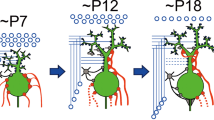
Abstract
The climbing fiber input to the cerebellum is crucial for its normal function but those factors which control the development of this precisely organized pathway are not fully elucidated. The neurotrophins are a family of peptides, which have many roles during development of the nervous system, including the cerebellum. Since the cerebellum and inferior olive express neurotrophins and their receptors, we propose that neurotrophins are involved in the regulation of climbing fiber development. Here we review the temporo-spatial expression of neurotrophins and their receptors at key ages during climbing fiber development and then examine evidence linking neurotrophins to climbing fiber development, including some of the intracellular pathways involved. During prenatal development the expression of neurotrophins in the hindbrain coupled with their function in neurogenesis and migration, is consistent with a role of NT3 in inferior olivary genesis. Subsequently, cerebellar expression of two neurotrophins, NT3 and NT4, is concurrent with olivary receptor expression and the time of olivary axonal outgrowth and this continues postnatally during early climbing fiber synaptogenesis on Purkinje cells. The expression-pattern of neurotrophins changes with age, with falling NGF, NT3 and NT4 but increasing granule cell BDNF. Importantly, olivary expression of neurotrophin receptors, and therefore climbing fiber responsiveness to neurotrophins, falls specifically during maturation of the climbing fiber-Purkinje cell synapse. The function of BDNF is less certain, but experimental studies indicate that it has a role in climbing fiber innervation of Purkinje cells, particularly synaptogenesis and synaptic plasticity. Its importance is highlighted by the overlap of BDNF signalling with several cellular pathways, which regulate climbing fiber maturation. From the data presented, we propose not only that neurotrophins are involved in climbing fiber development, but also that several act in a specific temporal order.
Similar content being viewed by others
References
Ito M. The cerebellum and neural control, (1st edn). New York: Raven Press, 1984.
Azizi SA, Woodward DJ. Inferior olivary nuclear complex of the rat: morphology and comments on the principles of organisation within the olivocerebellar system. J Comp Neurol 1987; 263: 467–484
Chedotal A, Sotelo C. Early development of olivocerebellar projections in the fetal rat using CGRP immunocytochemistry. Eur J Neurosci 1992; 4: 1159–1179.
Zagrebelsky M, Rossi F. Postnatal development and adult organisation of the olivocerebellar projection map in the hypogranular cerebellum of the rat. J Comp Neurol 1999; 407: 527–542.
Sotelo C, Chedotal A. Development of the olivocerebellar projection. Persp Dev Neurobiol 1997; 5: 57–67.
Henderson CE. Role of neurotrophic factors in neuronal development. Curr Opin Neurobiol 1996; 6: 64–70.
Maisonpierre PC, Belluscio L, Friedman B, et al. NT-3, BDNF and NGF in the developing rat nervous system: parallel as well as reciprocal patterns of expression. Neuron 1990; 5: 501–509.
Bondy CA. Transient IGF-I gene expression during the maturation of functionally related central projection neurones. J Neurosci 1991; 11: 3442–3455.
Rocamora N, Garcia-Ladona FJ, Palacios JM, Mengod G. Differential expression of brain-derived neurotrophic factor, neurotrophin-3 and low-affinity nerve growth factor receptor during postnatal development of the rat cerebellar system. Mol Brain Res 1993; 17: 1–8.
Lindholm D, Hamner S, Zirrgiebel U. Neurotrophins and cerebellar development. Persp Dev Neurobiol 1997; 5: 83–94.
Ringstedt T, Largercrantz H, Persson H. Expression of members of thetrk family in the developing postnatal rat brain. Dev Brain Res 1993; 72: 119–131.
Riva-Depaty I, Dubreuil YL, Mariani J, Delhaye-Bouchaud N. Erachation of cerebellar granular cells alters the developmental expression of Trk receptors in the rat inferior olive. Int J Dev Neurosci 1998; 16: 49–62.
Nitz M, Bower AJ, Sherrard RM. Localization of low affinity nerve growth factor receptor in the rat inferior olivary complex during development and plasticity of climbing fibers. Dev Brain Res 2001; 126: 229–239.
Lewin GR, Barde Y-A. Physiology of neurotrophins. Ann Rev Neurosci 1996; 19: 289–317.
Radeke MJ, Misko TP, Hsu C, Herzenberg LA, Shooter EM. Gene transfer and molecular cloning of the rat nerve growth factor receptor. Nature 1987; 325: 593–597.
Barbacid M. The trk family of neurotrophin receptors. J Neurobiol 1994; 25: 1386–1403.
Barker PA. p75NTR: a study in contrasts. Cell Death Differ 1998; 5: 346–356.
Snider WD, Lichtman JW. Are neurotrophins synaptotrophins? Mol Cell Neurosci 1996; 7: 433–442.
Smith MA, Zhang LX, Lyons WE, Mamounas LA. Anterograde transport of endogenous brain-derived neurotrophic factor in hippocampal mossy fibers. Neuroreport 1997; 8: 1829–1834.
Tucker KL, Meyer M, Barde Y-A. Neurotrophins are required for nerve growth during development Nat Neurosci 2001; 4: 29–37.
Horch HW, Kruttgen A, Portbury SD, Katz LC. Destabilization of cortical dendrites and spines by BDNF. Neuron 1999; 23: 353–364.
Hanover JL, Huang ZJ, Tonegawa S, Stryker MP. Brain-derived neurotrophic factor overexpression induces precocious critical period in mouse visual cortex. J Neurosci 1999; 19: 1–5.
Altman J. Morphological development of the rat cerebellum and some of its mechanisms. In: Palay S, Chan-Palay V, editors. The cerebellum: new vistas. Berlin: Springer-Verlag, 1982: 8–49.
Crepel F, Mariani J, Delhaye-Bouchaud N. Evidence for a multiple innervation of Purkinje cells by climbing fibers in the immature rat cerebellum. J Neurobiol 1976; 7: 567–578.
Mariani J, Changeux J-P. Ontogenesis of olivocerebellar relationships. I. Studies by intracellular recordings of the multiple innervation of Purkinje cells by climbing fibers in the developing rat cerebellum. J Neurosci 1981; 1: 696–702.
Bourrat F, Sotelo C. Early development of the rat precerebellar system: migratory routes, selective aggregation and neuritic differentiation of the inferior olive and lateral reticular nucleus neurons. An overview. Arch Ital Biol 1990; 128: 151–170.
Lamballe F, Smeyne RJ, Barbacid M. Developmental expression of trkC, the neurotrophin-3 receptor, in the mammalian nervous system. J Neurosci 1994; 14: 14–28.
Wiklund P, Ekstrom PAR. Axonal outgrowth from adult mouse nodose ganglia in vitro is stimulated by neurotrophin-4 in a Trk receptor and mitogen-activated protein kinase-dependent way. J Neurobiol 2000; 45: 142–151.
Timmusk T, Belluardo N, Metsis M, Persson H. Widespread and developmentally regulated expression of neurotrophin-4 messenger RNA in rat brain and peripheral tissues. Eur J Neurosci 1993; 5: 605–613.
Yan O, Johnson EM, Jr. An immunohistochemical study of the nerve growth factor receptor in developing rats. J Neurosci 1988; 8: 3481–3498.
Taniuchi M, Johnson EMJ. Characterisation of the binding properties and retrograde axonal transport of a monoclonal antibody directed against nerve growth factor receptor. J Cell Biol 1985; 101: 1100–1106.
Ernfors P, Merlio JP, Persson H. Cells expressing mRNA for neurotrophins and their receptors during embryonic rat development. Eur J Neurosci 1992; 4: 1140–1158.
Atwal JK, Massie B, Miller FD, Kaplan DR. The trkB-Shc site signals neuronal survival and local axon growth via MEK and PI3-kinase. Neuron 2000; 27: 265–277.
Veeranna AND, Ahn NG, Jaffe H et al. Mitogen-activated protein kinases (Erk1,2) phosphorylate Lys-Ser-Pro (KSP) repeats in neurofilament proteins NF-H and NF-M. J Neurosci 1998; 18: 4008–4021.
Garcia-Rocha M, Avila J. Characterization of microtubule-associated protein phosphoisoforms present in isolated growth cones. Dev Brain Res 1995; 89: 47–55.
Mason CA, Christakos S, Catalano SM. Early climbing fiber interactions with Purkinje cells in the postnatal mouse cerebellum. J Comp Neurol 1990; 297: 77–90.
Chedotal A, Sotelo C. The “creeper stage” in cerebellar climbing fiber synaptogenesis precedes the “pericellular nest”—ultrastructural evidence with parvalbumin immunocytochemistry. Dev Brain Res 1993; 76: 207–220.
O'Leary J, Inukai J, Smith JM. Histogenesis of the cerebellar climbing fiber in the rat. J Comp Neurol 1971; 142: 377–392.
Neveu I, Arenas E. Neurotrophins promote the survival and development of neurons in the cerebellum of hypothyroid rats in vivo. J Cell Biol 1996; 133: 631–646.
Large TH, Bodary SC, Clegg DO et al. Nerve growth factor gene expression in the developing rat brain. Science 1986; 234: 352–355.
Lu B, Buck CR, Dreyfus CF, Black IB. Expression of NGF and NGF receptor mRNAs in the developing brain: evidence for local delivery and action of NGF. Exp Neurol 1989; 104: 191–199.
Das KP, Chao SL, White LD et al. Differential patterns of nerve growth factor, brain derived neurotrophic factor and neurotrophin-3 mRNA and protein levels in developing regions of rat brain. Neuroscience 2001; 103: 739–761.
Castren E, Thoenen H, Lindholm D. Brain derived neurotrophic factor messenger RNA is expressed in the septum, hypothalamus and adrenergic brainstem nuclei of adult rat brain and is increased by osmotic stimulation in the paraventricular nucleus. Neuroscience 1995; 64: 71–80.
Friedman WJ, Black IB, Kaplan DR. Distribution of the neurotrophins brain-derived neurotrophic factor, neurotrophin-3 and neurotrophin-4/5 in the postnatal rat brain: an immunocytochemical study. Neuroscience 1998; 84: 101–114.
McCallister AK, Katz LC, Lo DC. Neurotrophins and synaptic plasticity. Ann Rev Neurosci 1999; 22: 295–318.
Segal RA, Pomeroy SL, Stiles CD. Axonal growth and fasciculation linked to differential expression of BDNF and NT3 receptors in developing cerebellar granule cells. J Neurosci 1995; 15: 4970–4981.
Masana Y, Wanaka A, Kato H, Asai T, Tohyama M. Localisation of trkB mRNA in postnatal brain development. J Neurosci Res 1993; 35: 468–479.
Gao WQ, Zheng JL, Karihaloo M. Neurotrophin-4/5 (NT-4/5) and brain-derived neurotrophic factor (BDNF) act at later stages of cerebellar granule cell differentiation. J Neurosci 1995; 15: 2656–2667.
Yan Q, Radeke MJ, Matheson C et al. Immunocytochemical localisation of TrkB in the central nervous system of the adult rat. J Comp Neurol 1997; 378: 135–157.
Sugihara I, Wu HS, Shinoda Y. Morphology of single olivocerebellar axons labeled with biotinylated dextran amine in the rat. J Comp Neurol 1999; 414: 131–148.
Morrison ME, Mason CA. Granule neuron regulation of Purkinje cell development—striking a balance between neurotrophin and glutamate signaling. J Neurosci 1998; 18: 3563–3573.
Shimada A, Mason CA, Morrison ME. TrkB signaling modulates spine density and morphology independent of dendrite structure in cultured neonatal Purkinje cells. J Neurosci 1998; 18: 8559–8570.
Hirai H, Launey T. The regulatory connection between the activity of granule cell NMDA receptors and dendritic differentiation of cerebellar Purkinje cells. J Neurosci 2000; 20: 5217–5224.
Rabacchi SA, Bailly Y, Delhaye-Bouchaud N, Herrup K, Mariani J. Role of the target in synapse elimination: studies in cerebellum of developinglurcher mutants and adult chimeric mice. J Neurosci 1992; 12: 4712–4720.
Cohen-Cory S, Fraser SE. Effects of brain-derived neurotrophic factor on optic axonal branching and remodelingin vivo. Nature 1995; 378: 192–196.
Causing CG, Gloster A, Aloyz R et al. Synaptic innervation density is regulated by neuron-derived BDNF. Neuron 1997; 18: 257–267.
Campenot RB. Local control of neurite development by nerve growth factor. Proc Natl Acad Sci 1977; 76: 1494–1496.
Blanquet P. Interplay between BDNF and CaMK: an important regulatory mechanism of synaptic plasticity in the hippocampus. Med Sci 1998; 14: 722–728.
Fournier AE, Beer J, Arregui CO et al. Brain-derived neurotrophic factor modulates GAP-43 but not Tα1 expression in injured retinal ganglion cells of adult rats. J Neurosci Res 1997; 47: 561–572.
Schwab ME. Structural plasticity of the adult CNS. Negative control by neurite growth inhibitory signals. Int J Dev Neurosci 1996; 14: 379–385.
Console-Bram LM, Fitzpatrick-McElligott SG, McElligott JG. Distribution of GAP-43 mRNA in the immature and adult cerebellum: a role for GAP-43 in cerebellar development and neuroplasticity. Dev Brain Res 1996; 95: 97–106.
Buffo A, Fronte M, Oestreicher AB, Rossi F. Degenerative phenomena and reactive modifications of the adult rat inferior olivary neurons following axotomy and disconnection form their targets. Neuroscience 1998; 85: 587–604.
Whitmarsh AJ, Davis RJ. Signal transduction by target-derived neurotrophins. Nat Neurosci 2001; 4: 963–964.
Minichiello L, Klein R. TrkB and trkC neurotrophin receptors cooperate in promoting survival of hippocampal and cerebellar granule neurons. Gene Dev 1996; 10: 2849–2858.
Schwartz PM, Borghesani PR, Levy RL, Pomeroy SL, Segal RA. Abnormal cerebellar development and foliation in BDNF− mice reveals a role for neurotrophins in CNS patterning. Neuron 1997; 19: 269–281.
Bates B, Rios M, Trumpp A et al. Neurotrophin-3 is required for proper cerebellar development. Nat Neurosci 1999; 2: 115–117.
Rondi-Reig L, Delhaye-Bouchaud N, Mariani J, Caston J. Role of the inferior olivary complex in motor skills and motor learning in the adult rat. Neuroscience 1997; 77: 955–963.
Tojo H, Takami K, Kaisho Y et al. Neurotrophin-3 is expressed in the posterior lobe of mouse cerebellum, but does not affect the cerebellar development. Neurosci Letts 1995; 192: 169–172.
Sotelo C, Arsenio-Nunes ML. Development of Purkinje cells in the absence of climbing tibers. Brain Res 1976; 111: 389–395.
Bao SW, Chen L, Qiao XX, Knusel B, Thompson RF. Impaired eye-blink conditioning inwaggler, a mutant mouse with cerebellar BDNF deficiency. Learn Mem 1998; 5: 355–364.
Qiao XO, Chen L, Gao Het al. Cerebellar brain-derived neurotrophic factor-TrkB defect associated with impairment of eye-blink conditioning inStargazer mutant mice. J Neurosci 1998; 18: 6990–6999.
Chen L, Bao SW, Qiao XO, Thompson RF. Impaired cerebellar synapse maturation inwaggler, a mutant mouse with a disrupted neuronal calcium channel γ subunit. Proc Natl Acad Sci USA 1999; 96: 12132–12137.
Lindholm D, Dechant G, Heisenberg CP, Thoenen H. Brain-derived neurotrophic factor is a survival factor for cultured rat cerebellar granule neurons and protects them against glutamate-induced neurotoxicity. Eur J Neurosci 1993; 5: 1455–1464.
Thompson RF. Neural mechanisms of classical conditioning in mammals. Phil Trans R Soc Lond B 1990; 329: 161–170.
Bao SW, Chen L, Qiao XO, Thompson RF. Transgenic brain-derived neurotrophic factor modulates a developing cerebellar inhibitory synapse. Learn Mem 1999; 6: 276–283.
Angaut P, Alvarado-Mallart RM, Sotelo C. Ultrastructural evidence for compensatory sprouting of climbing and mossy afferents to the cerebellar hemisphere after ipsilateral pedunculotomy in the newborn rat. J Comp Neurol 1982; 205: 101–111.
Angaut P, Alvarado-Mallart RM, Sotelo C. Compensator climbing fiber innervation after unilateral pedunculotomy in the new born rat: origin and topographic organisation. J Comp Neurol 1985; 236: 161–178.
Sherrard RM, Bower AJ, Payne JN. Innervation of the adult rat cerebellar hemisphere by fibers from the ipsilateral inferior olive following unilateral neonatal pedunculotomy: an autoradiographic and retrograde fluorescent double-labelling study. Exp Brain Res 1986; 62: 411–421.
Zagrebelsky M, Strata P, Hawkes R, Rossi F. Reestablishment of the olivocerebellar projection may by compensatory transcommissural reinnervation following unilateral transection of the inferior cerebellar peduncle in the newborn rat. J Comp Neurol 1997; 379: 283–299.
Sherrard RM, Bower AJ. BDNF and NT-3 extend the critical period for developmental climbing fiber plasticity. Neuroreport 2001; 12: 2871–2874.
Dieringer N. ‘Vestibular compensation’: neural plasticity and its relations to functional recovery after labyrinthine lesions in frogs and other vertebrates. Prog Neurobiol 1995; 46: 97–129.
Li YX, Hashimoto T, Tokuyama W, Miyashita Y, Okuno H. Spatiotemporal dynamics of brain-derived neurotrophic factor mRNA induction in the vestibulo-olivary network during vestibular compensation. J Neurosci 2001; 21: 2738–2748.
Li YX, Tokuyama W, Okuno H, Miyashita Y, Hashimoto T. Differential induction of brain-derived neurotrophic factor mRNA in rat inferior olive subregions following unilateral labyrinthectomy. Neuroscience 2001; 106: 385–394.
Rabacchi SA, Bailly Y, Delhaye-Bouchaud N, Mariani J. Involvement of the N-methyl D-aspartate (NMDA) receptor in synapse elimination during cerebellar development. Science 1992; 256: 1823–1825.
Kakizawa S, Yamasaki M, Watanabe M, Kano M. Critical period for activity-dependent synapse elimination in developing cerebellum. J Neurosci 2000; 20: 4954–4961.
Suen PC, Wu K, Levine ES et al. Brain-derived neurotrophic factor rapidly enhances phosphorylation of the postsynaptic M-methyl-D-aspartate receptor subunit 1. Proc Natl Acad Sci USA 1997; 94: 8191–8195.
Kano M, Hashimoto K, Chen C et al. Impaired synapse elimination during cerebellar development in PKC gamma mutant mice. Cell 1995; 83: 1223–1231.
Chen C, Kano M, Abeliovich A et al. Impaired motor coordination correlates with persistent multiple climbing fiber innervation in PKCγ mutant mice. Cell 1995; 83: 1233–1242.
Mount HTJ, Elkabes S, Dreyfus CF, Black IB. Differential involvement of metabotropic and p75 neurotrophin receptors in effects of nerve growth factor and neurotrophin-3 on cultured Purkinje cell survival. J Neurochem 1998; 70: 1045–1053.
Zirrgiebel U, Ohga Y, Carter B et al. Characterization of trkB receptor-mediated signalling pathways in rat cerebellar granule neurons: involvement of protein kinase C in neuronal survival. J Neurochem 1995; 65: 2241–2250.
Coffey ET, Akerman KEO, Courtney MJ. Brain dervied neurotrophic factor induces a rapid upregulation of synaptophysin and tau proteins via the neurotrophin receptor TrkB in rat cerebellar granule cells. Neurosci Letts 1997; 227: 177–180.
Kano M, Hashimoto K, Watanabe M et al. Phospholipase cbeta4 is specifically involved in climbing fiber synapse elimination in the developing cerebellum. Proc Natl Acad Sci USA 1998; 95: 15724–15729.
Ribar TJ, Rodriguiz RM, Khiroug L et al. Cerebellar defects in Ca2+/calmodulin kinase IV-deficient mice. J Neurosci 2000; 20: 1–5.
Rondi-Reig L, Dubreuil YL, Martinou JC et al. Fear decrease in transgenic mice overexpressing bcl-2 in neurons. Neuroreport 1997; 8: 2429–2432.
Mariani J, Benoit P, Hoang MD, Thomson, MA, Delhaye-Bouchaud N. Extent of multiple innervation of cerebellar Purkinje cells by climbing fibers in adult X-irradiated rats. Comparison of different schedules of irradiation during the first postnatal week. Dev Brain Res 1990; 57: 63–70.
Qiao XO, Hefti F, Knusel B, Noebels JL. Selective failure of brain-derived neurotrophic factor mRNA expression in the cerebellum of stargazer, a mutant mouse with ataxia., J Neurosci 1996; 16: 640–648.
Dimberg Y, Vazquez M, Soderstrom S, Ebendal T. Effects of x-irradiation on nerve growth factor in the developing mouse brain. Tox Letts 1997; 90: 35–43.
Davies AM. The role of neurotrophins in the developing nervous system. J Neurosci 1994; 25: 1334–1348.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sherrard, R.M., Bower, A.J. Climbing fiber development: do neurotrophins have a part to play?. Cerebellum 1, 265–275 (2002). https://doi.org/10.1080/147342202320883579
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1080/147342202320883579