Abstract
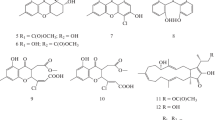
Phytotoxic components detected by a bean (Vicia faba) leaf assay were isolated from extracts of dried mycelia and liquid culture filtrates of Drechslern wirreganensis and D. campanulata by preparative thin layer chromatography. Mass spectrometric examination of chromatographically homogeneous phytotoxic fractions was consistent with the presence of cytochalasin B and dihydrocytochalasins in both fungi. Cytochalasin B was present at 10.3 g/kg dry weight of the mycelium for D. wirreganensis and 7.7 g/kg for D. campanulata. In addition, differences were observed in the minor cytochalasin components. In D. wirreganensis, a more hydrophobic cytochalasin derivative with an ion at m/z 675 was present. In D. campanulata a component of similar hydrophobicity was present, but with an ion at 481 m/z. Measurable inhibition of wheat seed germination by cytochalasin B was observed only at or above 70 μм in 1% dimethyl sulfoxide solution. The formation of bi-nucleate cells, a common feature in mammalian cells treated with cytochalasin B, is reported in barley root tips. Differences in lesion morphology caused by these two related pathogenic fungi are also associated with differences in types and amounts of the phytotoxic components they produce.
Similar content being viewed by others
References
Aldridge DC, Armstrong JJ, Speake RN, Turner WB (1967) The cytochalasins, a new class of biologically active mould metabolites. Chemical Communications 1, 26–27.
Betina V, Micekova D, Nemec P (1972) Antimicrobial properties of cytochalasins and their alteration of fungal morphology. Journal of General Microbiology 71, 343–349.
Bottalico A, Capasso R, Evidente A, Logreico A, Randazzo G, Vurro M (1989) Production of cytochalasins by isolates of Ascochyta heteromorpha from oleander and negative evidence for their role in leaf spot disease. In ‘Phytotoxins and plant pathogenesis’. (Eds A Graniti, RD Durbin) (Springer-Verlag: Berlin Heidelberg)
Botallico A, Capasso R, Evidente A, Randazzo G, Vurro M (1990) Cytochalasins structure—activity relationships. Phytochemistry 29, 93–96.
Campbell MA, Medd RW, Brown JF (1996) Cultural and infection studies on Pyrenophora semeniperda, a possible bioherbicide for annual grass-weeds. In ‘Proceedings of the 9th international symposium on biological control of weeds’. Stellenbosch, South Africa. (Eds VC Moran, JH Hoffmann) pp. 519–523. (University of Cape Town)
Capasso R, Evidente A, Retiene A (1988) Aschochalasin, a new cytochalasin from Aschochyta heteromorpha. Journal of Natural Products 51, 567–571.
Carter SB (1967) Effects of cytochalasins on mammalian cells. Nature 213, 261–264.
Copeland M (1973) The cellular response to cytochalasin B: a critical overview. Cytologia 39, 709–727.
Evidente A, Andolfi A, Vurro M, Zonno MC, Motta A (2002) Cytochalasins ZI, Z2 and Z3, three 24-oxa[14]cytochalasans produced by Pyrenophora semeniperda. Phytochemistry 60, 45–53. doi:10.1016/S0031-9422(02)00071-7
Herth W, Franke WW, Vanderwoude WJ (1972) Cytochalasin stops tip growth in plants. Die Naturwissenschaften 59, 38–39.
Kohmoto K (Ed.) (1992) ‘Determination of host selective toxins. Modern methods of plant analysis:plant toxin analysis.’ (Springer-Verlag: Germany)
Krishan A, Ray-Chaudhuri R (1969) Asynchrony of nuclear development in cytochalasin-induced multinucleate cells. The Journal of Cell Biology 43, 618–621.
Lin S, Spudich JA (1974) Biochemical studies on the mode of action of cytochalasin B. The Journal of Biological Chemistry 249, 5778–5783.
Palevitz BA (1980) Comparative effects of phalloidin and cytochalasin B on motility and morphogenesis in Allium. Canadian Journal of Botany 58, 773–785.
Paterson RRM, Bridge PD (1994) “Biochemical techniques for filamentous fungi. IMI Technical Handbooks: No. 1.’ (International Mycological Institute: UK)
Pope DG, Thorpe JR, Al-Azzawi MJ, Hall JL (1979) The effect of cytochalasin B on the rate of growth and ultrastructure of wheat coleoptiles and maize roots. Planta 144, 373–383.
Pribela A, Tomko J, Dolejs L (1975) Cytochalasin B from tomatoes contaminated by Hormisium sp. Phytochemistry 14, 285. doi: 10.1016/0031-9422(75)85057-6
Purvis MJ, Collier DC, Walls D (1964) “Laboratory techniques in botany.’ (Butterworth and Co.: London)
Ridler MAC, Smith GF (1968) The response of human cultured lymphocytes to cytochalasin B. Journal of Cell Science 3, 595–602.
Rothweiler W, Tamm C (1966) Isolation and structure of phomin. Experientia 22, 750–752.
Sawhney VK, Srivastava LM (1974) Cytochalasin-B-induced inhibition of root-hair in lettuce seedlings and its reversal by benzyladenine. Planta (Berlin) 119, 165–168.
Scott PM, Harwig J, Chen Y-K, Kennedy BPC (1975) Cytochalasins A and B from strains of Phoma exigua var. exigua and formation of cytochalasin Bin potato gangrene. Journal of General Microbiology 87, 177–180.
Tamm C (1978) Ch2. Chemistry and biosynthesis of cytochalasans. In ‘Cytochalasins — biochemical and cell biological aspects’. (Ed. SW Tanenbaum) pp. 15–51. (Elsevier/North Holland Biomedical Press: Amsterdam)
Thilly WG, Liber HL, Woogan GN (1978) Toxicity and structure-activity relationships of cytochalasins. In ‘Cytochalasins — biochemical and cell biological aspects’. (Ed. SW Tanenbaum) pp. 53–63. (Elsevier/North Holland Biomedical Press: Amsterdam)
Thomas DDS (1978) Ch 11. Cytochalasin effects in plants and eukaryotic microbial systems. In ‘Frontiers in biology’. (Ed. SW Tanenbaum) pp. 258–275. (Elsevier/North Holland Biomedical Press: Amsterdam)
Thomas DDS, Lager NM, Manavathu EK (1973) Cytochalasin B: effects on root morphogenesis in Allium cepa. Canadian Journal of Botany 51, 2269–2273.
Van Der Westhuizen GCA, Marasas WFO, Schneider DJ (1985) Drechslera campanulata rediscovered on oats in South Africa. Phytophylactica 17, 103–106.
Wallace HAH (1959) A rare seed-borne disease of wheat caused by Podosporietla verticillata. Canadian Journal of Botany 37, 509–515.
Wallwork H, Lichon A, Sivanesan A (1992) Drechslera wirreganensis — a new Hyphomycete affecting barley in Australia. Mycological Research 96, 886–888.
Wallwork H, Potter TD, Lichon A (1995) Occurrence of Wirrega blotch in barley and other grass species in Australia. Australasian Plant Pathology 24, 22–25.
Williamson RE (1975) Cytoplasmic streaming in Chara: a cell model activated ATP and inhibited by Cytochalasin B. Journal of Cell Science 17, 655–668.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Capio, E.R., Tate, M.E. & Wallwork, H. Phytotoxic metabolites from Drechslern wirreganensis and D. campanulata . Australasian Plant Pathology 33, 23–28 (2004). https://doi.org/10.1071/AP03069
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1071/AP03069