Abstract
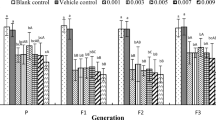
The effects of parental exposure to 17β-estradiol (E2) and 4-nonylphenol (4-NP) on life history parameters of three successive generations of the euryhaline cladoceran Diaphanosoma celebensis were assessed under laboratory conditions. Less than 24-h-old neonates (P) were exposed to five sublethal concentrations of E2 (0.1, 1, 10, 100 and 1000 μg/L) and 4-NP (0.01, 0.1, 1, 10 and 50 μg/L) at 25°C. Age at first reproduction, reproductive period, fecundity and lifespan were investigated. Successive generations (F1–F3) were monitored further in the absence of toxicants. Results showed that cladocerans exposed to 10–1000 μg/L E2 produced more neonates at a younger age compared to the control. The same effects were also observed in the F1 and F2 but ceased in F3. Cladocerans exposed to 1 μg/L 4-NP also produced significantly more neonates compared to the control, but this effect was not found in successive generations. The results suggest that vertebrate hormone can modify the reproduction of D. celebensis, and the effects are multigenerational.
Similar content being viewed by others
References
LeBlanc GA, Mu X, Rider CV. Embryotoxicity of the alklyphenol degradation product of 4-nonylphenol to the crustacean Daphnia magna. Environ. Health Perspect. 2000; 108: 1133–1138.
Oetken M, Bachmann J, Schulte-Oehlmann U, Oehlmann J, Evidence of endocrine disruption in invertebrates. Int. Rev. Cytol. 2004; 226: 1–44.
Shurin JB, Dodson SI. Sublethal toxic effects of cyanobacteria and nonylphenol on environmental sex determination and development in Daphnia. Environ. Toxicol. Chem. 1997; 16: 1269–1276.
Jaser W, Severin GF, Jütting U, Jüttner I, Schramm KW, Kettrup A. Effects of 17α-ethinylestradiol on the reproduction of the cladoceran species Ceriodaphnia reticulata and Sida crystallina. Environ. Int. 2003; 28: 633–638.
Zhang L, Gibble R, Baer KN. The effects of 4-nonylphenol and ethanol on acute toxicity, embryo development, and reproduction of Daphnia magna. Ecotoxicol. Environ. Saf. 2003; 55: 330–337.
Brennan SJ, Brougham CA, Roche JJ, Fogarty AM. Multigenerational effects of four selected environmental oestrogens on Daphnia magna. Chemosphere 2006; 64: 49–55.
Korovchinsky NM. Redescription of Diaphanosoma celebensis Stingelin, 1900 (Crustacea, Cladocera). Hydrobiologia 1989; 184: 7–22.
Segawa S, Yang WT. Growth, moult, reproduction and filtering rate of an estuarine cladoceran, Diaphanosoma celebensis, in laboratory culture. Bull. Plankton Soc. Jpn 1990; 37: 145–155.
Shrivastava Y, Mahambre GG, Achuthankutty CT, Fernandes B, Goswami SC, Madhupratap M. Parthenogenetic reproduction of Diaphanosoma celebensis (Crustacea: Cladocera). Effect of algae and algal density on survival, growth, life span and neonate production. Mar. Biol. 1999; 135: 663–670.
Achuthankutty CT, Shrivastava Y, Mahambre GG, Goswami SC, Madhupratap M. Parthenogenetic reproduction of Diaphanosoma celebensis (Crustacea: Cladocera): influence of salinity on feeding, survival, growth and neonate production. Mar. Biol. 2000; 137: 19–22.
de la Pena MR. Use of juvenile instar Diaphanosoma celebensis (Stingelin) in hatchery rearing of Asian sea bass Lates calcarifer (Bloch). Isr. J. of Aquaculture/Bamidgeh 2001; 53: 128–138.
Atienzar FA, Jha AN. The random amplified polymorphic DNA (RAPD) assay to determine DNA alterations, repair and transgenerational effects in B(a) P exposed Daphnia magna. Mutat. Res. 2004; 552: 125–140.
Anway MD, Cupp AS, Uzumcu M, Skinner MK. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005; 308: 1466–1469.
Lotka AJ. Vital statistics — a natural population norm. J. Wash. Acad. Sci. 1913; 3: 241–248.
Zar JH. Biostatistical Analysis. Prentice Hall. Upper Saddle River, NJ. 1999.
Comber MHI, Williams TD, Stewart KM. The effects of nonylphenol on Daphnia magna. Water Res. 1993; 27: 273–276.
Andersen HR, Halling-Sørensen B, Kusk KO. A parameter for detecting estrogenic exposure in the copepod Acartiatonsa. Ecotoxicol. Environ. Saf. 1999; 44: 56–61.
Watts MM, Pascoe D, Carroll KC. Population responses of the freshwater amphipod Gammarus pulex (L.) to an environmental estrogen, 17α-ethinylestradiol. Environ. Toxicol. Chem. 2002; 21: 445–450.
Sarojini R, Rao SS, Lakshmi KJ. Effects of steroids (estradiol and estrone) on the ovaries of the marine crab Scylla serrata. Comp. Physiol. Ecol. 1990; 15: 21–26.
Ghosh D, Ray AK. 17β-Hydroxysteriod dehydrogenase activity of ovary and hepatopancreas of freshwater prawn, Macrobrachium rosenbergii: relation to ovarian condition and estrogen treatment. Gen. Comp. Endocrinol. 1993; 89: 248–254.
Routledge ES, Sumpter JP. Estrogenic activity of surfactants and some of their degradation products assessed using recombinant yeast screen. Environ. Toxicol. Chem. 1996; 15: 241–248.
Song M, Xu Y, Jiang Q, Lam PKS, O’Toole DK, Giesy JP Jiang G. Measurement of estrogenic activity in sediments from Haine and Dagu River, China. Environ. Int. 2002; 32: 676–681.
Nunes BS, Carvalho FD, Guilhermino LM, Van Stappen G. Use of the genus Artemia in ecotoxicity testing. Environ. Pollut. 2006; 144: 453–462.
Blackburn MA, Waldock MJ Concentrations of alkylphenols in rivers and estuaries in England and Wales. Water Res. 1995; 29: 1623–1629.
Ying GG, Kookana RS, Ru YJ. Occurrence and fate of hormone steroids in the environment. Environt. Int. 2002; 28: 545–551.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marcial, H.S., Hagiwara, A. Multigenerational effects of 17β-estradiol and nonylphenol on euryhaline cladoceran Diaphanosoma celebensis . Fish Sci 73, 324–330 (2007). https://doi.org/10.1111/j.1444-2906.2007.01338.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2007.01338.x