V., and Kondrat’eva, T.F., Appl. Biochem. Microbiol., 2011, vol. 47, no. 6, pp. 607–614.
Fomchenko, N.V. and Muravyov, M.I., Hydrometallurgy, 2017, vol. 174, pp. 116–122.
Silverman, M.P. and Lundgren, D.C., J. Bacteriol., 1959, vol. 77, no. 5, pp. 642–647.
Reznikov, A.A., Mulikovskaya, E.P., and Sokolov, I.Yu., Metody analiza prirodnykh vod (Methods of Analysis of Natural Waters), Moscow: Nedra, 1970.
Kondrat’eva, T.F., Bulaev, A.G., and Muravyov, M.I., Mikroorganizmy v biogeotekhnologiyakh pererabotki sul’fidnykh rud (Microorganisms in Biogeotechnologies for Processing Sulfide-Containing Ores), Moscow: Nauka, 2015.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.V. Fomchenko, M.I. Muravyov, V.S. Melamud, 2018, published in Prikladnaya Biokhimiya i Mikrobiologiya, 2018, Vol. 54, No. 4.
Rights and permissions
About this article
Cite this article
Fomchenko, N.V., Muravyov, M.I. & Melamud, V.S. Bioregeneration of Leaching Solutions during Two-Step Processing of Copper-Zinc Concentrate. Appl Biochem Microbiol 54, 432–435 (2018). https://doi.org/10.1134/S0003683818040075
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683818040075