Abstract
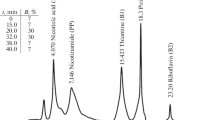
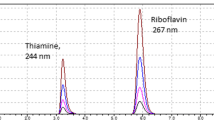
To overcome nutritional vitamin B12 deficiencies in certain populations in Southern Africa, fortified functional foods have been developed. However, current microbiological methods used to accurately determine vitamin B12 levels in foodstuffs, important for quality control and regulatory purposes, are time consuming. This study describes an extraction and detection method for vitamin B12 in dairy products and growth media cultured with Propionibacterium freudenreichii. Samples were extracted by mixing in KCN buffer (pH 4.5), autoclaving at 121 °C for 25 min, cooling and centrifugation. The resultant supernatant was syringe-filtered prior to reversed-phase HPLC analysis using a methanol-water gradient that was effective in resolving the B12 peak. The method offered excellent linearity with a regression coefficient R > 0.998. The limit of quantification was 0.005 μg·mL−1 sample. For samples with vitamin B12 concentrations well within the linear range of the assay, good repeatability was demonstrated for the same sample with mean concentrations of 2.62 ± 0.02 and 2.61 ± 0.02 μg·mL−1 detected on day 1 and day 2, respectively. Recovery values ranged from 98.6% to 103.2%, indicating that the extraction method ensured complete dissolution of vitamin B12 from the matrices under study. Sensitivity was enhanced by sample concentration and purification using a series of solid phase extraction steps which resulted in improved peak resolution and removal of interfering peaks. The method was validated by comparison of the HPLC results of the same sample with those obtained using traditional microbiological methods. The method is a rapid alternative to the more time-consuming microbiological assay.
Abstract
B12 B12 B12 (Propionibacterium freudenreichii) B12 KCN (pH 4.5) 121 °C 25 min, RP-HPLC R > 0.998 0.005 μg·mL−1 B12 2.62 ± 0.02 μg·mL−1 2.61 ± 0.02 μg·mL−1 98.6% ∼ 103.2%, B12 B12 B12
Résumé
Pour lutter contre les carences en vitamine B12 parmi certaines populations d’Afrique du Sud, des aliments fonctionnels enrichis ont été développés. Cependant, les méthodes actuelles utilisées pour analyser précisément le niveau de vitamine B12 dans les aliments, qui sont importantes à des fins de contrôle-qualité et de législation, sont coûteuses en temps. Cet article décrit une méthode d’extraction et de détection de la vitamine B12 dans les produits laitiers et les cultures de Propionibacterium freudenreichii. Les échantillons étaient extraits par mélange à du tampon KCN, pH 4,5 et autoclavage à 121 °C pendant 25 min, refroidissement et centrifugation. Le surnageant résultant était filtré puis analysé par HPLC en phase inverse en utilisant un gradient méthanol-eau, qui permettait une bonne résolution du pic de vitamine B12. La méthode montrait une bonne linéarité, avec un coefficient de régression R > 0,998. La limite de quantification était de 0,005 μg·mL−1 d’échantillon. Pour des échantillons contenant des concentrations dans la moyenne de la gamme de linéarité, la répétabilité a été établie, avec des valeurs obtenues pour le même échantillon à deux jours différents de 2,62 ± 0,02 μg·mL−1 et 2,61 ± 0,02 μg·mL−1. Les rendements d’extraction variaient entre 98,6–103,2 %, ce qui indique que la méthode d’extraction permettait une dissolution complète de la vitamine B12 des matrices étudiées. La méthode HPLC était validée en comparant les résultats obtenus avec ceux de la méthode microbiologique de référence. La sensibilité de la méthode était améliorée par une série d’étapes d’extraction en phase solide pour concentrer et purifier les échantillons, ce qui améliorait la résolution des pics et éliminait les pics contaminants. La méthode décrite est une alternative rapide à la méthode microbiologique de référence.
Article PDF
Similar content being viewed by others
References
Angyal G., Methods for the microbiological analysis of selected nutrients, AOAC International, Gaithersburg, Maryland, USA, 1996.
Anonymous, Vitamin B12 may slow AIDS, Tufts Univ. Health Nutr. Lett. 15 (1998) 2.
Anonymous, Regulations relating to the fortification of certain foodstuffs, G.N.R. 2003/2003, Foodstuffs, cosmetics and disinfectants act and regulations. Act no. 54 of 1972, Lex Patria Publishers, Johannesburg, South Africa, 2003.
Anonymous, Scientific opinion of the panel on biological hazards on a request from EFSA on the maintenance of the QPS list of microorganisms intentionally added to food or feed, EFSA J. 923 (2008) 1–48.
Babuchowski A., Laniewska-Moroz L., Warminska-Radyko I., Propionibacteria in fermented vegetables, Lait 79 (1999) 113–124.
Ball G.F.M., Vitamins in Foods, Analysis, Bioavailability and Stability, CRC Press, Boca Raton, FL, USA, 2006.
Britz T.J., Riedel K.-H.J., Propionibacterium species diversity in Leerdammer cheese, Int. J. Food Microbiol. 22 (1994) 257–267.
Celik M., Barkut I.K., Öncel C., Forta H., Involuntary movements associated with vitamin B12 deficiency, Parkinsonism Relat. Disord. 10 (2003) 55–57.
Cerná J., Hrabová H., Biologic enrichment of fermented milk beverages with vitamin B12 and folic acid, Milchwissenchaft 32 (1977) 274–277.
Dannhauser A., Joubert G., Nel M., Nutritional status of preschool children in the Bloemfontein district, South Afr. J. Food Sci. Nutr. 8 (1996) 14–22.
De Man J.M., Principles of food chemistry, 3rd edn., Aspen Publishers Inc., Gaithersburg, Maryland, USA, 1999.
Gao Y., Fuo F., Gokavi S., Chow A., Sheng Q., Guo M., Quantification of water-soluble vitamins in milk-based infant formulae using biosensor-based assays, Food Chem. 110 (2008) 769–776.
Gardner N., Champagne C.P., Production of Propionibacterium shermanii biomass and vitamin B12 on spent media, J. Appl. Microbiol. 99 (2005) 1365–2672.
Hepburn M.J., Dyal K., Runser L.A., Barfield R.L., Hepburn L.M., Fraser S.L., Low serum vitamin B12 levels in an outpatient HIV-infected population, Int. J. Sexually Transmitted Dis. & AIDS 15 (2004) 127–133.
Heudi O., Kilinç T., Fontannaz P., Marley E., Determination of vitamin B12 in food products and in premixes by reversed-phase high performance liquid chromatography and immunoaffinity extraction, J. Chromatogr. A 73 (2006) 63–68.
Hoffbrand V., Provan D., Macrocytic anaemias, Br. Med. J. 314 (1997) 430–433.
Kelly R.J., Detection of methyl cobalamin and adenosyl cobalamin using a nitroxide radical trap, Anal. Commun. 35 (1998) 257–259.
Labadarios D., The iron status of South African children 6–71 months of age: results of the national survey, 1994, South Afr. J. Food Sci. Nutr. 8 (1996) 10.
Lebiedzińska A., Marszałł M.L., Kuta J., Szefer P., Reversed-phase high-performance liquid chromatography method with coulometric electrochemical and ultraviolet detection for the quantification of vitamins B1 (thiamine), B6 (pyridoxamine, pyridoxal and pyridoxine) and B12 in animal and plant foods, J. Chromatogr. A 173 (2007) 71–80.
Li H.B., Chen F., Jiang Y., Determination of vitamin B12 in multivitamin tablets and fermentation medium by high-performance liquid chromatography with fluorescence detection, J. Chromatogr. A 891 (2000) 243–247.
Li K.-T., Liu D.-H., Li Y.-L., Chu J., Wang Y.-H., Zhuang Y.-P., Zhang S.-L., Improved large-scale production of vitamin B12 by Pseudomonas denitrificans with betaine feeding, Bioresour. Technol. 99 (2008) 8516–8520.
Markopoulou C.K., Kagkadis K.A., Koundourellis J.E., An optimized method for the simultaneous determination of vitamins B1, B6, B12, in multivitamin tables by high performance liquid chromatography, J. Pharm. Biomed. Anal. 30 (2002) 1403–1410.
Mathé G., The non-enumerable described retrovirus integrase inhibitors are not a lure, as evidenced by ten years of clinical experience, Biomed. Pharmacother. 54 (2000) 3–6.
Mazumder T.P., Nishio N., Fukuzaki S., Nagai S., Production of extracellular vitamin B12 compounds from methanol by Methanosarcina barkeri, Appl. Microbiol. Biotechnol. 26 (1987) 511–516.
Moreno P., Salvadó V., Determination of eight water- and fat-soluble vitamins in multivitamin pharmaceutical formulations by high-performance liquid chromatography, J. Chromatogr. A 870 (2000) 207–215.
Morita S., Miwa H., Kihira T., Kondo T., Cerebellar ataxia and leukoencephalopathy associated with cobalamin deficiency, J. Neurol. Sci. 216 (2003) 183–184.
Murooka Y., Piao Y., Kiatpapan P., Yamashita M., Production of tetrapyrrole compounds and vitamin B12 using genetically engineering of Propionibacterium freudenreichii. An overview, Lait 85 (2005) 9–22.
Ozbek Z., Kucukali C.I., Ozkok E., Orhan N., Aydin M., Kilic G., Sazci A., Kara I., Effect of the methylenetetrahydrofolate reductase gene polymorphisms on homocysteine, folate and vitamin B12 in patients with bipolar disorder and relatives, Prog. Neuropsychopharmacol. Biol. Psychiatry 32 (2008) 1331–1337.
Thiel R.J., Fowkes S.W., Down syndrome and epilepsy: a nutritional connection, Med. Hypotheses 62 (2004) 35–44.
Tucker K.L., Mahnken B., Wilson P.W.F., Jacques P., Selhub J., Folic acid fortification of the food supply. Potential risks and benefits for the elderly population, J. Am. Med. Assoc. 276 (1996) 1879–1885.
Vorobjeva L.I., Propionibacteria, Kluwer Academic Publishers, Dordrecht, The Netherlands, 1999.
Weinberg J.B., Sauls D.L., Misukonis M.A., Shugars D.C., Inhibition of productive Human Immunodeficiency Virus-1 infection by cobalamins, Blood 86 (1995) 1281–1287.
Xu H., Li Y., Liu C., Wu Q., Zhao Y., Lu L., Tang H., Fluorescence resonance energy transfer between acridine orange and rhodamine 6G and its analytical application for B12 with flow-injection laser-induced fluorescence detection, Talanta 77 (2008) 176–181.
Yang Y., Zhang Z., Lu J., Maekawa T., Continuous methane fermentation and the production of vitamin B12 in a fixed-bed reactor packed with loofah, Bioresour. Technol. 92 (2004) 285–290.
Ye K., Shijo M., Miyano K., Shimizu K., Metabolic pathway of Propionibacterium growing with oxygen: enzymes, 13C NMR analysis, and its application for vitamin B12 production with periodic fermentation, Biotechnol. Prog. 15 (1999) 201–207.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Van Wyk, J., Britz, T.J. A rapid HPLC method for the extraction and quantification of vitamin B12 in dairy products and cultures of Propionibacterium freudenreichii . Dairy Sci. Technol. 90, 509–520 (2010). https://doi.org/10.1051/dst/2009055
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1051/dst/2009055