Summary
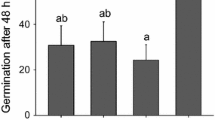
The hormone-stimulated and growth-related cell surface hydroquinone (NADH) oxidase activity of etiolated hypocotyls of soybeans oscillates with a period of about 24 min or 60 times per 24-h day. Plasma membranes of soybean hypocotyls contain two such NADH oxidase activities that have been resolved by purification on concanavalin A columns. One in the apparent molecular weight range of 14–17 kDa is stimulated by the auxin herbicide 2,4-dichlorophenoxyacetic acid (2,4-D). The other is larger and unaffected by 2,4-D. The 2,4-D-stimulated activity absolutely requires 2,4-D for activity and exhibits a period length of about 24 min. Also exhibiting 24-min oscillations is the rate of cell enlargement induced by the addition of 2,4-D or the natural auxin indole-3-acetic acid (IAA). Immediately following 2,4-D or IAA addition, a very complex pattern of oscillations is frequently observed. However, after several hours a dominant 24-min period emerges at the expense of the constitutive activity. A recruitment process analogous to that exhibited by prions is postulated to explain this behavior.
Similar content being viewed by others
References
Bridge, A.; Barr, R.; Morré, D. J. The plasma membrane NADH oxidase of soybean has vitamin K1 hydroquinone oxidase activity. Biochim. Biophys. Acta 1463:448–458; 2000.
Brightman, A. O.; Barr, R.; Crane, F. L.; Morré, D. J. Auxin-stimulated NADH oxidase purified from plasma membrane of soybean. Plant Physiol. 86:1264–1269; 1988.
Chueh, P.-J.; Morré, D. M.; Penel, C.; DeHahn, T.; Morré, D. J. The hormone-responsive NADH oxidase of the plant plasma membrane has properties of a NADH: protein disulfide reductase. J. Biol. Chem. 272:11221–11227; 1997.
Davies, P. J. Plant hormones. Dordrecht: Kluwer Academic Publishers; 1995.
DeHahn, T.; Barr, R.; Morré, D. J. NADH oxidase activity present on both the external and internal membrane surfaces of soybean membranes. Biochim. Biophys. Acta 1328:99–108; 1997.
Griffith, J. S. Self-replication and scrapie. Nature 315:1043–1044; 1967.
Kelker, M.; Kim, C.; Chueh, P.-J.; Guimont, R.; Morré, D. M.; Morré, D. J. Cancer isoform of a tumor-associated cell surface NADH oxidase (tNOX) has properties of a prion. Biochemistry 40:7351–7354; 2001.
Kishi, T.; Morré, D. M.; Morré, D. J. The plasma membrane NADH oxidase of HeLa cells has hydroquinone oxidase activity. Biochim. Biophys. Acta 1412:66–77; 1999.
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685; 1970.
Lambeth, J. D.; Cheng, G.; Arnold, R. S.; Edens, W. A. Novel homologs of gp91phox. TIBS 25:459–461; 2000.
Lüthen, H.; Böttger, M. A high-tech low-cost auxinometer for high-resolution determination of elongation rates in six simultaneous experimental setups. Mitt. Inst. Allg. Botanik, Hamburg 24:13–22; 1992.
Morré, D. J. NADH oxidase: a multifunctional ectoprotein of the eukaryotic cell surface. In: Asard, A.; Bérczi, A.; Caubergs, R., eds. Plasma membrane redox systems and their role in biological stress and disease. Dordrecht: Kluwer Academic Publishers; 1998:121–156.
Morré, D. J.; de Cabo, R.; Jacobs, E.; Morré, D. M. Auxin-modulated protein disulfide-thiol interchange activity from soybean plasma membrane. Plant Physiol. 109:573–578; 1995.
Morré, D. J.; Gomez-Rey, M. L.; Schramke, C.; Em, O.; Lawler, J.; Hobeck, J.; Morré, D. M. Use of dipyridyl-dithio substrates to measure directly the protein disulfide-thiol interchange activity of the auxin stimulated NADH: protein disulfide reductase of soybean plasma membranes. Mol. Cell Biochem. 200:7–13; 1999a.
Morré, D. J.; Grieco, P. A. Glaucarubolone and simalikalactone D, respectively, preferentially inhibit auxin-induced and constitutive components of plant cell enlargement and the plasma membrane NADH oxidase. Int. J. Plant Sci. 160:291–297; 1999.
Morré, D. J.; Morré, D. M. NADH oxidase activity of soybean plasma membranes oscillates with a temperature compensated period of 24 min. Plant J. 16:277–284; 1998.
Morré, D. J.; Morré, D. M.; Penel, C.; Greppin, H. NADH oxidase periodicity of spinach leaves synchronized by light. Int. J. Plant Sci. 160:855–860; 1999b.
Morré, D. J., Navas, P.; Penel, C.; Castillo, F. J. Auxin-stimulated NADH oxidase (semidehydroascorbate reductase) of soybean plasma membrane: role in acidification of cytoplasm? Protoplasma 133:195–197; 1986.
Morré, D. J.; Pogue, R.; Morré, D. M. Soybean cell enlargement oscillates with a temperature-compensated period length of ca. 24 min. In Vitro Cell. Dev. Biol. Plant 37:19–23; 2001.
Morré, D. J.; Ternes, P.; Morré, D. M. Cell enlargement of plant tissue explants oscillates with a temperature-compensated period length of ca. 24 min. In Vitro Cell Dev. Biol. Plant 38:18–28; 2002.
Pogue, R.; Morré, D. M.; Morré, D. J. CHO cell enlargement oscillates with a temperature-compensated period of 24 min. Biochim. Biophys. Acta 1498:44–51; 2000.
Prusiner, S. B. Biology and genetics of prion diseases. Annu. Rev. Microbiol. 48:655–686; 1994.
Satter, R. L.; Galston, A. W. Mechanisms of the control of leaf movements. Annu. Rev. Plant Physiol. 32:83–110; 1981.
Sedlak, D.; Morré, D. M.; Morré, D. J. A drug-unresponsive and protease-resistant CNXO protein from human sera. Arch. Biochem. Biophys. 386:106–116; 2001.
Smith, P. K.; Krohn, R. I.; Hermanson, G. T.; Mallia, A. K.; Gartner, F. H.; Provenzano, E. K.; Fujimoto, E. K.; Goeke, N. M.; Olson, B. J.; Klenk, D. C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 150:76–85; 1985.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morré, D.J., Morré, D.M. & Ternes, P. Auxin-activated nadh oxidase activity of soybean plasma membranes is distinct from the constitutive plasma membrane NADH oxidase and exhibits prion-like properties. In Vitro Cell.Dev.Biol.-Plant 39, 368–376 (2003). https://doi.org/10.1079/IVP2003417
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2003417