Summary
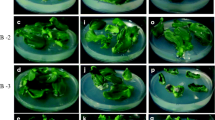
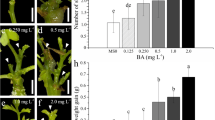
Guar (Cyamopsis tetragonoloba L. Taub) is a drought-tolerant multipurpose cash crop. A rapid regeneration system has been developed for four Indian guar genotypes. Investigations were carried out to assess the effect of different growth regulators and their combinations on a variety of explants such as the embryo, cotyledons, and cotyledonary nodes for shoot morphogenesis. It was established that Murashige and Skoog's culture medium containing 6-benzylaminopurine (13.3 μM or 3 mgl−1) in combination with indole-3-acetic acid (11.4 μM or 2mgl−1) with cotyledonary node explants gave the highest frequency of multiple shoot induction. In vitro rooting from cultured shoots was maximal on a half-salt concentration of Murashige and Skoog's culture medium fortified with indole-3-butyric acid (4.9 μM or 1 mgl−1). In vitro-regenerated plants were grown to pod setting and subsequent maturity in greenhouse conditions.
Similar content being viewed by others
References
Bansal, Y. K.; Chibbar, T.; Bansal, S.; Singh, M. Plant regeneration from hypocotyl and cotyledon explants of cluster bean (Cyamopsis tetragonoloba L.). J. Physiol. Res. 7:57–60; 1994.
Gamborg, O. L.; Miller, R. A.; Ojima, K. Nutritional requirements of suspension cultures of soybean root cells. Exp. Cell. Res. 50:151–158; 1968.
Gulati, A.; Jaiwal, P. K. Plant regeneration from cotyledonary node explants of mung bean (Vigna radiata L. Wilczek). Plant Cell Rep. 13:523–527; 1994.
Hoagland, D.R.; Arnon, D. I. The water-culture method for growing plants without soil. Univ. Calif. Coll. Agric. Exp. Sta. Berkeley, Circ. 347:1–39; 1950.
Joersbo, M.; Brunstedt, J.; Marcussen, J.; Okkels, F. T. Transformation of the endospermous legume guar (Cyamopsis tetragonoloba L.) and analysis of transgene transmission. Mol. Breeding 5:521–529; 1999.
Joersbo, M.; Marcussen, J.; Brunstedt, J. In vivo modification of the cell wall polysaccharide galactomannan of guar transformed with a α-galactosidase gene cloned from senna. Mol. Breeding 7:211–219; 2001.
McClean, P.; Grafton, K. F. Regeneration of dry bean (Phaseolus vulgaris L.) via organogenesis. Plant Sci. 60:117–122; 1989.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Prakash, N. S.; Pental, D.; Sarin, N. B.; Regeneration of pigeon pea (Caianus cajan) from cotyledonary node via multiple shoot formation. Plant Cell Rep. 13:623–627; 1994.
Ramulu, C. A.; Rao, D. Tissue culture studies of differentiation in a grain legume Cyamopsis tetragonoloba (L.) Taub. J. Physiol. Res. 4:183–185; 1991.
Ramulu, C. A.; Rao, D. In vitro effect of phytohormones on tissue cultures of cluster bean (Cyamopsis tetragonoloba L. Taub). J. Physiol. Res. 20:7–9; 1993a.
Ramulu, C. A.; Rao, D. Induced pesticide resistant lines in cluster bean through tissue culture. J. Physiol. Res. 6:71–72; 1993b.
Sainy, M. L.; Paroda, R. S. Guar cultivation in Haryana, vol. 1. Hisar, Haryana, India: Department of Plant Breeding, Chaudhary Charan Singh Haryana Agricultural University; 1984:1–24.
Saxena, P. K.; Gill, R. Removal of browning and growth enhancement by polyvinylpolypyrrolidone in protoplast cultures of Cyamopsis tetragonoloba. Biol. Plant. 28:313–315; 1986.
Saxena, P. K.; Gill, R.; Rashid, A. Isolation and culture of protoplasts from mesophyll tissue of legume Cyamopsis tetragonoloba (L.). Plant Cell Tiss. Organ Cult. 6:173–176; 1986.
Subhadra, S.; Vashisht, R. K.; Chawdhury, J. B.; Singh, M.; Sareen, P. K. Multiple shoots from the cotyledonary node explants of non-nodulating genotype (ICC 435M) of chick pea, Cicer arietinum L. Int. J. Exp. Biol. 36:1276–1278; 1998.
Whistler, R. L.; Hymowitz, T. Guar: agronomy, production, industrial use and nutrition. West Lafayette, IN: Purdue University Press; 1979.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prem, D., Singh, S., Gupta, P.P. et al. High-frequency multiple shoot regeneration from cotyledonary nodes of guar (Cyamopsis tetragonoloba L. Taub). In Vitro Cell.Dev.Biol.-Plant 39, 384–387 (2003). https://doi.org/10.1079/IVP2003425
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2003425