Abstract
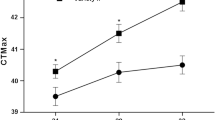
Thermal limits, induced thermotolerance and the expression of heat shock protein 70 (Hsp70) in an echinoderm Apostichopus japonicus were studied. The sublethal and lethal temperatures for the juveniles were 30 and 34°C, respectively; a previous sublethal heat shock exposure (30°C, 2 h) could increase the survival rates of the sea cucumbers when they were exposed to 34°C. This induced thermotolerance could last for at least 2 days. Levels of Hsp70 increased substantially after sublethal heat shock exposure and linearly decreased with time. This result indicated that a close relationship existed between the induction of thermotolerance and the levels of Hsp70 in A. japonicus.
Similar content being viewed by others
References
Weber IA. Relationship of heat shock proteins and induced thermal resistance. Cell. Prolif. 1992; 25: 101–113.
Trent JD, Gabrielsen M, Jensen B, Neuhard J, Olsen J. Acquired thermotolerance and heat shock proteins in thermophiles from the three phylogenetic domains. J. Bacteriol. 1994; 176: 6148–6152.
Clegg JS, Uhlinger KR, Jackson SA, Cherr GN, Rifkin E, Friedman CS. Induced thermotolerance and the heat shock protein-70 family in the Pacific oyster Crassostrea gigas. Mol. Mar. Biol. Biotechnol. 1998; 7: 21–30.
Hofmann GE, Somero GN. Evidence for protein damage at environmental temperatures: seasonal changes in levels of ubiquitin conjugates and hsp70 in the intertidal mussel, Mytilus trossulus. J. Exp. Biol. 1995; 198: 1509–1518.
Tomanek L, Somero GN. Evolutionary and acclimation-induced variation in the heat-shock responses of congeneric marine (genus Tegula) from different thermal habitats: implications for limits of thermotolerance and biogeography. J. Exp. Biol. 1999; 202: 2925–2936.
Buckley BA, Owen ME, Hofmann GE. Adjusting the thermostat: the threshold induction temperature for the heat-shock responses in intertidal mussels (genus Mytilus) changes as a function of thermal history. J. Exp. Biol. 2001; 204: 3571–3579.
Helmuth BS, Hofmann GE. Microhabitats, thermal heterogeneity, and patterns of physiological stress in the rocky intertidal zone. Biol. Bull. 2001; 201: 374–384.
Parsell DA, Lindquist S. The function of heat shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu. Rev. Genet. 1993; 27: 437–496.
Morimoto RI, Tissières A, Georgopoulos C. Stress Proteins in Biology and Medicine. Cold Harbor Laboratory Press, Cold Spring Harbor, NY. 1990.
Frydman J, Höhfeld J. Chaperones get in touch: the hip-hop connection. Trends Biochem. Sci. 1997; 22: 87–92.
Feder ME, Hofmann GE. Heat-shock proteins, molecular chaperones, and the stress responses: evolutionary and ecological physiology. Annu. Rev. Physiol. 1999; 61: 243–282.
Hamdoun AM, Cheney DP, Cherr GN. Phenotypic plasticity of HSP70 and HSP70 gene expression in the pacific oyster (Crassostrea gigas): implications for thermal limits and induction of thermal tolerance. Biol. Bull. 2003; 205: 160–169.
Sorte CJB, Hofmann GE. Thermotolerance and heat shock protein expression in Northeastern Pacific Nucella species with different biogeographical ranges. Mar. Biol. 2005; 148: 985–993.
Liao YL. Fauna Sinica: Phylum Echinodermata Class Holothuroidea, Science Press, Beijing. 1997.
Gavrilova GS. Regularities of distribution and present state of commercial echinoderm stocks in Peter the Great Bay (Japan Sea). In: Mooi R, Telford M (eds). Echinoderms: San Francisco: Proceedings of the Ninth International Echinoderm Conference San Francisco, California, USA, 5–9 August. Balkema, Rotterdam. 1996; 35.
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970; 227: 680–685.
Kyhse-Andersen J. Electroblotting of multiple gels: a simple apparatus without buffer tank for rapid transfer of proteins from polyacrylamide to nitrocellulose. J. Biochem. Biophys. Methods 1984; 10: 203–209.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principles of protein-dye binding. Anal. Biochem. 1976; 72: 248–254.
Finney DJ. Probit Analysis. Cambridge University Press, Cambridge. 1971.
Dong YW, Dong SL, Tian XL, Zhang MZ, Wang F. Effects of water temperature on growth, respiration and body composition of young sea cucumber Apostichopus japonicus. J. Fish. Sci. China 2005; 12: 33–38.
Dong YW, Dong SL. Growth and oxygen consumption of the juvenile sea cucumber Apostichopus japonicus (Selenka) at constant and fluctuating water temperatures. Aquac. Res. 2006; 37: 1327–1333.
Dong YW, Dong SL, Tian XL, Wang F, Zhang MZ. Effects of diel temperature fluctuations on growth, oxygen consumption and proximate body composition in the sea cucumber Apostichopus japonicus Selenka. Aquaculture 2006; 255: 514–521.
Nover L. Heat Shock Response. CRC Press, Boca Raton, CA. 1991.
Feige U, Morimoto RI, Yahara I, Polla BS. Stress-Inducible Cellular Responses. Birkhäuser-Verlag, Berlin. 1996.
Bonaventura R, Poma V, Costa C, Matranga V. UVB radiation prevents skeleton growth and stimulates the expression of stress markers in sea urchin embryos. Biochem. Biophys. Res. Commun. 2005; 328: 150–157.
Ellis RJ. The Chaperonins. Academic Press, New York, NY. 1996.
Lindquist S. The heat-shock response. Annu. Rev. Biochem. 1986; 55: 1151–1191.
Pörtner HO. Climate variations and the physiological basis of temperature dependent biogeography: systemic to molecular hierarchy of thermal tolerance in animals. Comp. Biochem. Physiol. 2002; 132A: 739–761.
Landry J, Zbernier D, Chretien P, Nicole LM, Tanguay RM, Marceau N. Synthesis and degradation of heat shock proteins during development and decay of thermotolerance. Cancer Res. 1982; 42: 2457–2461.
Tomanek L, Somero GN. Time course and magnitude of synthesis of heat-shock proteins in congeneric marine snails (genus Tegula) from different tidal heights. Physiol. Biochem. Zool. 2000; 73: 249–256.
Lindquist S. Varying patterns of protein synthesis in Drosophila during heat shock: implications for regulations. Dev. Biol. 1980; 77: 463–479.
Lindquist S. Regulation of protein synthesis during heat shock. Nature 1981; 293: 311–314.
Lindquist S. Autoregulation of the heat-shock response. In: Ilan J (ed.). Translational Regulation of Gene Expression 2. Plenum Press, New York. 1993: 279–320.
Storti RV, Scott MP, Rich A, Pardue ML. Translational control of protein synthesis in response to heat shock in D. melanogaster cells. Cell 1980; 22: 825–834.
Petersen NS, Mitchell HK. Recovery of protein synthesis after heat shock: prior heat treatment affects the ability of cells to translate mRNA. Proc. Natl. Acad. Sci. USA 1981; 78: 1708–1711.
Beck SC, De Maio A. Stabilization of protein synthesis in thermotolerant cells during heat shock. J. Biol. Chem. 1994: 269: 21803–21811.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dong, Y., Dong, S. Induced thermotolerance and expression of heat shock protein 70 in sea cucumber Apostichopus japonicus . Fish Sci 74, 573–578 (2008). https://doi.org/10.1111/j.1444-2906.2008.01560.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2008.01560.x