Abstract
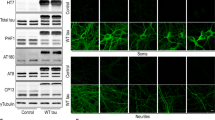
It has been suggested that terminally differentiated neuronal cells and mitotic cells respond differently in many aspects to herpes simplex virus type 1 (HSV-1) infection. The ICP4-deleted, Us3-defective, HSV-1 mutant strain d120 induces classical apoptosis in a variety of mitotic cell lines. Its behavior in postmitotic cells is not known. Here the authors report that mutant d120 virus failed to induce apoptosis in neuronal-like, nerve growth factor (NGF)-differentiated PC12 cells. More strikingly, rather than inducing apoptosis, d120 infection prolonged the life of nondividing NGF-differentiated PC12 cells in the culture flask. The virus genome had a half-life of 30 days. Unlike in other cells, such as Vero, neither wild-type nor d120 infection of NGF-differentiated PC12 cells induced the nuclear factor (NF)-κB p65 pathway, which has been associated with virus-induced apoptosis. Thus, the authors demonstrate, for the first time, that a potent apoptosis inducer mutant d120 failed to induce apoptosis in neuronal-like NGF-differentiated PC12 cells, unlike a number of other cell lines studied. The possible mechanisms involved in the failure of d120 to induce apoptosis in neuronal-like NGF-differentiated PC12 cells are discussed.
Similar content being viewed by others
References
Amici C, Belardo G, Rossi A, Santoro MG (2001). Activation of Ikappa B kinase by herpes simplex virus type 1. A novel target for anti-herpetic therapy. J Biol Chem 276: 28759–28766.
Amici C, Belardo G, Rozera C, Bernasconi D, Santoro MG (2004). Inhibition of herpesvirus-induced HIV-1 replication by cyclopentenone prostaglandins: role of I[kappa]B kinase (IKK). AIDS 18: 1271–1280.
Aubert M, Blaho JA (1999). The herpes simplex virus type 1 regulatory protein ICP27 is required for the prevention of apoptosis in infected human cells. J Virol 73: 2803–2813.
Azoitei N, Wirth T, Baumann B (2005). Activation of the IkappaB kinase complex is sufficient for neuronal differentiation of PC12 cells. J Neurochem 93: 1487–1501.
Chou J, Roizman B (1992). The {gamma}34.5 gene of herpes simplex virus 1 precludes neuroblastoma cells from triggering total shutoff of protein synthesis characteristic of programmed cell death in neuronal cells. Proc Natl Acad Sci U S A 89: 3266–3270.
Cui C, Griffiths A, Li G, Silva LM, Kramer MF, Gaasterland T, Wang XJ, Coen DM (2006). Prediction and identification of herpes simplex virus 1-encoded microRNAs. J Virol 80: 5499–5508.
DeLuca NA, McCarthy A, Schaffer PA (1985). Isolation and characterization of deletion mutants of herpes simplex virus type 1 in the gene encoding immediate-early regulatory protein ICP4. J Virol 56: 558–570.
Galvan V, Brandimarti R, Roizman B (1999). Herpes simplex virus 1 blocks caspase-3-independent and caspase-dependent pathways to cell death. J Virol 73: 3219–3226.
Galvan V, Roizman B (1998). Herpes simplex virus 1 induces and blocks apoptosis at multiple steps during infection and protects cells from exogenous inducers in a cell-type-dependent manner. Proc Natl Acad Sci U S A 95: 3931–3936.
Ghosh S, May MJ, Kopp EB (1998). NF-κB and REL proteins: evolutionarily conservedmediators of immune responses. Annu Rev Immunol 16: 225–260.
Goodkin ML, Ting AT, Blaho JA (2003). NF-{kappa}B is required for apoptosis prevention during herpes simplex virus type 1 infection. J Virol 77: 7261–7280.
Greene LA, Tischler AS (1976). Establishment of a nonadrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci U S A 73: 2424–2428.
Gregory D, Hargett D, Holmes D, Money E, Bachenheimer SL (2004). Efficient replication by herpes simplex virus type 1 involves activation of the I{kappa}B kinase-I{kappa}B-p65 pathway. J Virol 78: 13582–13590.
Gupta A, Gartner JJ, Sethupathy P, Hatzigeorgiou AG, Fraser NW (2006). Anti-apoptotic function of a microRNA encoded by the HSV-1 latency-associated transcript. Nature 442: 82–85.
Ishige K, Tanaka M, Arakawa M, Saito H, Ito Y (2005). Distinct nuclear factor-[kappa]B/Rel proteins have opposing modulatory effects in glutamate-induced cell death in HT22 cells. Neurochem Int 47: 545–555.
Jerome KR, Fox R, Chen Z, Sears AE, Lee HY, Corey L (1999). Herpes simplex virus inhibits apoptosis through the action of two genes, Us5 and Us3. J Virol 73: 8950–8957.
Koyama AH, Fukumori T, Fujita M, Irie H, Adachi A (2000). Physiological significance of apoptosis in animal virus infection. Microb Infect 2: 1111–1117.
Leopardi R, Roizman B (1996). The herpes simplex virus major regulatory protein ICP4 blocks apoptosis induced by the virus or by hyperthermia. Proc Natl Acad Sci U S A 93: 9583–9587.
Leopardi R, Van Sant C, Roizman B (1997). The herpes simplex virus 1 protein kinase US3 is required for protection from apoptosis induced by theávirus. Proc Natl Acad Sci U S A 94: 7891–7896.
Medici MA, Sciortino MT, Perri D, Amici C, Avitabile E, Ciotti M, Balestrieri E, De Smaele E, Franzoso G, Mastino A (2003). Protection by herpes simplex virus glycoprotein D against Fas-mediated apoptosis: role of nuclear factor {kappa}B. J Biol Chem 278: 36059–36067.
Moxley M, Block T, Liu H-C, Fraser N, Perng G-C, Wechsler S, Su Y-H (2002). Herpes simplex virus type 1 infection prevents detachment of nerve growth factor-differentiated PC12 cells in culture. J Gen Virol 83: 1591–1600.
Munger J, Chee A, Roizman B (2001). The Us3 protein kinase blocks apoptosis induced by the d120 mutant of herpes simplex virus 1 at premitochondrial stage. J Virol 75: 5491–5497.
Ono T, Sekino-Suzuki N, Kikkawa Y, Yonekawa H, Kawashima S (2003). Alivin 1, a novel neuronal activity-dependent gene, inhibits apoptosis and promotes survival of cerebellar granule neurons. J Neurosci 23: 5887–5896.
Patel A, Hanson J, McLean TI, Olgiate J, Hilton M, Miller WE, Bachenheimer SL (1998). Herpes simplex virus type 1 induction of persistent NF-[kappa]B nuclear translocation increases the efficiency of virus replication. Virology 247: 212–222.
Pizzi M, Goffi F, Boroni F, Benarese M, Perkins SE, Liou HC, Spano P (2002). Opposing roles for NF-kappa B/Rel Factors p65 and c-Rel in the modulation of neuron survival elicited by glutamate and interleukin-1beta. J Biol Chem 277: 20717–20723.
Santoro MG, Rossi A, Amici C (2003). NF-kB and virus infection: who controls whom. EMBO J 22: 2552–2560.
Su Y-H, Meegalla RL, Chowan R, Cubitt C, Oakes JE, Lausch RL, Fraser NW, Block TM (1999). Human corneal cells and other fibroblasts can stimulate the appearance of herpes simplex virus from quiescently infected PC12 cells. J Virol 73: 4171–4180.
Su Y-H, Moxley M, Kejariwal R, Mehta A, Fraser NW, Block TM (2000). The HSV 1 genome in quiescently infected NGF differentiated PC12 cells can not be stimulated by HSV superinfection. J NeuroVirol 6: 341–349.
Su Y-H, Moxley M, Ng AK, Lin J, Jordan R, Fraser NW, Block TM (2002). Stability and circularization of herpes simplex virus type 1 genomes in quiescently infected PC12 cultures. J Gen Virol 83: 2943–2950.
Taddeo B, Esclatine A, Roizman B (2002). The patterns of accumulation of cellular RNAs in cells infected with a wild-type and a mutant herpes simplex virus 1 lacking the virion host shutoff gene. Proc Natl Acad Sci U S A 99: 17031–17036.
Taddeo B, Luo TR, Zhang W, Roizman B (2003). Activation of NF-{kappa}B in cells productively infected with HSV-1 depends on activated protein kinase R and plays no apparent role in blocking apoptosis. Proc Natl Acad Sci U S A 100: 12408–12413.
Taddeo B, Zhang W, Lakeman F, Roizman B (2004). Cells lacking NF-{kappa}B or in which NF-{kappa}B is not activated vary with respect to ability to sustain herpes simplex virus 1 replication and are not susceptible to apoptosis induced by a replication-incompetent mutant virus. J Virol 78: 11615–11621.
Yedowitz JC, Blaho JA (2005). Herpes simplex virus 2 modulates apoptosis and stimulates NF-[kappa]B nuclear translocation during infection in human epithelial HEp-2 cells. Virology 342: 297–310.
Zhou G, Galvan V, Campadelli-Fiume G, Roizman B (2000). Glycoprotein D or J delivered in trans blocks apoptosis in SK-N-SH cells induced by a herpes simplex virus 1 mutant lacking intact genes expressing both glycoproteins. J Virol 74: 11782–11791.
Zhou G, Roizman B (2000). Wild-type herpes simplex virus 1 blocks programmed cell death and release of cytochrome c but not the translocation of mitochondrial apoptosis-inducing factor to the nuclei of human embryonic lung fibroblasts. J Virol 74: 9048–9053.
Zhou G, Roizman B (2001). The domains of glycoprotein D required to block apoptosis depend on whether glycoprotein D is present in the virions carrying herpes simplex virus 1 genome lacking the gene encoding the glycoprotein. J Virol 75: 6166–6172.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by National Institutes of Health Grant NS 33768 and an appropriation from The Commonwealth of Pennsylvania through The Institute for Hepatitis and Virus Research.
Rights and permissions
About this article
Cite this article
Aiamkitsumrit, B., Zhang, X., Block, T.M. et al. Herpes simplex virus type 1 ICP4 deletion mutant virus d120 infection failed to induce apoptosis in nerve growth factor-differentiated PC12 cells. Journal of NeuroVirology 13, 305–314 (2007). https://doi.org/10.1080/13550280701361490
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1080/13550280701361490