Abstract
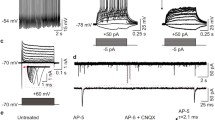
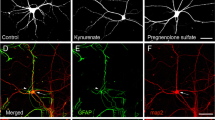
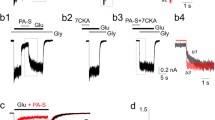
We charactrized glutamate receptor-mediated toxicity in mouse fibroblasts expressing the human NR1a/2A or NR1a/2B NMDA receptor. After induction of NMDA receptor, cells in both lines died over a 24 h time period. This toxicity was associated with a progressive increase in the glutamate content of the media. Cell death could be prevented by including either the non-competitive NMDA receptor antagonist ketamine or the competitive antagonist, d,l-AP-5. Cells expressing NR1a/2A receptors were maximally protected by 0.5 mM d,l-AP-5, while those expressing NR1a/2B receptors required 2 mM d,l-AP-5 for maximal protection. The neurosteroid pregnanolone sulfate, which negatively modulates NMDA receptor function, partially protected fibroblasts containing NR1a/2A or NR1a/2B NMDA receptor constructs. However, the neurosteroid pregnenolone sulfate, which has been reported to act as a positive allosteric modulator of the NMDA receptor, had no effect on the toxicity caused by endogenous glutamate. Our results on cells expressing human NMDA receptors suggest that certain neurosteriods may protect against NMDA induced toxicity while having low neurotoxic liabilities of their own.
Similar content being viewed by others
References
Anegawa, N.J., Lynch, D.R., Verdoorn, T.A. and Pritchett, D.D. (1995) “Transfection of N-methyl-d-aspartate receptors in a nonneuronal cell line leads to cell death” J. Neurochem. 64, 2004–2012.
Boeckman, F.A. and Aizenman, E. (1996) “Pharmacological properties of acquired excitotoxicity in Chinese hamster ovary cells transfected with N-methyl-d-aspartate receptor subunits”,J. Pharmacol. Exp. Ther. 279, 515–523.
Bowlby, M.R. (1993) “Pregnenolone sulfate potentiation of N-methyl-d-aspartate receptor channels in hippocampal neurons” Mol. Pharm. 43, 813–819.
Bruno, V., Battaglia, G., Copani, A., Giffard, R.G., Raciti, G., Raffaele, R., Shinozaki, H. and Nicoletti, F. (1995) “Activation of class II or class III metabotropic glutamate receptors protects cultured cortical neurons against excitotoxic degeneration” Eur. J. Neurosci. 7, 1906–1913.
Cascio, C., Guarneri, R., Russo, D., De Leo, G., Guarneri, M., Piccoli, F. and Guarneri, P. (2000) “Pregnenolone sulfate, a naturally occurring excitotoxin involved in delayed retinal cell death” J. Neurochem. 74, 2380–2391.
Connoly, D.T., Knight, M.D., Harakas, N.K., Wittwer, A.J. and Feder, J. (1986) “Determination of the number of epithelial cells in culture using an acid phosphatase assay”, Anal. Biochem. 152, 136–140.
Corpechot, C., Robel, P., Axelson, M., Sjovall, J. and Baulieu, E.E. (1981) “Characterization and measurement of dehydroepiandrosterone sulfate in rat brain”, Proc. Natl Acad. Sci. USA 78, 135–144.
Durand, G.M., Bennett, M.V.L. and Zurkin, R.S. (1993) “Splice variants of the N-methyl-d-aspartate receptor NR1 identify domains involved in regulation by polyamines and protein kinase C”, proc. Natl Acad. Sci. USA 90, 6731–6735.
Frandsen, A., Drejer, J. and Schousboe, A. (1989) “Direct evidence that excitotoxicity in cultured neurons is mediated via NMDA as well as non-NMDA receptors”, J. Neurochem. 55, 297–299.
Garcia-Gallo, M., Behrus, M.M., Renart, J. and Diaz-Guerra, M. (1999) “Expression of N-methyl-d-aspartate receptors using vaccinia virus causes excitotoxic death in human kidney cells”, J. Cell. Biochem. 72, 135–144.
Grimwood, S., Le Bourdelles, B., Atack, J.R., Barton, C., Cockett, W., Cook, S.M., Gilbert, E., Hutson, P.H., McKernan, R.M., Myers, J., Ragan, C.I., Wingrove, P.B. and Whiting, P.J. (1996) “Generation and characterization of stable cell lines expressing recombinant human NMDA receptor subtypes”, J. Neurochem. 66, 2239–2247.
Guarneri, P., Russo, D., Cascio, C., De Leo G., Piccoli, T., Sciuto, V., Piccoli, F. and Guarneri, R. (1989) “Pregnenolone sulfate modulates NMDA receptors, inducing and potentiating acute excitotoxicity in isolated retina”, J. Neurosci. Res. 54, 787–797.
Hollman, M., Boulter, J., Maron, C., Beasely, L., Sullivan, J., Pecht, G. and Heinemann, S. (1993) “Zinc potentiates agonist induced currents at certain splice variants of the NMDA receptor”, Neuron 10, 943–954.
Ishii, T., Moriyoshi, K., Sugihara, H., Sakurada, K., Kadotania, H., Yokoi, M., Akazawa, C., Shigemoto, R., Mizuno, N., Masu, M. and Nakanishi, S. (1993) “Molecular characterization of the family of the NMDA receptor subunits”, J. Biol. Chem. 268, 2836–2843.
Jhamandas, K., Boegman, R.J., Beninger, R.J., Miranda, A.F. and Lipic, K.A. (2000) “Excitotoxicity of quinolinic acid: modulation by endogenous antagonists”, Neurotox. Res. 2, 139–155.
Kashiwagi, K., Pahk, A.J., Masuko, T., Igarashi, K. and Williams, K. (1997) “Block and modulation of N-methyl-d-aspartate receptors by polyamines and protons: role of amino acid residues in the transmembrane and pore-forming regions of NR1 and NR2 subunits”, Mol. Pharmacol. 52, 701–713.
Koh, J.Y., Goldberg, M.P., Hartley, D.M. and Choi, D.W. (1990) “Non-NMDA receptor-mediated neurotoxicity in cortical culture”, J. Neurosci. 10, 693–705.
Le Bourdellès, B., Waffard, K.A., Kemp, G., Marshall, G., Bain, C., Wilcox, A.S., Sikela, J.M. and Whiting, P.J. (1994) “Cloning, functional co-expression and pharmacological characterisation of human cDNAs encoding NMDA receptor NR1a and NR2A subunits”, J. Neurochem. 62, 2091–2098.
Moryoshi, K., Masu, M., Ishii, T., Shigemoto, R., Mizuno, N. and Nakanishi, S. (1991) “Molecular cloning and characterization of the rat NMDA receptor”, Nature 354, 31–37.
Olney, J.W. (1969) “Glutamate-induced retinal degeneration in neonatal mice. Electron microscopy of the acutely evolving lesion”, J. Neuropathol. Exp. Neurol. 28, 455–474.
Park-Chung, M., Wu, F.-S. and Farb, D.H. (1994) “3″-hydroxy-5β-pregnan-20-one sulfate: a negative modulator of the NMDA-induced current in cultured neurons”, Mol. Pharm. 46, 146–150.
Park-Chung, M., Wu, F.-S., Purdy, R., Malayev, A., Gibbs, T.T. and Farb, D.H. (1997) “Distinct sites for inverse modulation of NMDA receptors by sulfated steroids”, Mol. Pharm. 52, 1113–1123.
Pawley, A.C., Flesher, S., Boegman, R.J., Beninger, R.J. and Jhamandas, K.J. (1996) “Differential action of NMDA antagonists on cholinergic neurotoxicity produced by injection of N-methyl-d-aspartate and quinolinic acid”, Br. J. Pharmacol. 117, 1059–1064.
Priestley, T., Laughton, P., Myers, J., Le Bourdellès, B., Kerby, J. and Whiting, P.J. (1995) “Pharmacological properties of recombinant human N-methyl-d-aspartate receptors comprising NR1a/2A and NR1a/2B subunit assemblies expressed in permanently transfected mouse fibroblast cells”, Mol. Pharm. 48, 841–848.
Raymond, L.A., Moshaver, A., Tingley, W.G., Shalaby, I. and Huganir, R.L. (1996) “Glutamate receptor ion channel properties predict vulnerability to cytotoxicity in a transfected nonneuronal cell line”, Mol. Cell. Neurosci. 7, 102–115.
Schinder, A.F., Olson, E.C., Spitzer, N.C. and Montal, M. (1996) “Mitochondrial dysfunction is a primary event in glutamate neurotoxicity”, J. Neurosci. 16, 6125–6133.
Tymianski, M., Charlton, M.P., Carlen, P.L. and Tator, C.H. (1994) “Properties of neuroprotective cell-permeant [Ca2+]i and glutamate neurotoxicity in vitro”, J. Neurophys. 72, 1973–1983.
Varney, M.A., Jachec, C., Deal, C., Hess, S.D., Dagget, L.P., Skvoretz, R., Urcan, M., Morrison, J.H., Moran, T., Johnson, E.C. and Veliçelebi, G. (1996) “Stable expression and characterization of recombinant human N-methyl-d-aspartate receptor subtypes NMDAR1a/2A and NMDAR1a/2B in mammalian cells”, J. Pharmacol. Exp. Ther. 279, 367–378.
Vrooman, L., Jhamandas, K., Boegman, R.J. and Beninger, R.J. (1993) “Picolinic acid modulates kainic acid-evoked glutamate release from the striatum in vitro”, Brain Res. 12, 193–198.
Yaghoubi, N., Malayev, A., Russek, S.J., Gibbs, T.T. and Farb, D.H. (1998) “Neurosteroid modulation of recombinant ionotropic glutamate receptors”, Brain Res. 803, 153–160.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scott, M., Tanguay, J.J., Beninger, R.J. et al. Neurosteroids and glutamate toxicity in fibroblasts expressing human NMDA receptors. neurotox res 4, 183–190 (2002). https://doi.org/10.1080/10298420290033223
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1080/10298420290033223