Abstract
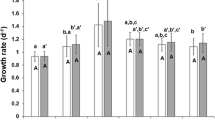
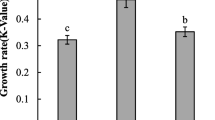
A continuous culture of rotifer was conducted to investigate the effect of combination feeding of both a high density of Nannochloropsis oculata (N) and condensed freshwater Chlorella (FC) on the fatty acid composition of L-type rotifer Brachionus plicatilis in a continuous culture system. The algal feeding of the rotifers was carried out in three successive steps: N-feeding → N+FC-feeding → FC-feeding. The culture was conducted at 24°C and 25–27 psu in a 2000mL bottle with 50% of water exchanged daily. The combination N+FC-feeding was effective in increasing rotifer density. The rotifers fed on N+FC (N+FC-R) had more non-polar lipids than polar ones, similar to those on N (N-R), opposite to the rotifers fed on FC (FC-R). N+FC-R contained higher levels of 16:2, 18:2n-6 (linoleic acid [LA]) and 20:2n-6, but lower levels of 18:1, 20:4n-6 (arachidonic acid), 20:5n-3 (eicosapentaenoic acid [EPA]) and 22:5n-3 (docosapentaenoic acid [DPA]) compared with N-R. Whereas N+FC-R contained higher levels of 16:1n-7, EPA and DPA, but lower levels of 16:2 and LA compared with FC-R. N+FC-R had more DPA in polar lipids than in non-polar ones. The ∑n-6/∑n-3 ratio in N+FC-R was 0.9–1.0, significantly different from those in N-R (0.4) and FC-R (6.6–8.4). Therefore, it is inferred that the fatty acid profile of the N+FC-R cultured in a continuous culture system was affected by both N and FC. Also, the combination N+FC-feeding may be effective in manipulating the ∑n-6/∑n-3 ratio in continuously cultured rotifers.
Similar content being viewed by others
References
Takeuchi T. A review of feed development for early life stages of marine finfish in Japan. Aquaculture 2001; 200: 203–222.
Takeuchi T, Wang Q, Furuita H, Hirota T, Ishida S, Hayasawa H. Development of microparticle diets for Japanese flounder Paralichthys olivaceus larvae. Fish. Sci. 2003; 69: 547–554.
Teshima S, Koshio S, Ishikawa M, Alam MS, Hernandez LHH. Effects of protein and lipid sources on the growth and survival of red sea bream Pagrus major and Japanese flounder Paralichthys olivaceus receiving micro-bound diets during larval and early juvenile stage. Aquacult. Nutr. 2004; 10: 279–287.
Takeuchi T. Food production for next generations with aquaculture: future direction and current scenario of seed production and aquaculture technology. Aquabiology 2005; 27: 120–128.
Maruyama I, Nakao T, Shigeno I, Ando Y, Hirayama K. Application of unicellular algae Chlorella vulgaris for the mass-culture of marine rotifer Brachionus. Hydrobiologia 1997; 358: 133–138.
Fu Y, Hada A, Yamashita T, Yoshida Y, Hino A. Development of a continuous culture system for stable mass production of the marine rotifer Brachionus. Hydrobiologia 1997; 358: 145–151.
Yoshimura K, Hagiwara A, Yoshimatsu T, Kitajima C. Culture technology of marine rotifers and the implications for intensive culture of marine fish in Japan. Mar. Freshwater Res. 1996; 47: 217–222.
Yoshimura K, Usuki K, Yoshimatsu T, Kitajima C, Hagiwara A. Recent development of a high density mass culture system for the rotifer Brachionus rotundiformis Tschugunoff. Hydrobiologia 1997; 358: 139–144.
Hino A. Advancements and prospects of the studies relating mass culture of marine rotifers. Nippon Suisan Gakkaishi 2001; 67: 1136–1137.
Kobayashi T, Nagase T, Kurano N, Hino A. Fatty acid composition of the L-type rotifer Brachionus plicatilis produced by a continuous culture system under the provision of high density Nannochloropsis. Nippon Suisan Gakkaishi 2005; 71: 328–334.
Kuwada H. Restructuring of a rotifer culture system as a live food for seed production of marine fish larvae. Res. J. Food Agric. 2004; 27: 18–23.
James CM, Rezeq TA. Intensive rotifer cultures using chemostats. Hydrobiologia 1989; 186/187: 423–430.
Rainuzzo JR, Reitan KI, Olsen Y. The significance of lipids at early stages of marine fish: a review. Aquaculture 1997; 155: 103–115.
Folch J, Lees M, Stanley GHS. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957; 226: 497–509.
Yu JP, Hino A, Ushiro M, Maeda M. Function of bacteria as vitamin B12 producers during mass culture of the rotifer Brachionus plicatilis. Nippon Suisan Gakkaishi 1989; 55: 1799–1806.
Scott JM. The vitamin B12 requirement of the marine rotifer Brachionus plicatilis. J. Mar. Biol. Ass. UK 1981; 61: 983–994.
Hirayama K, Funamoto H. Supplementary effect of several nutrients on nutritive deficiency of baker’s yeast for population growth of the rotifer Brachionus plicatilis. Nippon Suisan Gakkaishi 1983; 49: 505–510.
Yu JP, Hino A, Hirano R, Hirayama K. Vitamin B12-producing bacteria as a nutritive complement for a culture of the rotifer Brachionus plicatilis. Nippon Suisan Gakkaishi 1988; 54: 1873–1880.
Maruyama I, Hirayama K. The culture of the rotifer Brachionus plicatilis with Chlorella vulgaris containing vitamin B12 in its cells. J. World. Aquacult. Soc. 1993; 24: 194–198.
Kayama M. Essential fatty acids, their metabolism and function. In: Kayama M (ed.). AA, EPA, DHA-Highly Unsaturated Fatty Acids. Koseisha-Koseikaku, Tokyo. 1995; 44–81.
Sprecher H, Luthria DL, Mohammed BS, Baykousheva SP. Re-evaluation of the pathways for the biosynthesis of polyunsaturated fatty acids. J. Lipid Res. 1995; 36: 2471–2477.
Sargent JR, McEvoy LA, Bell JG. Requirements, presentation and sources of polyunsaturated fatty acids in marine fish larval feeds. Aquaculture 1997; 155: 117–127.
Berge JP, Barnathan G. Fatty acids from lipids of marine organisms; molecular biodiversity, roles as biomarkers, biologically active compounds, and economical aspects. Adv. Biochem. Eng. Biotechnol. 2005; 96: 49–125.
Teshima S, Kanazawa A, Horinouchi K, Yamasaki S, Hirata H. Phospholipids of the rotifers, prawn, and larval fish. Nippon Suisan Gakkaishi 1987; 53: 609–615.
Lubzens E, Marko A, Tietz A. De novo synthesis of fatty acids in the rotifer, Brachionus plicatilis. Aquaculture 1985; 47: 27–37.
Takeuchi T, Masuda R, Ishizaki Y, Watanabe T, Kanematsu M, Imaizumi K, Tsukamoto K. Determination of the requirement of larval striped jack for eicosapentaenoic acid and docosahexaenoic acid using enriched Artemia nauplii. Fish. Sci. 1996; 62: 760–765.
Furuita H, Takeuchi T, Uematsu K. Effects of eicosapentaenoic and docosahexaenoic acids on growth, survival and brain development of larval Japanese flounder (Paralichthys olivaceus). Aquaculture 1998; 161: 269–279.
Furuita H, Takeuchi T, Toyota M, Watanabe T. EPA and DHA requirements in early juvenile red sea bream using HUFA enriched Artemia nauplii. Fish. Sci. 1996; 62: 246–251.
Ishizaki Y, Takeuchi T, Watanabe T, Arimoto M, Shimizu K. A preliminary experiment on the effect of Artemia enriched with arachidonic acid on survival and growth of yellowtail. Fish. Sci. 1998; 64: 295–299.
Takeuchi T, Kobayashi T, Shimizu T, Sekiya S. The necessity and suitable feeding schedule of Artemia nauplii for larval mud crab. Nippon Suisan Gakkaishi 2000; 66: 984–992.
Kanazawa A. Biological activities of polyunsaturated fatty acids and phospholipids in aquatic animals. In: Fujimoto K (ed.). Properties and Physiological Activities of Fish Oils. Koseisha-Koseikaku, Tokyo. 1993; 69–79.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobayashi, T., Nagase, T., Hino, A. et al. Effect of combination feeding of Nannochloropsis and freshwater Chlorella on the fatty acid composition of rotifer Brachionus plicatilis in a continuous culture. Fish Sci 74, 649–656 (2008). https://doi.org/10.1111/j.1444-2906.2008.01570.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2008.01570.x