Abstract
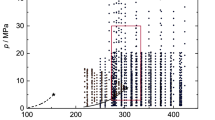
The pVT properties of the 0.075 n-C5H12 + 0.925H2O binary system were studied experimentally. Thermal properties were measured along nine different isochores over the density and temperature ranges 87–698 kg/m3 and 303–684 K at pressures up to 60 MPa. Measurements were performed using a constant-volume piezometer. Inflection and break points of the p(T) dependences were used to construct phase equilibrium curves, which were described by scaling equations.
Similar content being viewed by others
References
S. M. Rasulov and M. M. Khamidov, Prib. Tekh. Eksp., No. 1, 148 (1999).
P. H. Van Konynenburg and R. L. Scott, Philos. Trans. R. Soc. London, Ser. A 298(1442), 495 (1980).
E. Brunner, J. Chem. Thermodyn. 22, 335 (1990).
Th. W. de Loos, W. G. Penders, and R. N. Lichtenthaler, J. Chem. Thermodyn. 14, 83 (1982).
M. A. Anisimov, E. E. Gorodetskii, V. D. Kulikov, and J. V. Sengers, Phys. Rev. E: Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top. 51(2), 1199 (1995).
M. A. Anisimov, Critical Phenomena in Liquids and Liquid Crystals (Nauka, Moscow, 1987) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.M. Rasulov, A.R. Rasulov, 2009, published in Zhurnal Fizicheskoi Khimii, 2009, Vol. 83, No. 5, pp. 873–876.
Rights and permissions
About this article
Cite this article
Rasulov, S.M., Rasulov, A.R. The pVT properties and phase equilibria in the n-pentane-water binary system. Russ. J. Phys. Chem. 83, 756–759 (2009). https://doi.org/10.1134/S0036024409050136
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024409050136