Abstract
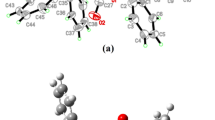
In this research, the formation of Hfm-Tin nanocluster in six configurations of Hfm-Tin and physicochemical behavior of CO2 adsorption on Hfm-Tin nanoclusters have been studied. The formation of the Hf-CO2 and Ti-CO2 bonds has been calculated using the DFT method. The effects of CO2 adsorption on structural variations and electronic properties have been studied. The adsorption energy ΔEads, energy gap (Eg), HOMO and LUMO energies, and dipole moments have been calculated at the 6-311++G** basis set, considering the constant bond lengths of the adsorbed CO2 molecules on Hfn-Ti5-n. The geometrical optimization has been performed by the B3PW91 method. The obtained results of adsorption values allow us to suggest that Hf doping on Ti nanocluster can noticeably improve the adsorption properties of all nanocluster models. Therefore, the decrease in global hardness and energy gaps after CO2 adsorption on Hfm-Tin nanoclusters can be attributed to the increase of chemical reactivity and hence leads to the lower stability of systems.
Similar content being viewed by others
References
P. A. León-Plata, M. R. Coan, and J. M. Seminario, J. Mol. Model. 19, 4419 (2013).
H. Akbarzadeh, H. Yaghoubi, A. Nasser Shamkhali, and F. Taherkhani, J. Phys. Chem. C 117, 26287 (2013).
F. Baletto and R. Ferrando, Rev. Mod. Phys. 77, 371 (2005).
Q. Zhang, J. Xie, Y. Yu, and J. Y. Lee, Nanoscale 2, 1962 (2010).
C. W. Bauschlicher, Jr., J. Chem. Phys. Lett. 462, 183 (2008).
J. H. Rose, J. R. Smith, F. Guinea, and J. Ferrante, Phys. Rev. B 29, 2963 (1984).
Z. Hu, J. G. Dong, J. R. Lombardi, and D. M. Lindsay, J. Phys. Chem. 97, 9263 (1993).
H. Wang, H. Haouari, R. Craig, Y. Liu, J. R. Lom-bardi, and D.M. Lindsay, J. Chem. Phys. 106, 2101 (1997).
M. D. Morse, J. Chem. Rev. 86, 1049 (1986).
U. Frenzel, U. Hammer, H. Westje, and D. Kreisle, J. Z. Phys. D: At., Mol. Clusters, 40, 108 (1997).
D. Dai, S. Roszak, and K. Balasubramanian, J. Chem. Phys. Lett. 308, 495 (1999).
D. Majumdar, D. Dai, and K. Balasubramanian, J. Chem. Phys. 113, 7919 (2000).
P. C. Jin, B. Han, and Z. W. Dai, and Z. J. Wu, J. Mol. Struct. (Theochem.) 680, 1 (2004).
C. Liu, H. He, P. Zapol, and L. A. Curtiss, Phys. Chem. Chem. Phys.: PCCP 48, 26584 (2014).
H. Kuze and S. Okude, Open J. Phys. Chem. 1, 109 (2011).
J.-Y. Zhao, Y. Zhang, F.-Q. Zhao, and X.-H. Ju, J. Phys. Chem. A 117, 12519 (2013).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
A. D. Becke, J. Chem. Phys. 98, 1372 (1993).
M. J. Frisch et al., Gaussian 03 (Gaussian, Pittsburgh, PA, 2003).
A. S. Ghasemi, E. Binaeian, H. A. Tayebi, and Y. Modanlou Jouybari, Int. J. Nano Dimens. 7, 247 (2016).
C. Lee, W. Yang, and R. G. Parr, J. Phys. Rev. B 37, 785 (1998).
P. K. Chattaraj and A. Poddar, J. Phys. Chem. A 103, 8691 (1999).
P. Geerlings, F. De Proft, and W. Langenaeker, Chem. Rev. 103, 1793 (2003).
R. G. Parr and W. Yang, Density-Functional Theory of Atoms and Molecules (Oxford Univ. Press, New York, 1989).
A. Soltani, S. Ghafouri Raz, V. Joveini Rezaei, A. Khalaji Dehno, and M. Savar, J. Appl. Surf. Sci. 263, 619 (2012).
A. Soltani, N. Ahmadian, A. Mirazami, A. Masoodi, E. Tazikeh Lemeski, and A. V. Moradi, J. Appl. Surf. Sci. 261, 262 (2012).
T. Koopmans, Physica, 1, 104 (1933).
Q. Sun, M. Wang, Z. Li, Y. Mad, and A. Du, Chem. Phys. Lett. 575, 59 (2013).
B. Gao, J.-X. Zhao, Q.-H. Cai, X.-G. Wang, and X.-Z. Wang, J. Phys. Chem. A 115, 9969 (2011).
A. Fielicke, P. Gruene, G. Meijer, and D. M. Rayner, Surf. Sci. 603, 1427 (2009).
A. Soltani, N. Ahmadian, Y. Kanani, A. Dehnokhalaji, and H. Mighani, J. Appl. Surf. Sci. 258, 9536 (2012).
R. G. Pearson, Inorg. Chem. 27, 734 (1988).
R. G. Pearson, Hard and Soft Acids and Bases (Dowden Hurchison Ross, Stroudsburg (Pa), 1973).
R. G. Pearson, J. Am. Chem. Soc. 85, 3533 (1963).
R. G. Pearson, Coord. Chem. Rev. 100, 403 (1990).
A. Soltani, Z. Azmoodeh, M. Bezi Javan, E. Tazikeh Lemeski, and L. Karami, Appl. Surf. Sci. 384, 230 (2016).
C.-G. Zhan, J. A. Nichols, and D. A. Dixon, J. Phys. Chem. A 107, 4184 (2003).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Ghasemi, A.S., Ashrafi, F. A Density-Functional Theory of CO2 Interaction with a Hafnium-Titanium Nanocluster. Russ. J. Inorg. Chem. 64, 88–97 (2019). https://doi.org/10.1134/S0036023619010108
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619010108