Abstract
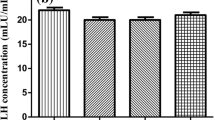
The androgen receptor (AR) is a ligand-dependent transcription factor involved in the regulation of many different physiological processes. Dysfunction of AR causes diverse clinical conditions, such as testicular feminization mutation (Tfm) syndrome and prostate cancer. However, the molecular basis of the AR in these disorders largely remains unknown, as a result of a lack of genetic models. Using conditional targeting technique with Cre-loxP system, we successfully generated null AR mutant (ARKO) mice. The ARKO males grew healthily, but they showed typical Tfm abnormalities. The ARKO males exhibited late onset of obesity with impaired bone metabolism and sexual behaviors. No overt abnormality was found in female ARKO mice, but a premature ovarian failurelike phenotype was found with impaired folliculogenesis. Thus, andorogen/AR system supports normal reproduction as well as normal female reproduction.
Similar content being viewed by others
References
Couse JF, Korach KS. Estrogen receptor null mice: what have we learned and where will they lead us?Endocr Rev 1999; 20: 358–417.
Wilson JD. The role of androgens in male gender role behavior.Endocr Rev 1999;20: 726–737.
Mangelsdorf DJ, Thummel C, Beato Met al. The nuclear receptor superfamily: the second decade.Cell 1995;83: 835–839.
He B, Kemppainen JA, Voegel JJ, Gronemeyer H, Wilson EM. Activation function 2 in the human androgen receptor ligand binding domain mediates interdomain communication with the NH (2)-terminal domain.J Biol Chem 1999;274: 37219–37225.
Watanabe M, Yanagisawa J, Kitagawa Het al. A subfamily of RNA-binding DEAD-box proteins acts as an estrogen receptor alpha coactivator through the N-terminal activation domain (AF-1) with an RNA coactivator, SRA.Embo J 2001;20: 1341–1352.
Shang Y, Myers M, Brown M. Formation of the androgen receptor transcription complex.Mol Cell 2002;9: 601–610.
Kato S, Endoh H, Masuhiro Yet al. Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase.Science 1995;270: 1491–1494.
Beato M, Herrlich P, Schutz G. Steroid hormone receptors: many actors in search of a plot.Cell 1995;83: 851–857.
Tora L, White J, Brou Cet al. The human estrogen receptor has two independent nonacidic transcriptional activation functions.Cell 1989;59: 477–487.
Glass CK, Rosenfeld MG. The coregulator exchange in transcriptional functions of nuclear receptors.Genes Dev 2000;14: 121–141.
Yanagisawa J, Kitagawa H, Yanagida Met al. Nuclear receptor function requires a TFTC-type histone acetyl transferase complex.Mol Cell 2002;9: 553–562.
Onate SA, Tsai SY, Tsai MJ, O’Malley BW. Sequence and characterization of a coactivator for the steroid hormone receptor superfamily.Science 1995;270: 1354–1357.
Spencer TE, Jenster G, Burcin MMet al. Steroid receptor coactivator-1 is a histone acetyltransferase.Nature 1997;389: 194–198.
Chen H, Lin RJ, Schiltz RLet al. Nuclear receptor coactivator ACTR is a novel histone acetyltransferase and forms a multimeric activation complex with P/CAF and CBP/p300.Cell 1997;90: 569–580.
Kamei Y, Xu L, Heinzel Tet al. A CBP integrator complex mediates transcriptional activation and AP-1 inhibition by nuclear receptors.Cell 1996;85: 403–414.
Naar AM, Beaurang PA, Zhou S, Abraham S, Solomon W, Tjian R. Composite co-activator ARC mediates chromatindirected transcriptional activation.Mature 1999;398: 828–832.
Radiez C, Lemon BD, Suldan Zet al. Ligand-dependent transcription activation by nuclear receptors requires the DRIP complex.Nature 1999;398: 824–828.
Fondell JD, Ge H, Roeder RG. Ligand induction of a transcriptionally active thyroid hormone receptor coactivator complex.Proc Matl Acad Sci U S A 1996;93: 8329–8333.
Murayama A, Kim MS, Yanagisawa J, Takeyama K, Kato S. Transrepression by a liganded nuclear receptor via a bHLH activator through co-regulator switching.Embo J 2004;23: 1598–1608.
Heinzel T, Lavinsky RM, Mullen TMet al. A complex containing N-CoR, mSin3 and histone deacetylase mediates transcriptional repression [see comments].Nature 1997;387: 43–48.
Ito T, Bulger M, Pazin MJ, Kobayashi R, Kadonaga JT. ACF, an ISWI-containing and ATP-utilizing chromatin assembly and remodeling factor.Cell 1997;90: 145–155.
Fyodorov DV, Kadonaga JT. The many faces of chromatin remodeling: Switching beyond transcription.Cell 2001;106: 523–525.
Lemon B, Inouye C, King DS, Tjian R. Selectivity of chromatinremodelling cofactors for ligand-activated transcription. Nature 2001;414: 924–928.
Kitagawa H, Fujiki R, Yoshimura Ket al. The chromatinremodeling complex WINAC targets a nuclear receptor to promoters and is impaired in Williams syndrome.Cell 2003;113: 905–917.
Fujiki R, Kim MS, Sasaki Y, Yoshimura K, Kitagawa H, Kato S. Ligand-induced transrepression by VDR through association of WSTF with acetylated histories.Embo J 2005;24: 3881–3894.
Narlikar GJ, Fan HY, Kingston RE. Cooperation between complexes that regulate chromatin structure and transcription.Cell 2002;108: 475–487.
DiRenzo J, Shang Y, Phelan Met al. BRG-1 is recruited to estrogen-responsive promoters and cooperates with factors involved in histone acetylation.Mol Cell Biol 2000;20: 7541–7549.
Chiba H, Muramatsu M, Nomoto A, Kato H. Two human homologues of Saccharomyces cerevisiae SWI2/SNF2 and Drosophila brahma are transcriptional coactivators cooperating with the estrogen receptor and the retinoic acid receptor.Nucl Acids Res 1994;22: 1815–1820.
Belandia B, Orford RL, Hurst HC, Parker MG. Targeting of SWI/SNF chromatin remodelling complexes to estrogenresponsive genes.Embo J 2002;21: 4094–4103.
Lubahn DB, Joseph DR, Sullivan PM, Willard HF, French FS, Wilson EM. Cloning of human androgen receptor complementary DNA and localization to the X chromosome.Science 1988;240: 327–330.
Griffin JE. Androgen resistance — the clinical and molecular spectrum.N Engl J Med 1992;326: 611–618.
McPhaul MJ. Molecular defects of the androgen receptor.J Steroid Biochem Mol Biol 1999;69: 315–322.
Takeyama K, Ito S, Yamamoto Aet al. Androgen-dependent neurodegeneration by polyglutamine-expanded human androgen receptor in Drosophila.Neuron 2002;35: 855–864.
Quigley CA, De Bellis A, Marschke KB, El-Awady MK, Wilson EM, French FS. Androgen receptor defects: historical, clinical, and molecular perspectives.Endocr Rev 1995;16: 271–321.
Li M, Indra AK, Warot Xet al. Skin abnormalities generated by temporally controlled RXRalpha mutations in mouse epidermis.Nature 2000;407: 633–636.
Sato T, Matsumoto T, Kawano Het al. Brain masculinization requires androgen receptor function.Proc Natl Acad Sci U S A 2004;101: 1673–1678.
Sato T, Matsumoto T, Yamada T, Watanabe T, Kawano H, Kato S. Late onset of obesity in male androgen receptordeficient (AR KO) mice.Biochem Biophys Res Commun 2003;300: 167–171.
Kawano LI, Sato T, Yamada Tet al. Suppressive function of androgen receptor in bone resorption.Proc Natl Acad Sci U S A 2003;100: 9416–9421.
Shiina H, Matsumoto T, Sato Tet al. Premature ovarian failure in androgen receptor-deficient mice.Proc Natl Acad Sci U S A 2006;103: 224–229.
Laml T, Preyer O, Umek W, Hengstschlager M, Hanzal H. Genetic disorders in premature ovarian failure.Hum Reprod Update 2002;8: 483–491.
Davison RM, Davis CJ, Conway GS. The X chromosome and ovarian failure.Clin Endocrinol (Oxf) 1999;51: 673–679.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Matsumoto, T., Kawano, H., Shiina, H. et al. Androgen receptor functions in male and female reproduction. Reprod Med Biol 6, 11–17 (2007). https://doi.org/10.1111/j.1447-0578.2007.00159.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1447-0578.2007.00159.x