Summary
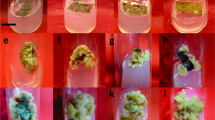
An effcient regeneration protocol has been developed from leaf explants of Pluchea lanceolata, a medicinally important plant of the family Asteracea. Nodular callus was initiated from young leaf segments cultured on Wood and Braun medium containing 2.0% sucrose (WB) supplemented with kinetin. On WB+5.0 mg l−1 kinetin, 100% plant regeneration with 14±0.5 plantlets was obtained. Regenerated plantlets with well-developed root systems were transferred to pots and subsequently to the field. With respect to the effect of metals on morphogenic events and quercetin content of the cultures, lower concentrations of CuSO4 proved to be beneficial but higher concentrations were detrimental. The presence of 200 μM ZnSO4 or 150 μM CuSO4 also resulted in the highest level of quercetin in regenerants. The yield of quercetin in culture varied with stages of differentiation.
Similar content being viewed by others
References
Ali, G.; Purohit, M.; Saba; Iqbal, M.; Srivastava, P. S. Morphogenic response and isozymes of Bacopa monniera L. Wettst cultures grown under salt stress. Phytomorphology 47:97–106; 1997.
Ali, G.; Srivastava, P. S.; Iqhal, M. Morphogenic response and proline content in Bacopa monniera cultures grown under copper stress. Plant Sci. 138:191–195; 1998.
Anonymous. The wealth of India. Raw materials, vol. VIII. New Delhi: Council of Scientific and Industrial Research; 1998.
Asami, T.; Kuboto, M.; Orikasa, K. Distribution of different fractions of cadmium, zinc, lead and copper in unpolluted and polluted soil. Soil Water Air Pollut. 83:187–194; 1995.
Bates, L. K.; Waldren, R. P.; Teare, J. D. Rapid determination of free proline for water stress studies. Plant Soil 39:205–207; 1973.
Bhattacharya, R.; Bhattacharya, S. High frequency in vitro propagation of Phyllanthus amarus Schum. & Thom. by shoot tip culture. Ind. J. Exp. Biol. 39:1184–1187; 2001.
Borthakur, M.; Dutta, K.; Nath, S. C.; Singh, R. S. Micropropagation of Eclipta alba and Eupatorium adenophorum using a single-step nodal cutting technique. Plant Cell Tiss. Organ Cult. 62:239–242; 2000.
Bradford, M. M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254; 1976.
Chen, C. T.; Kao, C. H. Osmotic stress and water stress, have opposite effects on putrescine and proline production in excised rice leaves. Plant Growth Regul 13:197–202; 1993.
Datta, A.; Srivastava, P. S. Variation in vinblastin production by Catharanthus roscus during in vivo and in vitro differentiation. Phytochemistry 46:135–137; 1997.
Davies, K. L.; Davies, M. S.; Francis, D. Zinc induced vacuolation in root meristematic cells of Festuca rubra L. Plant Cell Environ. 14:399–406; 1991.
Delauney, A. J.; Verma, D. P. S. Proline biosynthesis and osmoregulation in plants. Plant J. 4:215–223; 1993.
Fernandes, J. C.; Henriques, F. S. Biochemical, physiological and structural effects of excess copper in plants. Bot. Rev. 57:246–276; 1991.
Garcia-Sago, B.; Roig, L. A.; Moreno, V. Enhancement of morphogenetic response in cotyledon-derived explants of Cucumis melo induced by copper ions. Acta Hort. 289:229–230; 1991.
Godbold, D. L.; Horst, W. J.; Marschner, H.; Collins, J. C.; Thurman, D. A. Root growth and Zn uptake by two ecotypes of Deschampsia caespitosa as affected by high Zn concentrations. Z. Pflanzenphysiol. 112:315–324; 1983.
Grieve, T. M.; Gartland, K. M. A.; Elliott, M. C. Micropropagation of commercially important sugarbeet cultivars. Plant Growth Regul. 21:15–18; 1997.
Macnair, M. R. The genetics of metal tolerance in vascular plants. New Phytol. 124:641–559; 1992.
Marschner, H. Nutrition of higher plants. New York: Academic Press; 1986.
Mengel, K.; Kirby, E. A. Principles of plant nutrition, 4th edn. Bern: International Potash Institute; 1987:385–402.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Ouzounidou, G. Copper induced changes on growth, metal content and photosynthetic function of Alyssum montanum L. plants. Environ. Exp. Bot. 34:165–172; 1994.
Purohit, M.; Pande, D.; Datta, A.; Srivastava, P. S. Enhanced xanthotoxin content in regenerating cultures of Ammi majus and micropropagation. Planta Med. 61:481–482; 1995a.
Purohit, M.; Pande, D.; Datta, A.; Srivastava, P. S. In vitro flowering and high xanthotoxin in Ammi majus L. J. Plant Biochem. Biotechnol. 4:73–76; 1995b.
Saba; Pande, D.; Iqbal, M.; Srivastava, P. S. Effect of zinc sulphate and copper sulphate on regeneration and lepidine content in Lepidium sativum, L Biol. Plant. 43:1–4; 1999.
Saunders, J. W.; Doley, W. P. One step shoot regeneration form callus of whole plant leaf explants of sugarbeet lines and somaclonal variation of in vitro culture behaviour. J. Plant Physiol. 124:473–479; 1986.
Srivastava, P. S.; Pande, D. In vitro propagation and conservation of medicinal plants. In: Srivastava, P. S., ed. Plant tissue culture and molecular biology. New Delhi: Narosa Publishing House; 1998:254–281.
Srivastava, P. S.; Purohit, M.; Pande., D.; Dutta, A. Phenotypic variation and alkaloid content in the androgenic plantlets of Datura innoxia. Phytomorphology 13:209–216; 1993.
Srivastava, V.; Verma, N.; Tandon, J. S.; Srimal, R. C. Anti-inflammatory activity of Pluchea lanccolata: isolation of an active principle. Int. J. Crude Drug Res. 28:135–137; 1990.
Tao, R.; Murayama, H.; Moriguchi, K.; Sugiura, A. Plant regeneration from callus cultures derived from primordial leaves of adult Japanese persimmon. HortScience 23:1055–1056; 1998.
Tao, R.; Sugiura, A. Adventitious bud formation from callus cultures of Japanese persimmon. HortScience 27:259–261; 1992.
Tetu, T.; Sangwan, R. S.; Sangwan-Norreel, B. S. Hormonal control of organogenesis and somatic embryogenesis in Beta vulgaris callus. J. Exp. Bot. 38:506–517; 1987.
Tiwati, V.; Tiwari, K. N.; Singh, B. D. Comparative studies of cytokinins on in vitro propagation of Bacopa monnierd. Plant Cell Tiss. Organ Cult. 66:9–16; 2001.
Watad, A. A.; Kochba, M.; Nissima., A.; Gaba, V. Improvement of Aconitum napellus micropropagation by liquid culture on floating membrane raft. Plant Cell Rep. 40:345; 1995.
Wood, H. N.; Braun, A. C. Studies on the regulation of certain essential biosynthetic systems in normal and crown gall tumour Proc. Natl Acad. Sci. USA 147:1507; 1961.
Zhong, Z.; Smith, H. G.; Thomas, T. H. In vitro culture of petioles and intact leaves of sugar beet (Beta vulgaris) Plant Growth Regul. 12:59–66; 1993.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, S., Narula, A., Sharma, M.P. et al. In vitro propagation of Pluchea lanceolata, a medicinal plant, and effect of heavy metals and different aminopurines on quercetin content. In Vitro Cell.Dev.Biol.-Plant 40, 171–176 (2004). https://doi.org/10.1079/IVP2003490
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2003490