Summary
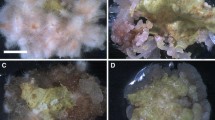
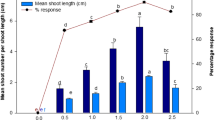
An efficient in vitro propagation system was developed for Arnebia euchroma, an important Chinese traditional medicinal plant. The present study utilized thidiazuron (TDZ) for the induction of shoot organogenesis on cotyledon and hypocotyl explants. The maximal number of shoots was obtained on the modified Linsmaier and Skoog (LS) medium supplemented with 1.0 mgl−1 (4.5 μM) TDZ for 12d on cotyledon explants (8.6 shoots per cotyledon explant). Other cytokinins (kinetin and 6-benzyladenine) and auxin (α-naphthaleneacetic acid) were not efficient in inducing regeneration on cotyledon explants. Browning of the basal portion of the subcultured shoots could be significantly reduced when they were cultured on the modified LS medium supplemented with 100 mgl−1 (33.3 μM) polyvinylpyrrolidone. Well-developed shoots formed roots on the same medium containing 1.0 mgl−1 (4.9 μM) indole-3-butyric acid. The efficient regeneration protocol reported here provides an important means of micropropagation of this plant. Furthermore, this protocol is essential to future genetic improvement of plants via transformation protocols.
Similar content being viewed by others
References
Andrade, L. B.; Echeverrigaray, S.; Fracaro, F.; Pauletti, G. F.; Rota, L. The effect of growth regulators on shoot propagation and rooting of common lavender (Lavandula vera DC). Plant Cell Tiss. Organ Cult. 56:79–83; 1999.
Chen, X.; Oppenheim, J.; Zack Howard, O. M. Shikonin: a component of anti-inflammatory Chinese herbal medicine, selectively blocks chemokine binding to CC chemokinereceptor-1. Int. J. Immunopharmacol. 1:229–236; 2001.
Du, J. Z.; Wang, J. X.; Sun, Y.; Cheng, G. E.; Zhao, Z. G. Factors to browning reaction of external implant during tissue culture in Brassica. J. Shanxi Agric. Sci. 32:29–32; 2004.
Fukui, H.; Hasan, A. F. M. F.; Kyo, M. Formation and secretion of a unique quinone by hairy root cultures of Lithospernum erythrohizon. Phytochemistry 51:511–515; 1999.
Ge, F.; Wang, X. D.; Wang, Y. C. Advances in studies on medicinal Radix Arnebiae Seu Lithospermi. Chin. Trad. Herbal Drugs 34:7–10; 2003.
Harter, H. L. Critical values for Duncan’s Multiple Range test. Biometric 16:671–685; 1960.
Hutchinson, M. J.; Murch, S. J.; Saxena, P. K. Morphoregulatory role of thidiazuron: evidence of the involvement of endogenous auxin in thidiazuron-induced somatic embryogenesis of geranium (Pelargonium x hortorum Bailey). J. Plant Physiol. 149:573–579; 1996.
Ji, Q. L.; Wang, W. G. Asexual propagation of Arnebla euchroma Johnston and exploration of hereditary stability in regenerated plantlets. Plant Physiol. Commun. 37:499–502; 2001.
Jiang, Y. L.; Song, J. D. Apoptotic induction of Xinjiang shikonin on human colorectal cancer CCL229 cell. Chin. J. Cancer 20:1355–1358; 2001.
Kadota, M.; Niimi, Y. Effects of cytokinin types and their concentrations on shoot proliferation and hyperhydricity in in vitro pear cultivar shoots. Plant Cell. Tiss. Organ. Cult. 72:261–265; 2003.
Li, Z. L.; Yang, S. Y. Reduction of abscisic acid and induction of sprouting in potato Solanum tuberosum L. by TDZ. J. Plant Growth Regul. 7:37–44; 1988.
Linsmaier, E. M.; Skoog, F. Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 18:100–127; 1965.
Liu, C. Z.; Murch, S. J.; El-Demerdash, M.; Saxena, P. K. Regeneration of the Egyptian medicinal plant Artemisia judaica L. Plant Cell Rep. 21:525–530; 2003.
Mizukami, H.; Konoshima, M.; Tabata, M. Effect of nutritional factors on shikonin derivative formation in Lithospermum erythrorhizon callus cultures. Phytochemistry 16:1183–1186; 1977.
Mok, M. C.; Mok, D. W. S.; Armstrong, D. J.; Shudo, K.; Isogai, Y.; Okamoto, T. Cytokinin activity of N-phenyl-N′-1,2,3-thidiazol-5-yl urea (thidiazuron). Phytochemistry 21:1509–1511; 1982.
Murch, S. J.; Choffe, K. L.; Victor, J. M. R.; Slimmon, T. Y. KrishnaRaj, S.; Saxena, P. K. Thidiazuron-induced plant regeneration from hypocotyl cultures of St. John’s wort (Hypericum perforatum cv ‘Anthos’). Plant Cell Rep. 19:576–581; 2000a.
Murch, S. J.; KrishnaRaj, S.; Saxena, P. K. Phytomaceuticals: mass production, standardization, and conservation. Sci. Rev. Alternative Med. 4:39–43; 2000b.
Murch, S. J.; Saxena, P. K. Modulation of mineral and fatty acid profiles during thidiazuron mediated somatic embryogenesis in peanuts (Arachis hypogeae L.). J. Plant Physiol. 151:183–191; 1997.
Murch, S. J.; Saxena, P. K. Molecular fate of thidiazuron and its effects on auxin transport in hypocotyl tissues of Pelargonium x hortorum Bailey. Plant Growth Regul. 35:269–275; 2001.
Murthy, B. N. S.; Murch, S. J.; Saxena, P. K. TDZ-induced somatic embryogenesis in intact seedlings of peanut (Arachis hypogaea): endogenous growth regulator levels and significance of cotyledons. Physiol. Plant. 94:268–276; 1995.
Murthy, B. N. S.; Murch, S. J.; Saxena, P. K. Thidiazuron: a potent regulator of in vitro plant morphogenesis. In Vitro Cell. Dev. Biol.-Plant 34:267–275; 1998.
Murthy, B. N. S.; Victor, J.; Singh, R. P.; Fletcher, R. A.; Saxena, R. K. In vitro regeneration of Chickpea (Cicer arientinum L.): stimulation of direct organogenesis and somatic embryogenesis by thidiazuron. Plant Growth Regul. 19:233–240; 1996.
Nandi, S. K.; Letham, D. S.; Palni, L. M. S.; Wong, O. C.; Summons, R. E. 6-Benzylaminopurine and its glycosides as naturally occurring cytokinins. Plant Sci. 61:189–196; 1989.
Singh, N. D.; Sahoo, L.; Sarin, N. B.; Jaiwal, P. K. The effect of TDZ on organogenesis and somatic embryogenesis in pigeonpea (Cajanus cajan L. Millsop). Plant Sci. 164:341–347; 2003.
Steward, F. C.; Mapes, M. O.; Kent, A. E.; Holsten, R. D. Growth and development of cultured plant cells. Science 143:20–27; 1964.
Tabata, M.; Fujita, Y. Production of shikonin by plant cell cultures. In: Zaitlin, M., ed. Biotechnology in Plant Science. New York: Academic Press; 1985:207–218.
Tabata, M.; Misukami, H.; Hiraka, N. Pigment formation in callus culture of Lithospermum erythrorhizon. Phytochemistry 13:927–932; 1974.
Tepper, H. B.; Mante, S. The mature dicot cotyledon as an organogenic structure. Phytomorphology 40:163–168; 1990.
Ye, H. C.; Yin, Z. H.; Li, G. F.; Wu, X.; Dong, J. W.; Wu, Z. R. Effects of physical and chemical factors on callus growth and shikonin derivative formation in the callus cultures of Arnebia euchroma. Acta. Bot. Sin. 33:927–931; 1991.
Yip, W. K.; Yang, S. F. Effect of thidiazuron in cytokinin dependent ethylene production system. Plant Physiol. 80:515–519; 1986.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, B., Yang, YG., Guo, YM. et al. Thidiazuron-induced in vitro shoot organogenesis of the medicinal plant Arnebia euchroma (Royle) Johnst. In Vitro Cell.Dev.Biol.-Plant 41, 677–681 (2005). https://doi.org/10.1079/IVP2005650
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2005650