Abstract
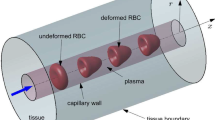
Multiple sets of experimental data have shown that the red blood cell (RBC) consumes nitric oxide (NO) about 600–1000-fold slower than the equivalent concentration of cell-free hemoglobin (Hb). Diffusion barriers of various sources have been suggested to explain this observation. In this work, a multicellular, spatially distributed, two-dimensional model, that describes the production, transport, and consumption of NO in blood vessels and the surrounding tissue, is developed. The model is used to assess the relative significance of NO transport barriers that reduce the rate of NO consumption in the blood. Unlike previous models of this system, the model developed here accounts explicitly for the presence of, and interactions among, a population of RBCs inside the lumen of the blood vessel and is, therefore, better suited to analyze, quantitatively, the contribution of each transport barrier as NO diffuses from its site of synthesis to the interior of the RBCs where it interacts with Hb. The model, which uses experimentally derived parameters, shows that extracellular unstirred boundary layer diffusion alone cannot account for the reduced NO consumption by RBC compared to an equivalent concentration of cell-free Hb. Since this result is reached using a two-dimensional representation of the RBCs, which overestimates the importance of the boundary layer diffusion resistance, it would be expected that in the real three-dimensional case, diffusion through the extracellular boundary layer would contribute even less to the overall mass transfer resistance. Consistent with recent experimental findings, the results of our model suggest that, under physiological conditions, transmembrane (membrane and its associated cytoskeleton layer) diffusion limitations in RBCs represent a key source of resistance for NO uptake by RBCs. © 2003 Biomedical Engineering Society.
PAC2003: 8716Uv, 8714Ee, 8715Vv, 8717Aa
Similar content being viewed by others
REFERENCES
Buerk, D. G.Can we model nitric oxide biotransport? A survey of mathematical models for a simple diatomic molecule with surprisingly complex biological activities. Annu. Rev. Biomed. Eng.3:109–143, 2001.
Butler, A. R., I. L. Megson, and P. G. Wright. Diffusion of nitric oxide and scavenging by the blood in the vasculature. Biochim. Biophys. Acta1425:168–176, 1998.
Carlsen, E., and J. H. Comroe, Jr.The rate of uptake of carbon monoxide and of nitric oxide by normal human erythrocytes and experimentally produced spherocytes. J. Gen. Physiol.42:83–107, 1958.
Cassoly, R., and Q. H. Gibson. Conformation, cooperativity, and ligand binding in human hemoglobin. J. Mol. Biol.91:3301–3313, 1974.
Eich, R. F., T. S. Li, D. D. Lemon, D. H. Doherty, S. R. Curry, J. F. Aitken, A. J. Mathews, K. A. Johnson, R. D. Smith, G. N. Phillips, and J. S. Olson. Mechanism of NO-induced oxidation of myoglobin and hemoglobin. Biochemistry35:6976–6983, 1996.
Kutchai, H., and N. C. Staub. Steady-state, hemoglobin-facilitated O2 transport in human erythrocytes. J. Gen. Physiol.53:576–589, 1969.
Herold, S., M. Exner, and T. Nauser. Kinetic and mechanistic studies of the NO-mediated oxidation of oxymyoglobin and oxyhemoglobin. Biochemistry40:3385–3395, 2001.
Huang, K., T. H. Han, D. R. Hyduke, M. W. Vaughn, H. V. Herle, T. W. Hein, C. Zhang, L. Kuo, and J. C. Liao. Modulation of nitric oxide bioavailability by erythrocytes. Proc. Natl. Acad. Sci. U.S.A.98:11771–11776, 2001.
L. J. Ignarro, Nitric Oxide: Biology and Pathobiology. CA: Academic, San Diego, 2000.
Jia, L., C. Bonaventura, J. Bonaventura, and J. S. Stamler. S-nitrosolhaemoglobin: A dynamic activity of blood involved in vascular control. Nature (London)380:221–226, 1996.
Lancaster, J.A tutorial on the diffusivity and reactivity of free nitric oxide. Nitric Oxide1:18–30, 1997.
Liao, J. C., T. W. Hein, M. W. Vaughn, K. Huang, and L. Kuo. Intravascular flow decreases erythrocyte consumption of nitric oxide. Proc. Natl. Acad. Sci. U.S.A.96:8757–8761, 1999.
Liu, X., M. J. S. Miller, M. S. Joshi, H. Sadowska-Krowicka, D. A. Clark, and J. R. Lancaster. Diffusion-limited reaction of free nitric oxide with erythrocytes. J. Biol. Chem.273:18709–18713, 1998.
Liu, X. P., A. Samouilov, J. R. Lancaster, and J. L. Zweier. Nitric oxide uptake by erythrocytes is primarily limited by extracellular diffusion not membrane resistance. J. Biol. Chem.277:26194–26199, 2002.
Malinski, T., Z. Taha, S. Grunfeld, S. Patton, M. Kapturczak, and P. Tomboulian. Diffusion of nitric oxide in the aorta wall monitored by porphyrinic microsensors. Biochem. Biophys. Res. Commun.193:1076–1082, 1993.
McMahon, T. J., R. E. Moon, B. P. Luschinger, M. S. Carraway, A. E. Stone, B. W. Stolp, A. J. Gow, J. R. Pawloski, P. Watke, D. J. Singel, C. A. Piantadosi, and J. S. Stamler. Nitric oxide in the human respiratory cycle. Nat. Med.8: 711–717 (2002).
Merchuk, J. C., Z. Tzure, and N. Lightfoot. Diffusional resistances to oxygen transfer in whole blood. Chem. Eng. Sci.38:1315–1321, 1983.
Pohl, U., and D. Lamontagne. Impaired tissue perfusion after inhibition of endothelium-derived nitric oxide. Basic Res. Cardiol.86:97–105, 1991.
Rice, S. A. Hydrodynamic and diffusion considerations of rapid-mix experiments with red blood cells. Biophys. J.29:65–78, 1980.
Schmidt-Schönbein, H., T. Fisher, G. Driessen, and H. Rieger. In: Microcirculation. Baltimore, MD: University Park Press, 1979, pp. 353–418.
Swihart, A. H., J. M. Mikrut, J. B. Ketterson, and R. C. Macdonald. Atomic force microscopy of the erythrocyte membrane skeleton. J. Microsc.204:212–225, 2001.
Takeuchi, M., H. Miyamoto, Y. Sako, H. Komizu, and A. Kusumi. Structure of the erythrocyte membrane skeleton as observed by atomic force microscopy. Biophys. J.74:2171–2183, 1998.
Tsoukias, N. M., and A. S. Popel. Erythrocyte consumption of nitric oxide in presence and absence of plasma-based hemoglobin. Am. J. Physiol.282:H2265-H2277, 2002.
Vandegriff, K. D., and J. S. Olson. The kinetics of O release by human red blood cells in the presence of external sodium dithionite. J. Biol. Chem.259:12609–12618, 1984.
Vandegriff, K. D., and J. S. Olson. Morphological and physiological factors affecting oxygen uptake and release by red blood cells. J. Biol. Chem.259:12619–12627, 1984.
Vaughn, M. W., K. T. Huang, L. Kuo, and J. C. Liao. Erythrocytes possess an intrinsic baarrier to nitric oxide consumption. J. Biol. Chem.275:2342–2348, 2000.
Vaughn, M. W., K. T. Huang, L. Kuo, and J. C. Liao. Erythrocytes consumption of nitric oxide: Competition experiment and model analysis. Nitric Oxide5:18–31, 2001.
Vaughn, M. W., L. Kuo, and J. C. Liao. Effective diffusion distance of nitric oxide in the micocirculation. Am. J. Physiol.43:H1705-H1714, 1998.
Vaughn, M. W., L. Kuo, and J. C. Liao. Estimation of nitric oxide production and reaction rates in tissue using a mathematical model. Am. J. Physiol.43:H2163-H2176, 1998.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Farra, N.H., Christofides, P.D. & Liao, J.C. Analysis of Nitric Oxide Consumption by Erythrocytes in Blood Vessels using a Distributed Multicellular Model. Annals of Biomedical Engineering 31, 294–309 (2003). https://doi.org/10.1114/1.1553454
Issue Date:
DOI: https://doi.org/10.1114/1.1553454