Abstract
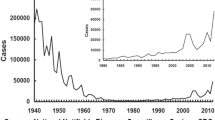
Pertussis is the only vaccine-preventable disease that has re-emerged in Israel. In the last two decades, despite high primary immunization coverage, crude incidence increased over tenfold, with especially high morbidity among infants and adolescents and with 19 infant deaths. Two pertussis vaccine boosters were added, in 2005 for 7-year-olds and in 2011 for 13-year-olds. We reviewed age group incidence from 1999 to 2016, before and after the booster program introduction. We compared three groups of 13–15 year-olds with identical primary immunization but different booster immunization histories. Vaccine effectiveness was calculated before and after adjustment for specific incidence in those aged 65 and over. Two years after one booster, adjusted vaccine effectiveness was 74.5%. Two years after two boosters, adjusted vaccine effectiveness was 91.8%. However, crude morbidity rates were not reduced. The booster program has been effective only among recipient groups. The program will be continued. Israel is now encouraging pregnant women to be vaccinated against pertussis to improve protection of infants.


Similar content being viewed by others
References
European Centre for Disease Prevention and Control. Expert consultation on pertussis—Barcelona. http://ecdc.europa.eu/en/publications/Publications/pertussis-meeting-2012.pdf. Accessed 20 Nov 2012.
Clark TA. Changing pertussis epidemiology: everything old is new again. J Infect Dis. 2014;209:978–81. http://jid.oxfordjournals.org/content/209/7/978.full.pdf.
Moerman L, Leventhal A, Slater PE, Anis E, Yishai R, Marva E. The re-emergence of pertussis in Israel. IMAJ 2006;8:308–11. http://www.health.gov.il/PublicationsFiles/pert_2006.pdf.
Public Health England. Guidelines for the Public Health Management of Pertussis in England Produced by the Pertussis Guidelines Group. Update to the 2012 Health Protection Agency (HPA) Guidelines for the public health management of pertussis. 2016: London. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/576061/Guidelines_for_the_Public_Health_Management_of_Pertussis_in_England.pdf.
Statistical Abstract of Israel. Central Bureau of Statistics, Jerusalem, Israel. http://www.cbs.gov.il/reader/shnatonenew_site.htm.
Bisgard KM, Pascual FB, Ehresmann KR, Miller CA, Cianfrini C, Jennings CE, Rebmann CA, Gabel J, Schauer SL, Lett SM. Infant pertussis: who was the source? Pediatr Infect Dis J. 2004;23(11):985–9.
Brooks DA, Clover R. Pertussis infection in the United States: role for vaccination of adolescents and adults. JABFM. 2006;19(6). http://www.jabfm.org/content/19/6/603.full.pdf+html.
OECD. Fertility rates (interactive chart-map, comparisons). 2016. https://data.oecd.org/pop/fertility-rates.htm.
Stein-Zamir C, Shoob H, Abramson N, Zentner G. The impact of additional pertussis vaccine doses on disease incidence in children and infants. Vaccine. 2011;29:207–11.
Skoff TH, Cohn AC, Clark TA, Messonnier NE, Martin SW. Early impact of the US Tdap vaccination program on pertussis trends. Arch Pediatr Adolesc Med. 2012;166(4):344–9. http://archpedi.jamanetwork.com/article.aspx?articleid=1148395.
Lavine J, Bjornstad O, De Blasio BF, Storsaeter J. Short-lived immunity against pertussis, age-specific routes of transmission, and the utility of a teenage booster vaccine. Vaccine. 2012;30(3):544–51. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3246080/pdf/nihms340699.pdf.
Tartof SY, Lewis M, Kenyon C, White K, Osborn A, Liko J, et al. Waning immunity to pertussis following 5 doses of DTaP. Pediatrics. 2013; 131(4): e1047–52. http://pediatrics.aappublications.org/content/pediatrics/early/2013/03/06/peds.2012-1928.full.pdf.
McGirr A, Fisman DN. Duration of pertussis immunity after DTaP immunization: a meta-analysis. Pediatrics. 2015;135(2):331–43. http://pediatrics.aappublications.org/content/pediatrics/early/2015/01/01/peds.2014-1729.full.pdf.
Wendelboe AM, Van Rie A, Salmaso S, Englund JA. Duration of immunity against pertussis after natural infection or vaccination. Pediatr Infect Dis J. 2005; 24(5):S58–61. http://journals.lww.com/pidj/Fulltext/2005/05001/Duration_of_Immunity_Against_Pertussis_After.11.as.
Klein NP, Bartlett J, Rowhani-Rahbar A, Fireman B, Baxter R. Waning protection after fifth dose of acellular pertussis vaccine in children. N Engl J Med. 2012;367(11):1012–9. https://doi.org/10.1056/NEJMoa1200850.
Klein NP, Bartlett J, Fireman B, Rowhani-Rahbar A, Baxter R. Comparative effectiveness of acellular versus whole-cell pertussis vaccines in teenagers. Pediatrics. 2013;131(6):e1716–22. http://pediatrics.aappublications.org/content/early/2013/05/15/peds.2012-3836.full.pdf.
Witt MA, Arias L, Katz PH, Truong ET, Witt DJ. Reduced risk of pertussis among persons ever vaccinated with whole cell pertussis vaccine compared to recipients of acellular pertussis vaccines in a large US cohort. CID. 2013;56(9). http://cid.oxfordjournals.org/content/56/9/1248.long.
Sheridan SL, Ware RS, Grimwood K, Lambert SB. Number and order of whole cell pertussis vaccines in infancy and disease protection. JAMA. 2012;308(5):454–6. http://jama.jamanetwork.com/article.aspx?articleid=1273011.
Choi YH, Campbell H, Amirthalingam G, van Hoek AJ, Miller E. Investigating the pertussis resurgence in England and Wales, and options for future control. BMC Med. 2016;14:121. https://doi.org/10.1186/s12916-016-0665-8.
Mooi FR, Van Der Mas NAT, De Melker H. Pertussis resurgence: waning immunity and pathogen adaptation—two sides of the same coin. Epidemiol Infect. 2014;142:685–94. https://doi.org/10.1017/S0950268813000071.
Schmidtke AJ, Boney KO, Martin SW, Skoff TH, Tondella ML, Tatti KM. Population diversity among Bordetella pertussis isolates, United States, 1935–2009. Emerg Infect Dis 2012; 18:1248–5. https://wwwnc.cdc.gov/eid/article/18/8/12-0082_article.
Octavia S, Wu SZ, Kaur S, Valinsky L, Marva E, Moran-Gilad J, et al. Whole-genome sequencing and comparative genomic analysis of Bordetella. pertussis isolates from the 2007-2008 epidemic in Israel. J Infect. 2016. https://doi.org/10.1016/j.jinf.2016.11.012.
Raya BA, Bamberger E, Spiegel G, Sprecher H, Davidson S, Geffen Y, Srugo I. Two closely related strains associated with pertussis resurgence in Israel. Pediatr Infect Dis J. 2012;31:761–2.
Litt DJ, Neal SE, Fry NK. Changes in genetic diversity of the Bordetella pertussis population in the United Kingdom between 1920 and 2006 reflect vaccination coverage and emergence of a single dominant clonal type. J Clin Microbiol. 2009;47:680–688. http://jcm.asm.org/content/47/3/680.full.pdf.
Healy CM, Rench MA, Baker CJ. Importance of timing of maternal combined tetanus, diphtheria, and acellular pertussis (Tdap) immunization and protection of young infants. Clin Infect Dis. 2013;56:539–44. http://cid.oxfordjournals.org/content/56/4/539.full.pdf.
Dabrera G, Amirthalingam G, Andrews N, Campbell H, Ribeiro D, Kara E, et al. A case-control study to estimate the effectiveness of maternal pertussis vaccination in protecting newborn infants in England and Wales, 2012–2013. Clin Infect Dis. 2015;60(3)333–7. http://cid.oxfordjournals.org/content/60/3/333.full.pdf.
Baxter R, Bartlett J, Fireman B, Lewis E, Klein NP. Effectiveness of vaccination during pregnancy to prevent infant pertussis. Pediatrics. 2017;139(5):e20164091.
Donegan K, King B, Bryan P. Safety of pertussis vaccination in pregnant women in UK: observational study. BMJ. 2014;349:g4219. http://www.bmj.com/content/bmj/349/bmj.g4219.full.pdf.
Petousis-Harris H, Walls T, Watson D, et al. Safety of Tdap vaccine in pregnant women: an observational study. BMJ Open. 2016;6:e010911. https://doi.org/10.1136/bmjopen-2015-010911.
Advisory Committee on Immunization Practices. Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) in pregnant women and persons who have or anticipate having close contact with an infant aged < 12 months. MMWR. 2011;60(41)1424–6. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6041a4.htm.
World Health Organization. Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE), April 2014—conclusions and recommendations. Wkly Epidemiol Rec. 2014;89(21):221–36. http://www.who.int/wer/2014/wer8921.pdf.
Tan T, Trindade E, Skowronski D. Epidemiology of Pertussis. Pediatr Infect Dis J. 2005;24(5):S1–18.
Bamberger ES, Srugo I. What is new in pertussis? Eur J Pediatr. 2008;167:133–9. https://doi.org/10.1007/s00431-007-0548-2.
Gabutti G, Rota MC. Pertussis: a review of disease epidemiology worldwide and in Italy. Int J Environ Res Public Health. 2012;9:4626–38. http://www.mdpi.com/1660-4601/9/12/4626/htm.
Zamir CS, Dahan DB, Shoob H. Pertussis in infants under one year old: risk markers and vaccination status—A case-control study. Vaccine. 2015;33(17):2073–8.
Warfel JM, Zimmerman LI, and Merkel TJ. Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model. PNAS. 2014;111(2): 787–92. http://www.pnas.org/content/111/2/787.full.pdf.
Poland GA. Pertussis outbreaks and pertussis vaccines: new insights, new concerns, new recommendations? Vaccine. 2012;30:6957–9. http://www.sipps.it/pdf/newsfromlett/Newinsights.pdf.
Cherry JD. Pertussis: challenges today and for the future. PLoS Pathog. 2013;9(7):e1003418. https://doi.org/10.1371/journal.ppat.1003418. http://journals.plos.org/plospathogens/article/file?id=10.1371/journal.ppat.1003418&type=printable.
Rosenau MJ. Preventive medicine and hygiene. 6th ed. New York: Appleton-Century Company; 1916.
Disclaimer
The opinions expressed in this article are those of the authors alone and are not the opinions of the institutions with which they are associated.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anis, E., Moerman, L., Ginsberg, G. et al. Did two booster doses for schoolchildren change the epidemiology of pertussis in Israel?. J Public Health Pol 39, 304–317 (2018). https://doi.org/10.1057/s41271-018-0130-3
Published:
Issue Date:
DOI: https://doi.org/10.1057/s41271-018-0130-3